Balancing Equations Balancing Equations Balancing Equations n Law

Balancing Equations

Balancing Equations

Balancing Equations n Law of Conservation of Matter: n In a chemical reaction, matter can be neither created nor destroyed. n In a chemical reaction, the amount of reactants equal the amount of products.

Balancing Equations Paraphrased: n Law of Conservation of Atoms: n The number of atoms of each type of element must be the same on each side of the equation.

Chemical Equations

Balancing Chemical Equations n n Draw a reactant/ product Zn(s) + _HCl(aq) Zn. Cl (aq) + H (g) 2 2 table, listing each atom type. Count each atom on R P both sides of equation. Balance by trial & error Zn 1 1 (one atom at a time) until you have the same H 1 2 # of atoms of each element on both sides of Cl 1 2 the equation. Balance by adding coefficients, not subscripts.

Balancing Equations n Hydrogen n H 2 + oxygen + O 2 water H 2 O

Balancing Equations n N 2 n + H 2 Nitrogen + hydrogen NH 3 ammonia

Balancing Equations n Ca 3(PO 4)2 + H 2 SO 4 Ca. SO 4 + H 3 PO 4

Balancing Equations Cu + H 2 SO 4 n Cu. SO 4 + H 2 O + SO 2
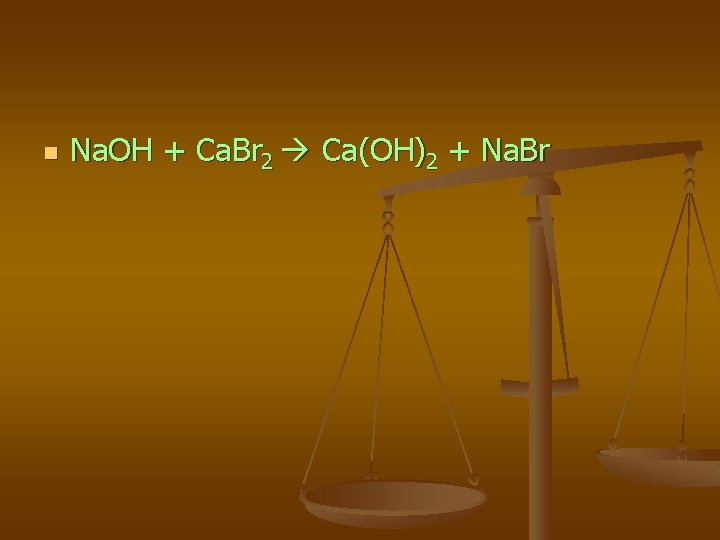
n Na. OH + Ca. Br 2 Ca(OH)2 + Na. Br
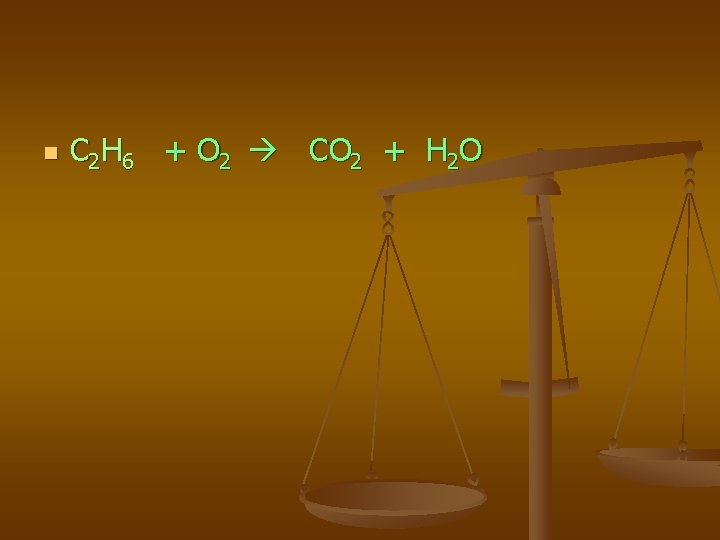
n C 2 H 6 + O 2 CO 2 + H 2 O

Key to Balancing Equations n Practice! n Trial & Error!!!

Balancing chemical equations _ Na + _Cl 2 → _ Na. Cl 2. _ Fe(s) + _ Cl 2(g) → _ Fe. Cl 3 3. _ Ag 2 O → _ Ag + _O 2 4. _ Cu. Cl 2 + _ Al → _ Al. Cl 3 + _ Cu 5. _ Ag. NO 3 + _Mg. Cl 2 → _ Ag. Cl + _Mg(NO 3)2 1.

Types of Chemical Rxns n Classifying reactions n There are 5 general types of reactions n n n Combustion Synthesis (Combination) Decomposition Single-replacement (single-displacement) Double-replacement (double-displacement) Not all chemical reactions fit uniquely into only one of these classes n Sometimes, one rxn may fit 2 or 3 different types
- Slides: 15