Balancing Chemical Equations What Do You See Look

Balancing Chemical Equations

What Do You See? Look closely! n When the pans are both at the same height, what does that mean if you have masses on both pans? n Keep this in mind as we continue n

What Does the Law State n n The law of conservation of matter states that matter (mass) can neither be created nor destroyed. It can, however, can be rearranged. In a chemical reaction, the mass of the reactants must equal the mass of the products.
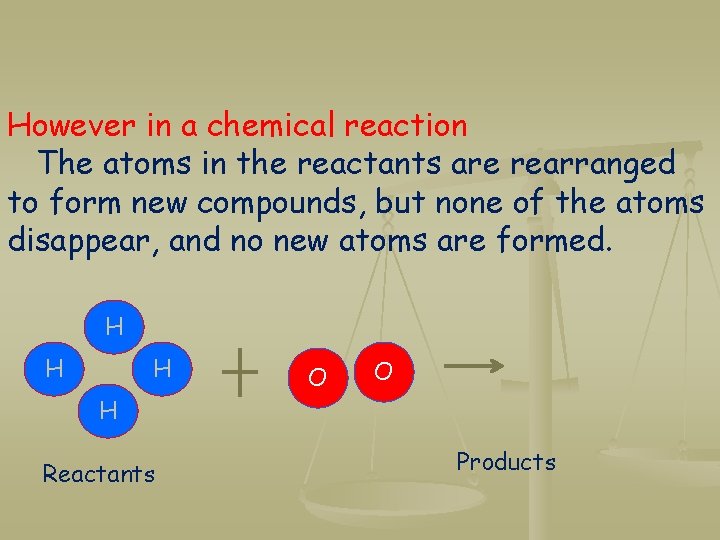
However in a chemical reaction The atoms in the reactants are rearranged to form new compounds, but none of the atoms disappear, and no new atoms are formed. H H Reactants O O Products
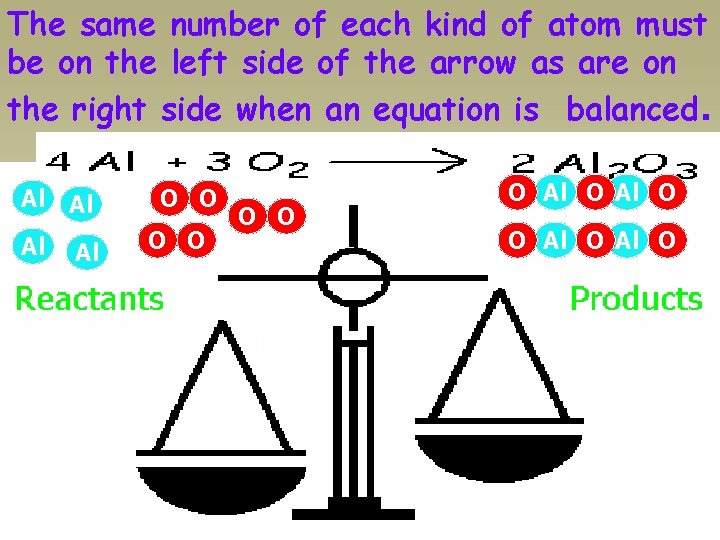
The same number of each kind of atom must be on the left side of the arrow as are on the right side when an equation is balanced. Al Al O O O O Al O
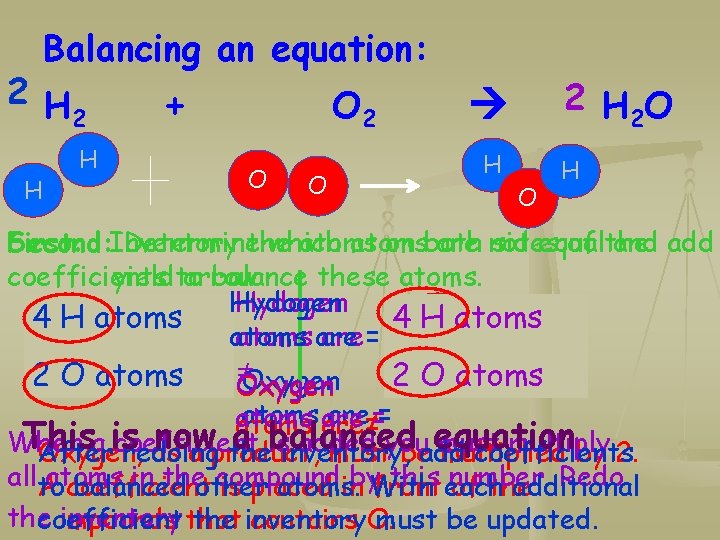
Balancing an equation: 2 H 2 H H + O 2 O O 2 H 2 O H O H Second: Inventory First: Determinethe which atoms on both are not sides equal of the and add coefficients yieldto arrow balance these atoms. Hydogen 4 H 2 atoms Hydogen 42 HH atoms are = 2 O 2 atoms O atoms ≠ 21 OO atoms atom Oxygen atoms are≠== atoms are This now the a is balanced equation When a is coefficient added must multiply After redoing inventory, coefficients Oxygen, as a product, mustyou beadd multiplied by 2. allto in theother compound by. With this each number. Redo balanced atoms. additional Aatoms coefficient is placed in front of the inventory that coefficient the inventory must be updated. compounds contains O.
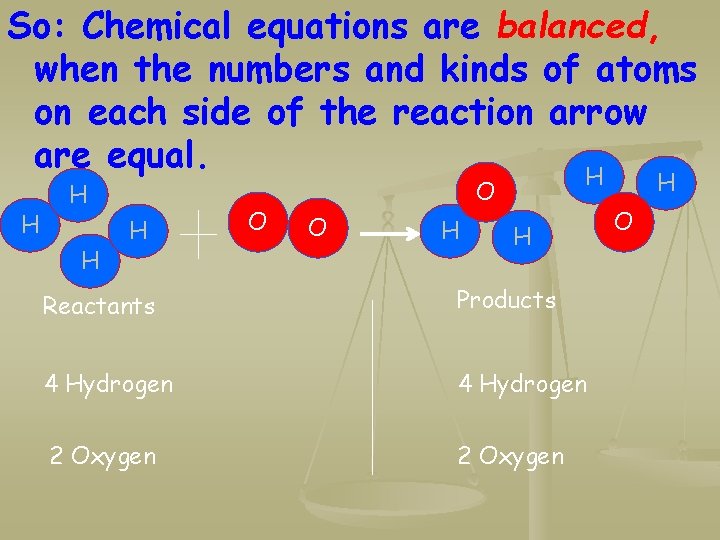
So: Chemical equations are balanced, when the numbers and kinds of atoms on each side of the reaction arrow are equal. H H O H O O H H Reactants Products 4 Hydrogen 2 Oxygen H O

Because atoms are conserved, the mass of the reactants in the chemical reaction is equal to the mass of the products. H 2 + F 2 H H F 2 g + 38 g 40 g H F = = 2 HF F H 2( 20 g) 40 g Reactants Products 40 g F

Law of Conservation of Matter (Mass) n If the amount of matter (mass) was not the same before and after the change (remember, it is the law), you must offer an explanation as to why.

Just because you cannot see it… Sometimes in chemical reactions mass is converted to a gas and/or energy that it is released as a product.

Background research st n 1 paragraph – describe 1 variable n 2 nd paragraph – describe nd 2 variable rd n 3 paragraph – What is your hypothesis and why
- Slides: 11