Balancing Chemical Equations What goes in must come

Balancing Chemical Equations What goes in must come out!

Standard SPS 2. Students will explore the nature of matter, its classifications, and its system for naming types of matter. e. Apply the Law of Conservation of Matter by balancing the following types of chemical equations: l Synthesis l Decomposition l Single Replacement l Double Replacement

Essential Questions How do you balance chemical equations? How do you know if they are balanced? Where is a good place to start when you are balancing chemical equations?

Learning Targets l You should know how to balance chemical reactions You should know how to check your work & know what the Law of Conservation of Matter is

Balancing Chemical Equations l Balancing a chemical equation is much like the work of an accountant who has to show every penny that comes in and where it has gone to.

Objectives l l Learn the steps to balancing chemical equations. Take notes to help you understand. Test yourself with a set of equations to balance. Enter your own equations to see if they balance.

Law of Conservation of Mass You need to remember this law! l l The Law of Conservation of Mass states: that mass is neither created nor destroyed in any chemical reaction. Therefore balancing of equations requires the same number and types of atoms on both sides of a chemical reaction. The number of atoms in the Reactants must equal the Number of atoms in the Products

Chemical Equations Because of the principle of the Conservation of Matter, an equation must be balanced. It must have the same number of atoms of the same kind on both sides. Lavoisier, 1788

Law of Conservation of Mass l The mass of all the reactants (the substances going into a reaction) must equal the mass of the products (the substances produced by the reaction). l Reactant + Reactant = Product

A simple equation, such as the synthesis of Iron (II) sulfide, l l l iron + sulfur Fe + S Iron (II) sulfide Fe. S Note that in a chemical equation, by convention, we use the arrow “ " instead of the equals “ = ".

l The last stage is to put in state of matter symbols, (s, l, g, aq), as appropriate (solid, liquid, gas, aqueous or dissolved in water) l Fe(s) + S(s) Fe. S(s)
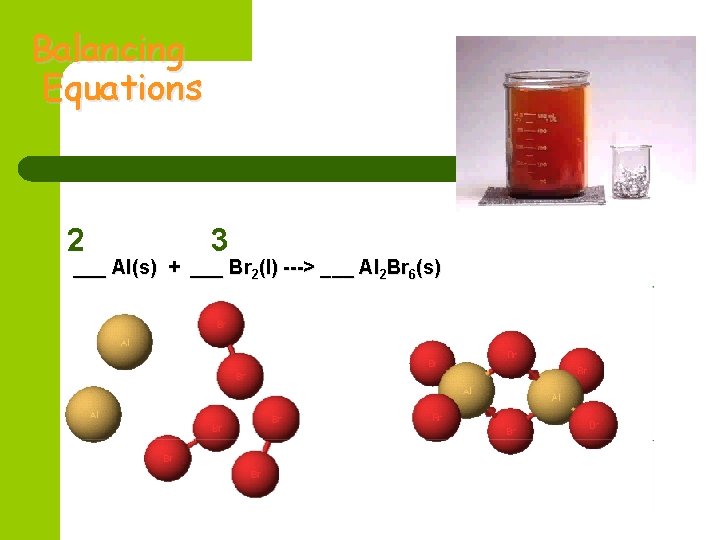
Balancing Equations 2 3 ___ Al(s) + ___ Br 2(l) ---> ___ Al 2 Br 6(s)

Steps to Balancing a Chemical Equation 1. Write all reactants 2. Use coefficients in front of each formula on the left and all to balance the products on the number of atoms on right side of the each side. equation arrow. In the equation 2 Li the Make sure you 2 is a coefficient. write the correct In the equation H 2 O formula for each the 2 is a subscript. element.

Remember! You can change Coefficients but never subscripts!

Steps to Balancing a Chemical Equation 3. Multiply the coefficient of each element by the subscript of the element to count the atoms. Then list the number of atoms of each element on each side. 4. It is often easiest to start balancing with an element that appears only once on each side of the arrow. These elements must have the same coefficient. Next balance elements that appear only once on each side but have different numbers of atoms. Finally balance elements that are in two formulas in the same side.

Re-cap of steps from rule 4: Balance elements that appear only once on each side of the arrow. l Next balance elements that appear only once on each side but have different numbers of atoms. l Finally balance elements that are in two formulas in the same side. l

Balancing Chemical Equations An easier way

First you need an equation with the correct “formula” ………. You’ll probably be given this in the question Just like this one Mg + O 2 Mg. O Then all you do is list the atoms that are involved on each side of the arrow Mg + O 2 Mg. O Mg O
![Then start balancing: [1] Just count up the atoms on each side Mg + Then start balancing: [1] Just count up the atoms on each side Mg +](http://slidetodoc.com/presentation_image_h2/a7ab4dc93f905233cbe6a16f88d4c617/image-19.jpg)
Then start balancing: [1] Just count up the atoms on each side Mg + O 2 Mg. O 1 Mg 1 2 O 1 [2] The numbers aren’t balanced so then add “BIG” numbers to make up for any shortages Mg + O 2 2 Mg. O And adjust totals 1 Mg 1 2 2 O 1 2

But the numbers still aren’t equal, so add another “BIG” number 2 Mg + O 2 2 Mg. O 2 1 Mg 2 2 O 2 And adjust totals again NOW BOTH SIDES HAVE EQUAL NUMBERS OF ATOMS WE SAY THAT THE EQUATION IS BALANCED!!
![Try to balance these equations using the same method: [1] Na + Cl 2 Try to balance these equations using the same method: [1] Na + Cl 2](http://slidetodoc.com/presentation_image_h2/a7ab4dc93f905233cbe6a16f88d4c617/image-21.jpg)
Try to balance these equations using the same method: [1] Na + Cl 2 Na. Cl [2] CH 4 + O 2 CO 2 + H 2 O [3] Li + HNO 3 Li. NO 3 + H 2 [4] Al + O 2 Al 2 O 3
![How did you get on? ? Here are the answers: [1] 2 Na + How did you get on? ? Here are the answers: [1] 2 Na +](http://slidetodoc.com/presentation_image_h2/a7ab4dc93f905233cbe6a16f88d4c617/image-22.jpg)
How did you get on? ? Here are the answers: [1] 2 Na + Cl 2 2 Na. Cl [2] CH 4 + 2 O 2 CO 2 + 2 H 2 O [3] 2 Li + 2 HNO 3 2 Li. NO 3 + H 2 [4] 4 Al + 3 O 2 2 Al 2 O 3 HOPE YOU’VE GOT THE IDEA… REMEMBER TO CHECK THAT YOU CAN DO ELECTROLYSIS EQUATIONS TOO
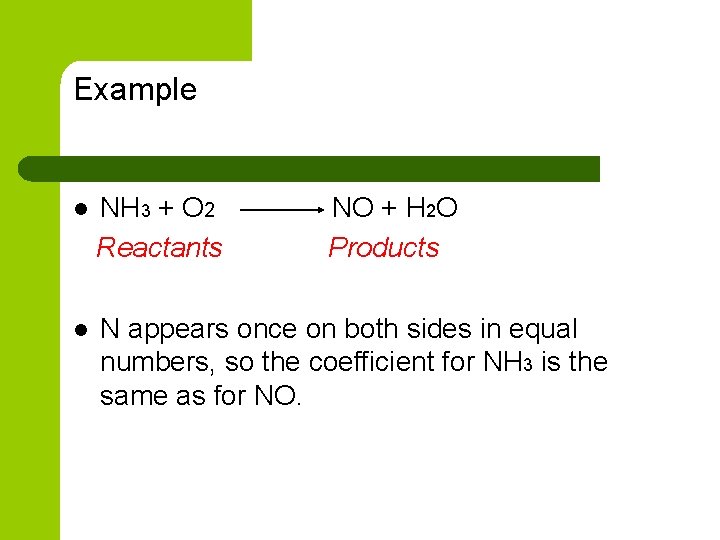
Example l NH 3 + O 2 Reactants NO + H 2 O Products l N appears once on both sides in equal numbers, so the coefficient for NH 3 is the same as for NO.
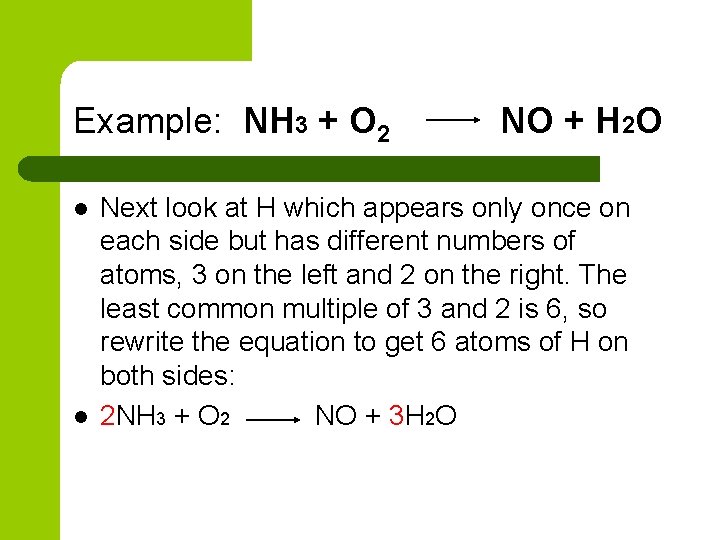
Example: NH 3 + O 2 l l NO + H 2 O Next look at H which appears only once on each side but has different numbers of atoms, 3 on the left and 2 on the right. The least common multiple of 3 and 2 is 6, so rewrite the equation to get 6 atoms of H on both sides: 2 NH 3 + O 2 NO + 3 H 2 O
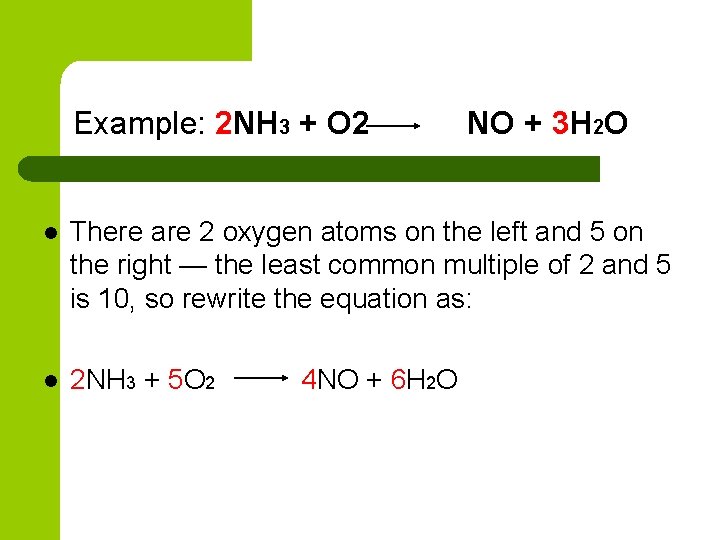
Example: 2 NH 3 + O 2 NO + 3 H 2 O l There are 2 oxygen atoms on the left and 5 on the right — the least common multiple of 2 and 5 is 10, so rewrite the equation as: l 2 NH 3 + 5 O 2 4 NO + 6 H 2 O

Now count the atoms on each side: l l l 2 NH 3 + 5 O 2 4 NO + 6 H 2 O Write them out keeping them on the appropriate side of the chemical equation 2 N (nitrogen atoms) 4 N (nitrogen atoms) 6 H (hydrogen atoms) 12 H (hydrogen atoms) 10 O (oxygen atoms) This shows the equation not to be balanced “YET”

Check the number again: l If you double the N and H on the left the equation will be balanced: l 4 NH 3 + 5 O 2 4 NO + 6 H 2 O
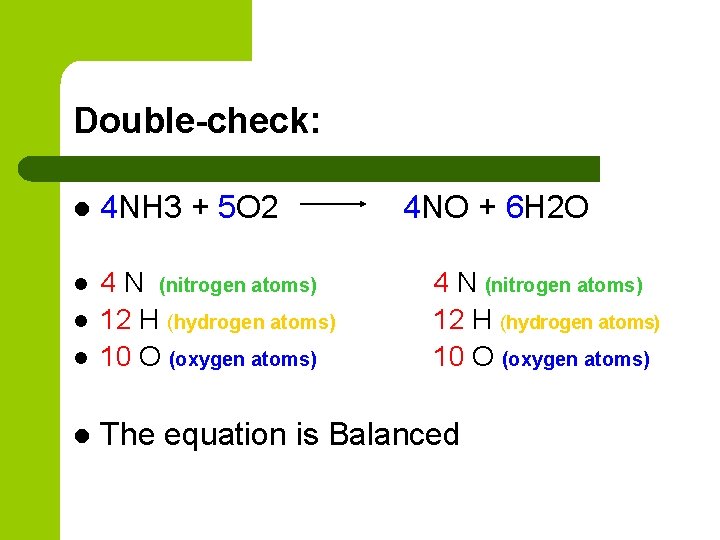
Double-check: l 4 NH 3 + 5 O 2 l l 4 N (nitrogen atoms) 12 H (hydrogen atoms) 10 O (oxygen atoms) l The equation is Balanced l 4 NO + 6 H 2 O 4 N (nitrogen atoms) 12 H (hydrogen atoms) 10 O (oxygen atoms)

Balancing Practice n For more help go to: http: //richardbowles. tripod. com/chemistry/bal ance. htm#part 0 n For some fun balancing equations go to: n http: //www. mpcfaculty. net/mark_bishop/bala ncing_equations_tutorial. htm
- Slides: 29