Balancing Chemical Equations What goes in must come

Balancing Chemical Equations What goes in must come out!

Law of Conservation of Mass You need to remember this law! The Law of Conservation of Mass states: that mass is neither created nor destroyed in any chemical reaction. Therefore balancing of equations requires the same number of atoms on both sides of a chemical reaction. The number of atoms in the Reactants must equal the Number of atoms in the Products The mass of all the reactants (the substances going into a reaction) must equal the mass of the products (the substances produced by the reaction). Reactant + Reactant = Product REACTANTS - PRODUCTS, Reactants on left, products on right

Chemical Equations Because of the principle of the Conservation of Matter, an equation must be balanced. It must have the same of atoms of the both sides. https: //www. youtube. com/watch? v=2 S 6 e 11 NBwiw number same kind on Lavoisier, 1788

Symbols in Reactions Symbol Explanation “yields” ; indicates result of a reaction Used in place of a single arrow to indicate a reversible reaction (s) Reactant or product in the solid state. Also a precipitate (ppt) Precipitate formed in a reaction (l) Reactant or product in a pure liquid state, H 2 O. (aq) Reactant or product in an aqueous solution (dissolved in water) (g) Reactant or product in the gaseous state ∆ Reactants are heated

A simple equation, such as the synthesis of Iron (II) sulfide, iron + sulfur Iron (II) sulfide Fe + S Fe. S Note that in a chemical equation, we use the arrow “ " instead of the equals “ = ". The last stage is to put in state of matter symbols, (s, l, g, aq), as appropriate (solid, liquid, gas, aqueous or dissolved in water) Fe(s) + S(s) Fe. S(s) TIP: BALANCED EQUATIONS WILL ALMOST ALWAYS HAVE AN ODD NUMBER FOR ONE OF THE COEFFICIENTS. MUST REDUCE IF POSSIBLE.

Steps to Balancing a Chemical Equation 1. Write all reactants on the 3. Multiply the coefficient of each element by the subscript of the element to left and all products on the count the atoms. Then list the number right side of the equation of atoms of each element on each side. arrow. Make sure you write 4. It is often easiest to start balancing with the correct formula for an element that appears only once on each element each side of the arrow. These elements must have the same 2. Use coefficients in front coefficient. Next balance elements of each formula to balance that appear only once on each side but the number of atoms on have different numbers of atoms. Finally balance elements that are in each side. two formulas in the same side.

Balancing Equations 2 3 2 ___ Al(s) + ___ Br (l) ---> ___ Al. Br (s) 2 3

Re-cap of steps from rule 4: Balance elements that appear only once on each side of the arrow. Next balance elements that appear only once on each side but have different numbers of atoms. Finally balance elements that are in two formulas in the same side.

CHART METHOD FOR BALANCING EQUATIONS Just like this one Mg + O 2 Mg. O Then all you do is list the atoms that are involved on each side of the arrow Mg + O 2 Mg. O Mg O
![Then start balancing: [1] Just count up the atoms on each side Mg + Then start balancing: [1] Just count up the atoms on each side Mg +](http://slidetodoc.com/presentation_image_h2/ae0cb88c07662a2195f9f6c93c49ab70/image-10.jpg)
Then start balancing: [1] Just count up the atoms on each side Mg + O 2 Mg. O 1 Mg 1 2 O 1 [2] The numbers aren’t balanced so then add “BIG” numbers to make up for any shortages Mg + O 2 2 Mg. O And adjust totals 1 Mg 1 2 2 O 1 2

But the numbers still aren’t equal, so add another “BIG” number 2 Mg + O 2 2 Mg. O 2 1 Mg 2 2 O 2 And adjust totals again NOW BOTH SIDES HAVE EQUAL NUMBERS OF ATOMS WE SAY THAT THE EQUATION IS BALANCED!!

Example: NH 3 + O 2 NO + H 2 O Reactants Products N appears once on both sides in equal numbers, so the coefficient for NH 3 is the same as for NO. Next look at H which appears only once on each side but has different numbers of atoms, 3 on the left and 2 on the right. The least common multiple of 3 and 2 is 6, so rewrite the equation to get 6 atoms of H on both sides: 2 NH 3 + O 2 NO + 3 H 2 O http: //www. youtube. com/watch? v=Rn. Gu 3 x. O 2 h 74 khan balance 14. 5 http: //www. youtube. com/watch? v=_B 735 tur. Do. M Bozeman begin bal 9. 3

Count the atoms on each side: NH 3 + O 2 NO + H 2 O Write them out keeping them on the appropriate side of the chemical equation 1 N (nitrogen atoms) 3 H (hydrogen atoms) 2 H (hydrogen atoms) 2 O (oxygen atoms) This shows the equation not to be balanced “YET” NOTE: find a common factor of 2 and 3 NOTE: add all the O’s on product side

Double-check: NH 3 + O 2 N (nitrogen atoms) H (hydrogen atoms) O (oxygen atoms) NO + H 2 O N (nitrogen atoms) H (hydrogen atoms) O (oxygen atoms)

Try to balance these equations using the same method: Na + Cl 2 Na. Cl 2 Na + Cl 2 2 Na. Cl CH 4 + O 2 CO 2 + H 2 O CH 4 + 2 O 2 CO 2 + 2 H 2 O Li + HNO 3 Li. NO 3 + H 2 2 Li + 2 HNO 3 2 Li. NO 3 + H 2 Al + O 2 Al 2 O 3 4 Al + 3 O 2 2 Al 2 O 3 Tip: Balance polyatomic ions as if they were single atoms.

TYPES OF REACTIONS SYNTHESIS DECOMPOSITION SINGLE REPLACEMENT DOUBLE REPLACEMENT AD + CB

TYPES OF REACTIONS COMBUSTION Cx. Hx + O 2 CO 2 + H 2 O A hydrocarbon, Cx. Hx burns in the presence of O 2 to produce carbon dioxide and water. Hydrocarbons include: candles, gasoline, propane, butane, methane, acetylene….

TYPES OF REACTIONS Synthesis two or more elements or compounds may combine to form a more complex compound. Basic form: A + X → AX Decomposition: A single compound breaks down into its component parts or simpler compounds. Basic form: AX → A + X

TYPES OF REACTIONS Single Replacement: a more active element takes the place of another element in a compound and sets the less active one free. Basic form: A + BX → AX + B or AX + Y → AY + X Double Replacement: Two positive ions switch places. Basic form: AB + CD AD + CB

TYPES OF REACTIONS Neutralization: A double replacement reaction between an acid and a base. HCl + Na. OH H 2 O + Na. Cl The products of a neutralization reaction are salt and water.

TYPES OF REACTIONS List what type the following reactions are: 1) Na. OH + KNO 3 --> Na. NO 3 + KOH DR 2) CH 4 + 2 O 2 --> CO 2 + 2 H 2 O COMBUSTION 3) 2 Fe + 6 Na. Br --> 2 Fe. Br 3 + 6 Na SR 4) Ca. SO 4 + Mg(OH)2 --> Ca(OH)2 + Mg. SO 4 DR 5) NH 4 OH + HBr --> H 2 O + NH 4 Br DR NEUTRALIZATION 6) Pb + O 2 --> Pb. O 2 SYNTHESIS 7) Na 2 CO 3 --> Na 2 O + CO 2 DECOMPOSITION https: //www. youtube. com/watch? v=sin. Q 06 Yzb. JI

TYPES OF REACTIONS List what type the following reactions are: 1) Na. OH + KNO 3 --> Na. NO 3 + KOH DR 2) CH 4 + 2 O 2 --> CO 2 + 2 H 2 O COMBUSTION 3) 2 Fe + 6 Na. Br --> 2 Fe. Br 3 + 6 Na SR 4) Ca. SO 4 + Mg(OH)2 --> Ca(OH)2 + Mg. SO 4 DR 5) NH 4 OH + HBr --> H 2 O + NH 4 Br DR NEUTRALIZATION 6) Pb + O 2 --> Pb. O 2 SYNTHESIS 7) Na 2 CO 3 --> Na 2 O + CO 2 DECOMPOSITION

PREDICTING PRODUCTS pg. 30 CTP DETERMINE THE PRODUCTS FROM THE REACTANTS SINGLE REPLACEMENT REACTIONS METALS CHANGE PLACES Ex: K + Zn(NO 3)2 Zn + 2 KNO 3 Use the Activity series to determine if the reaction takes place. The single metal or element must be higher than the one in the compound for the reaction to occur. TRY THESE: Al + Pb(NO 3)2 --> 2 Al + 3 Pb(NO 3)2 --> 2 Al(NO 3)3 + 3 Pb K + Fe. SO 4 2 K + Fe. SO 4 K 2 SO 4 + Fe Ag + Ca. Br 2 no reaction Zn + Sn. C 2 O 4 Zn. C 2 O 4 + Sn
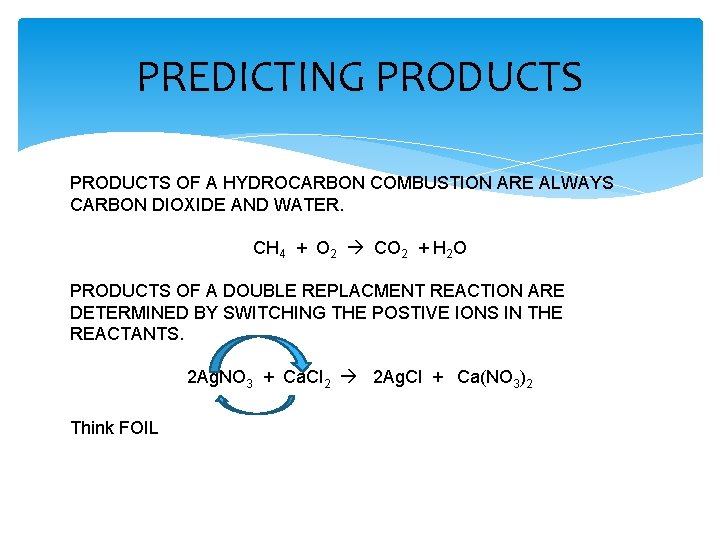
PREDICTING PRODUCTS OF A HYDROCARBON COMBUSTION ARE ALWAYS CARBON DIOXIDE AND WATER. CH 4 + O 2 CO 2 + H 2 O PRODUCTS OF A DOUBLE REPLACMENT REACTION ARE DETERMINED BY SWITCHING THE POSTIVE IONS IN THE REACTANTS. 2 Ag. NO 3 + Ca. Cl 2 2 Ag. Cl + Ca(NO 3)2 Think FOIL

SOLUBILITY RULES pg. 28 -30 in CTP SOLUBILITY REFERS TO THE ABILITY OF A COMPOUND TO STAY SOLUBLE IN SOLUTION OR FORM A PRECIPITATE. SOLUBILITY RULES AND CHARTS CAN BE USED TO DETERMINE THE STATES OF MATTER OF REACTANT AND PRODUCTS. Read the chart: S= soluble which means aq. I = insoluble which means ppt. s= partially soluble which means ppt. Ag. NO 3(aq) + Na. Cl(aq) Ag. Cl(ppt) + Na. NO 3(aq)

SOLUBILITY RULES TRY THESE: WRITE PRODUCTS, BALANCE, AND ALL STATES OF MATTER Ag. NO 3(aq)+ Li. OH(aq) Ag. OH(ppt) + Li. NO 3 (aq) Al(NO 3)3(aq) + H 2 SO 4(aq) Al 2(SO 4)3 + HNO 3 (NH 4) 3 PO 4(aq) + Ba(OH)2(aq) Ba 3(PO 4)2 + NH 4 OH NH 4) 3 PO 4(aq) + Ba(OH)2(aq) Ba 3(PO 4)2(ppt) + NH 4 OH(aq)

SOLUBILITY RULES Ba. Cl 2(aq)+ Na 2 SO 4(aq) Ba. SO 4 (ppt) + 2 Na. Cl (aq) Ca. SO 4(aq) + Mg(OH)2(aq) --> Ca(OH)2(ppt) + Mg. SO 4 (aq)

TYPES OF REACTIONS List what type the following reactions are: 1) Na. OH + KNO 3 --> Na. NO 3 + KOH 2) CH 4 + 2 O 2 --> CO 2 + 2 H 2 O 3) 2 Fe + 6 Na. Br --> 2 Fe. Br 3 + 6 Na 4) Ca. SO 4 + Mg(OH)2 --> Ca(OH)2 + Mg. SO 4 5) NH 4 OH + HBr --> H 2 O + NH 4 Br 6) Pb + O 2 --> Pb. O 2 7) Na 2 CO 3 --> Na 2 O + CO 2



- Slides: 31