Balancing equations Atoms must be conserved Counting Atoms

Balancing equations Atoms must be conserved!
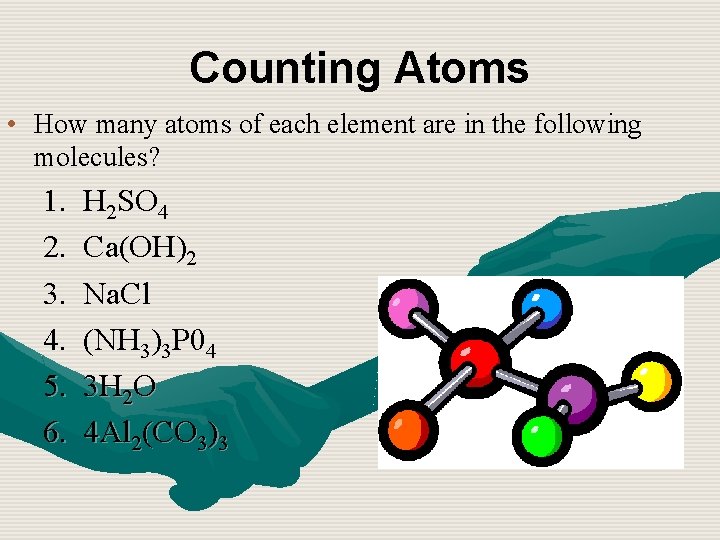
Counting Atoms • How many atoms of each element are in the following molecules? 1. 2. 3. 4. 5. 6. H 2 SO 4 Ca(OH)2 Na. Cl (NH 3)3 P 04 3 H 2 O 4 Al 2(CO 3)3

Answers n 1. 2 hydrogen, 1 sulfur, 4 oxygen n 2. 1 calcium, 2 oxygen, 2 hydrogen n 3. 1 sodium, 1 chlorine n 4. 3 nitrogen, 9 hydrogen, 1 phosphorus, 4 oxygen n 5. 6 hydrogen, 3 oxygen n 6. 8 aluminum, 12 carbon, 36 oxygen

Balancing Equations n Law of Conservation of Matter: n In a chemical reaction, matter can be neither created nor destroyed. n But merely changed from one form to another. n

Is this equation balanced? • Na. Cl + H 2 O Na. OH + Cl 2 • left side has 1 Na, 1 Cl, 2 H, and 1 O • right side has 1 Na, 1 O, 1 H, and 2 Cl • NO!!!!!! It is not balanced!!

Is this equation balanced? • HCl + Na. OH Na. Cl+ H 2 O • left side has 2 H, 1 Cl, 1 Na, and 1 O • right side has 1 Na, 1 Cl, 2 H, and 1 O • Yes!!!!!! It is balanced!!

Is this equation balanced? • Ca + H 2 O Ca(OH)2 + H 2 • left side has 1 Ca, 2 H, 1 O • right side has 1 Ca, 2 O, 4 H • No!!!!!! It is not balanced!! • How can we make it balance? ?

Ca + H 2 O Ca(OH)2 + H 2 n We need more H’s, but we can’t add just the H atom, we have to add the entire molecule the H is attached to: Ca + H 2 O Ca(OH)2 + H 2 O Count the boxes. These are the coefficients: 1 Ca + 2 H 2 O 1 Ca(OH)2 + 1 H 2

1 Ca + 2 H 2 O 1 Ca(OH)2 + 1 H 2 n Just like in math, this equation may be written in a simpler way: n Ca + 2 H 2 O Ca(OH)2 + H 2 (be sure you write the “ 2” on your worksheet!)

What is balanced in a chemical equation? Three things are balanced in a chemical equation: 1. Atoms 2. Mass 3. Charge

• For now, you will only be concerned with balancing atoms. Remember, the number of atoms of each element on the reactants side must equal the number of atoms of each element on the products side!

The End l Let’s practice!
- Slides: 12