Balancing Chemical Equations Vocabulary chemical equation reversible chemical

Balancing Chemical Equations Vocabulary: *chemical equation *reversible *chemical reaction *conservation of mass *coefficient *yields *chemical change *products *reactants *skeleton equation *symbolic equation *subscript *aqueous *physical change
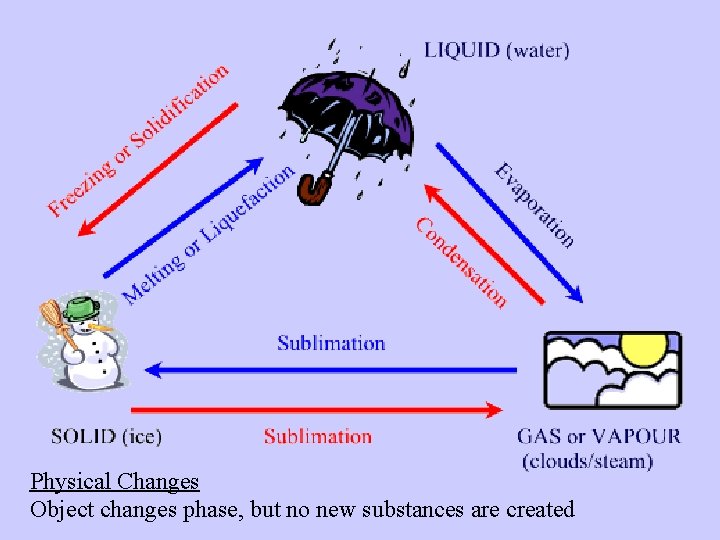
Physical Changes Object changes phase, but no new substances are created

• Chemical reactions result in chemical changes. – Chemical changes occur when new substances are created. – The original substance(s), called reactants, change into new substance(s) called products.

• Chemical reactions can be written in different ways. – A word equation: Nitrogen monoxide + oxygen nitrogen dioxide – A symbolic equation: State of matter 2 NO(g) + O 2(g) 2 NO 2(g) - Letters indicate the state of each compound Coefficients - indicate the ratio of compounds in the reaction - here, there is twice as much NO and NO 2 than there is O 2 (aq) =aqueous/ mixed with water (s) = solid (l) = liquid (g) = gas See pages 202 - 203

Conservation of Mass in Chemical Change • Chemical change means new compounds are created. – BUT no new atoms are created or destroyed; atoms are just rearranged. – All of the matter in the reactants = all of the matter in the products – Number of each atom in reactants = number of each atom in products

The Law of Conservation of Mass § In chemical reactions, atoms are neither created nor destroyed § Mass of reactants = Mass of products § Number of atoms (reactants side) = Number of atoms (product side)

Writing and Balancing Chemical Equations • The simplest form of chemical equation is a word equation Potassium metal + oxygen gas potassium oxide

Writing and Balancing Chemical Equations • A skeleton equation shows only the formulas of the elements/compounds – Shows atoms, but not how many atoms • K(s) + O 2 (g) K 2 O(s) • A balanced chemical equation shows all atoms and their how many there are – Balancing makes the number of each atom the same on both sides of the reaction arrow – Always use the smallest whole number ratio • 4 K(s) + O 2 (g) 2 K 2 O(s)

Counting Atoms to Balance an Equation • Because of the Law of Conservation of Mass, we can count atoms and use math to balance the number of atoms in chemical equations. – Word equation: Methane + oxygen water + carbon dioxide – Skeleton equation: CH 4(g) + O 2 (g) H 2 O(l) + CO 2(g) • To balance the compounds, take note of how many atoms of each element occur on each side of the reaction arrow. • 1 Carbon, 4 Hydrogen, 2 Oxygen 1 Carbon, 2 Hydrogen, 3 Oxygen
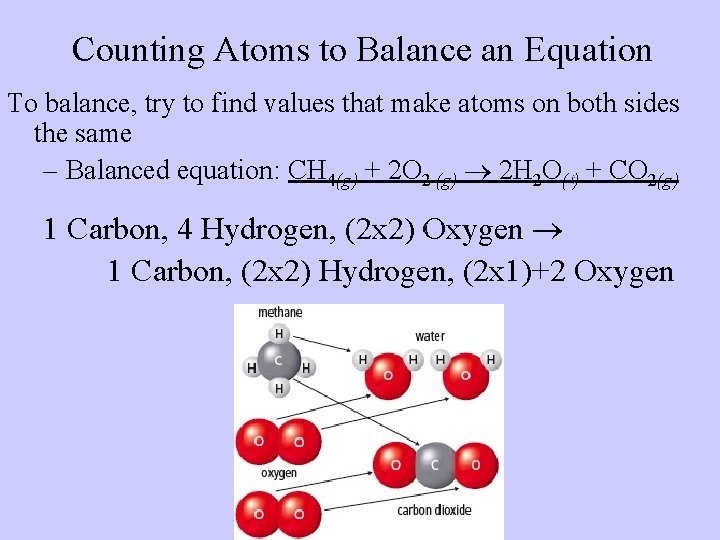
Counting Atoms to Balance an Equation To balance, try to find values that make atoms on both sides the same – Balanced equation: CH 4(g) + 2 O 2 (g) 2 H 2 O( ) + CO 2(g) 1 Carbon, 4 Hydrogen, (2 x 2) Oxygen 1 Carbon, (2 x 2) Hydrogen, (2 x 1)+2 Oxygen

Hints for Writing Chemical Equations – Use the chemical symbol to represent single elements. • Be careful of diatomic and polyatomic elements such as O 2, P 4 and S 8 • The “special seven” are all diatomic elements (hockey stick & puck) – H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2

Hints for Writing Chemical Equations – Several common covalent molecules containing hydrogen have common names that do not help in writing chemical formulas • For example, – methane = CH 4, – glucose = C 6 H 12 O 6, – Ethane = C 2 H 6, – Ammonia = NH 3

Strategies for Balancing Equations • Tips to balance chemical equations: – Trial and error will work, but can be very inefficient – Balance compounds first, elements last – Balance one compound at a time – Only add coefficients; NEVER change subscripts! – Balance metals first, then nonmetals, then H & O – If H and O appear in more than one place, attempt to balance them LAST – Polyatomic ions (such as SO 42–) can often be balanced as a whole group – Always double-check after you think you are finished!

Balancing Equations Examples • Balance the following: 2 Fe + 3 Br 2 2 Fe. Br 3 Fe + Br 2 Fe. Br 3

Balancing Equations Examples Balance the following: Sn(NO 2)4 + K 3 PO 4 KNO 2 + Sn 3(PO 4)4

Balancing Equations Examples Balance the following: C 2 H 6 + O 2 CO 2 + H 2 O
- Slides: 16