Balancing Equations Balancing Writing and Naming Equations Balancing

Balancing Equations Balancing, Writing, and Naming Equations

Balancing Equations

Balancing Equations Law of Conservation of Matter: In a chemical reaction, matter can be neither created nor destroyed. In a chemical reaction, the amount of reactants equal the amount of products.

Balancing Equations Paraphrase: Law of Conservation of Atoms: The number of atoms of each type of element must be the same on each side of the equation.

Balancing Equations n Hydrogen n H 2 + oxygen + O 2 water H 2 O Hydrogen and oxygen are diatomic elements. Their subscripts cannot be changed. The subscripts on water cannot be changed.

Balancing Equation n H 2 + O 2 H 2 O Count the atoms on each side. Reactant side: 2 atoms H and 2 atoms O Product side: 2 atoms H and 1 atom O

Balancing Equations H 2 + O 2 H 2 O If the subscripts cannot be altered, how can the atoms be made equal? Adjust the number of molecules by changing the coefficients.

Balancing Equations n H 2 + O 2 2 H 2 O Reactants: 2 atoms of H and 2 atoms of O Products: 4 atoms of H and 2 atoms of O H is no longer balanced!

Balancing Equations n 2 H 2 + O 2 2 H 2 O Reactant side: 4 atoms of H and 2 atoms of O Product side: 4 atoms of H and 2 atoms of O It’s Balanced!
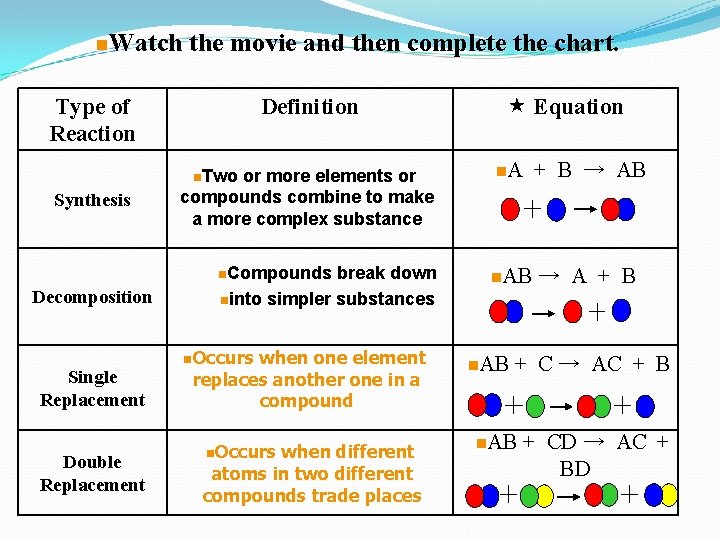
n. Watch the movie and then complete the chart. Type of Reaction Definition n. Two Synthesis or more elements or compounds combine to make a more complex substance n. Compounds Decomposition Single Replacement Double Replacement break down ninto simpler substances n. Occurs when one element replaces another one in a compound n. Occurs when different atoms in two different compounds trade places Equation n. A + B → AB n. AB → A + B n. AB + C → AC + B n. AB + CD → AC + BD

Balancing Equations n N 2 n + H 2 Nitrogen + hydrogen NH 3 ammonia Count atoms. Reactants: 2 atoms N and 2 atoms H Products: 1 atom N and 3 atoms of NH 3

Balancing Equations Nothing is balanced. Balance the nitrogen first by placing a coefficient of 2 in front of the NH 3. n N 2 + H 2 2 NH 3

Balancing Equations Hydrogen is not balanced. Place a 3 in front of H 2. Reactant side: 2 atoms N, 6 atoms H Product side: 2 atoms N, 6 atoms H n N 2 + 3 H 2 2 NH 3

Balancing Equations n Ca 3(PO 4)2 + H 2 SO 4 Ca. SO 4 + H 3 PO 4 Count atoms. Reactants: Ca – 3 atoms, P – 2 atoms, O – 8 atoms; H – atoms, S – 1 atom, O – 4 atoms

Balancing Equations Side note on Ca 3(PO 4)2 The subscript after the phosphate indicates two phosphate groups. This means two PO 43 - groups with two P and eight O atoms.

Balancing Equations Ca 3(PO 4)2 + H 2 SO 4 Ca. SO 4 + H 3 PO 4 Count atoms in the product. Ca atoms – 1, S atom – 1, O atoms – 4; H atoms – 3, P atom – 1, O atoms - 4

Balancing Equations In this equation, the ion groups do not break up. Instead of counting individual atoms, ion groups may be counted. Ca 3(PO 4)2 + H 2 SO 4 Ca. SO 4 + H 3 PO 4

Balancing Equations Ca 3(PO 4)2 + H 2 SO 4 Ca. SO 4 + H 3 PO 4 Reactants: Ca 2+ – 3, PO 43 - - 2, H+ – 2, SO 42+ - 1 Products: Ca 2+ - 1, SO 42 - - 1, H+ - 3, PO 43 - - 1

Balancing Equations Balance the metal first by placing a coefficient of 3 in front of Ca. SO 4. Products: Ca – 3 atoms, SO 42 - - 3 groups Ca 3(PO 4)2 + H 2 SO 4 3 Ca. SO 4 + H 3 PO 4

Balancing Equations Three sulfate groups are needed on the reactant side so place a coefficient of 3 in front of H 2 SO 4. 3 H 2 SO 4 gives 6 H+ and 3 SO 42 -. Neither phosphate nor calcium is balanced. Ca 3(PO 4)2 + 3 H 2 SO 4 3 Ca. SO 4 + H 3 PO 4

Balancing Equations A coefficient of 2 placed in front of H 3 PO 4 which balances both hydrogen and phosphate. Ca 3(PO 4)2 + 3 H 2 SO 4 3 Ca. SO 4 + 2 H 3 PO 4

Balancing Equations n n Cu + H 2 SO 4 Cu. SO 4 + H 2 O + SO 2 The sulfate group breaks up. Each atom must be counted individually. Ugh! Reactants: Cu – 1, H – 2, S – 1, O – 4 Products: Cu – 1, S – 1, O - 4, H – 2, O – 1, S – 1, O - 2

Balancing Equations Sulfur is not balanced. Place a two in front of sulfuric acid. Count atoms: 2 H 2 SO 4 H – 4, S – 2, O - 8 Cu + 2 H 2 SO 4 Cu. SO 4 + H 2 O + SO 2

Balancing Equations Hydrogen needs to be balanced so place a 2 in front of the H 2 O. Count the number of atoms. Cu + 2 H 2 SO 4 Cu. SO 4 + 2 H 2 O + SO 2

Balancing Equations Reactants: Cu – 1, H – 4, S – 2, O – 8 Products: Cu – 1, S – 1, O – 4, H – 4, O – 2, S – 1, O – 2 = Cu – 1, S – 2, H – 4, O – 8 It’s balanced! Cu + 2 H 2 SO 4 Cu. SO 4 + 2 H 2 O + SO 2

Balancing Equations Balancing hints: Balance the metals first. Balance the ion groups next. Balance the other atoms. Save the non ion group oxygen and hydrogen until the end.

Balancing Equations This method of balancing equations is the inspection method. The method is trial and error. Practice.

Writing and Naming n n Nickel + hydrochloric acid Nickel(II) chloride + hydrogen Write the corresponding formula equation and then balance the equation.

Writing and Naming Write each formula independently. Ignore the rest of the equation. Balance the equation after writing the formulas. Ni + HCl Ni. Cl 2 + H 2 Ni + 2 HCl Ni. Cl 2 + H 2 n n
Writing and Naming Remember the diatomic elements: H 2, N 2, O 2, F 2, Cl 2, Br 2, and I 2.

Writing and Naming Cu + H 2 SO 4 n Cu. SO 4 + H 2 O + SO 2 n Balance the formula equation. Write the word equation.

Writing and Naming Cu + 2 H 2 SO 4 Cu. SO 4 + 2 H 2 O + SO 2 Write the names: Cu by itself is just copper. Copper(I) or copper(II) would be incorrect. H 2 SO 4 should be named as an acid. Sulfuric acid

Writing and Naming Cu. SO 4 has a SO 42 - group so Cu must be 2+. Some metals must have Roman Numerals. Copper(II) sulfate H 2 O is known as water. SO 2 is a nonmetal compound. Its name is either sulfur dioxide or sulfur(IV) oxide.

Writing and Naming Cu + 2 H 2 SO 4 n Cu. SO 4 + 2 H 2 O + SO 2 n Copper + sulfuric acid Copper(II) sulfate + water + sulfur dioxide

Balancing Equations Chapter 8 Practice!
- Slides: 35