Balancing equation Chemical equation Word equation potassium water

Balancing equation

Chemical equation Word equation: potassium + water potassium hydroxide solution + hydrogen Chemical equation: reactants K (s) + H 2 O (l) products KOH (aq) + H 2(g) How many H in the reactants? 2 Is it equal to the number of H in the products? No There are 3 H in the products

What can we do? Balance it! Chemical equation: K (s) + H 2 O (l) KOH (aq) + H 2(g) 2 H(Hydrogen) in reactants We can’t change to H 3 O 3 H (Hydrogen) in the products

What can we do? Balance it! Chemical equation: K (s) + H 2 O (l) 2 H(Hydrogen) in reactants 2 KOH (aq) + H 2(g) 3 H (Hydrogen) in the products We can’t change to KOH 2, but we can multiple KOH by 2

What can we do? Balance it! Chemical equation: K (s) + H 2 O (l) 2 H(Hydrogen) in reactants 2 KOH (aq) + H 2(g) 4 H (Hydrogen) in the products

What can we do? Balance it! Chemical equation: K (s) + 2 H 2 O (l) 2 KOH 2 H(Hydrogen) in reactants multiple by 2 in the beginning (aq) + H 2(g) 4 H (Hydrogen) in the products

What can we do? Balance it! Chemical equation: K (s) + 2 H 2 O (l) 2 KOH (aq) + H 2(g) 4 H(Hydrogen) in reactants 4 H (Hydrogen) in the products YEAH~~~Hydrogen is now balanced~~ Oxygen is now balanced~~~

What can we do? Balance it! Chemical equation: 2 K (s) + 2 H 2 O (l) 2 KOH (aq) + H 2(g) Don’t worry~ Write 2 in front of K, it will be balanced~

What can we do? Balance it! Chemical equation: 2 K (s) + 2 H 2 O (l) 2 KOH (aq) + H 2(g)
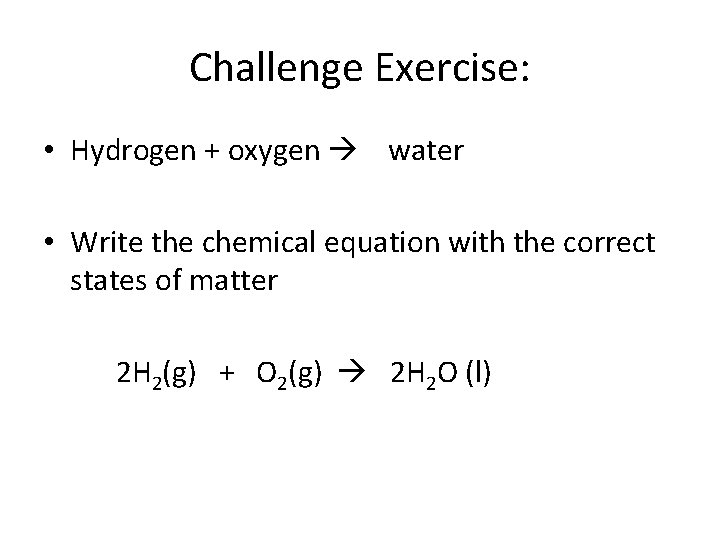
Challenge Exercise: • Hydrogen + oxygen water • Write the chemical equation with the correct states of matter 2 H 2(g) + O 2(g) 2 H 2 O (l)
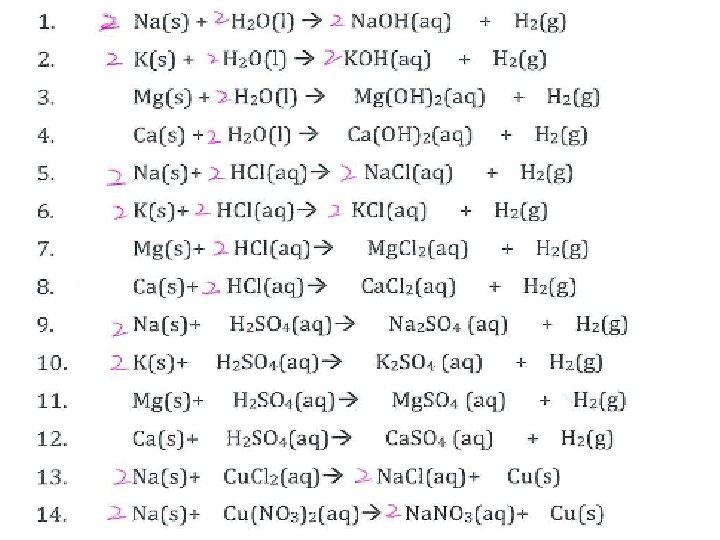


Common ions Cations Li+ Na+ K+ Mg 2+ Ca 2+ Al 3+ Group III Common metals: Ag+ Cu 2+ Fe 2+ Zn 2+ Pb 2+

Anions F- Cl- Br- I- Group VII O 2 Group VI Polyatomic Ion: OH- (hydroxide) NO 3 - (nitrate) CO 32 - (carbonate) SO 42 - (sulphate)
- Slides: 14