Balancing Chemical Equations Part II Balancing Chemical Reactions
Balancing Chemical Equations Part II
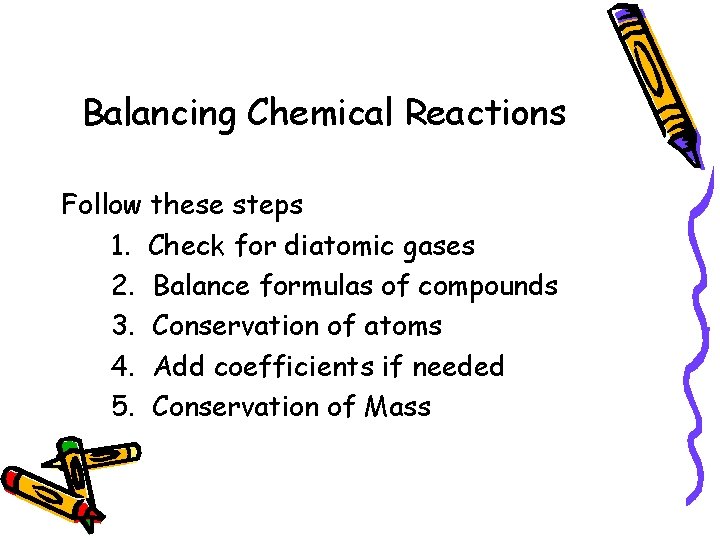
Balancing Chemical Reactions Follow these steps 1. Check for diatomic gases 2. Balance formulas of compounds 3. Conservation of atoms 4. Add coefficients if needed 5. Conservation of Mass

A. Check For Diatomic Gases • • Diatomic Gases – molecules containing only two atoms. You must include the subscript “ 2” when the symbol is written by itself. • Seven Diatomic Gases nitrogen, N 2 oxygen, O 2 fluorine, F 2 chlorine, Cl 2 bromine, Br 2 iodine, I 2 hydrogen, H 2 Name One Frozen Clam Broke In Hawaii

Diatomic Gases Example H 2(g) + O 2(g) → H 2 O(g) Hydrogen and oxygen are diatomic gases when written ALONE. You must include the subscript “ 2”. Note: The equation isn’t balanced.
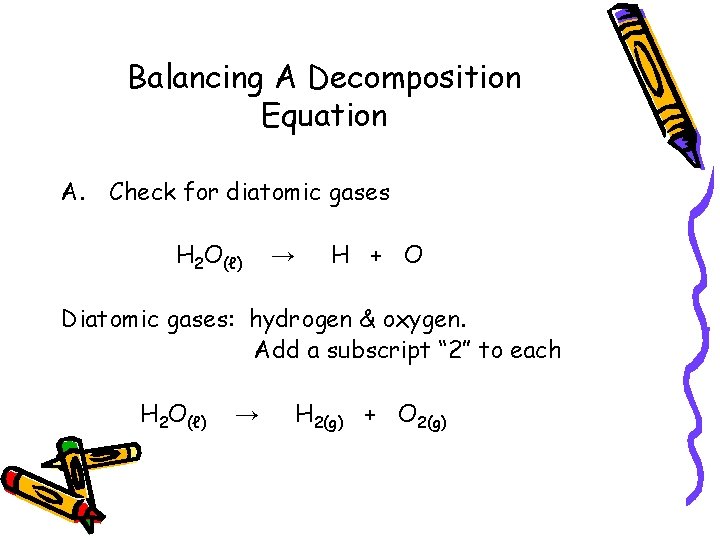
Balancing A Decomposition Equation A. Check for diatomic gases H 2 O(ℓ) H + O → Diatomic gases: hydrogen & oxygen. Add a subscript “ 2” to each H 2 O(ℓ) → H 2(g) + O 2(g)

B. Check The Formulas of Compounds • The compound in the equation is water. • The formula is balanced. H 2 O(ℓ) → 2 H 2(g) + O 2(g)
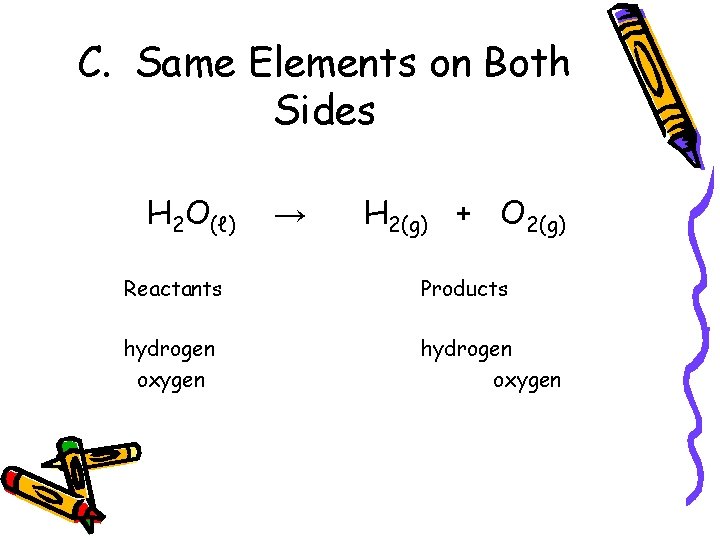
C. Same Elements on Both Sides H 2 O(ℓ) → H 2(g) + O 2(g) Reactants Products hydrogen oxygen

D. Conservation of Atoms H 2 O(ℓ) 2–H 1–O → 2 H 2(g) + O 2(g) 2 -H 2–O Atoms aren’t conserved. Equation isn’t balanced.

E. Balanced Equation 2 H 2 O(ℓ) → 2 H 2(g) + O 2(g) The coefficients “ 2” are placed in front of water, and hydrogen. 4–H 2–O The equation is balanced. 4–H 2–O
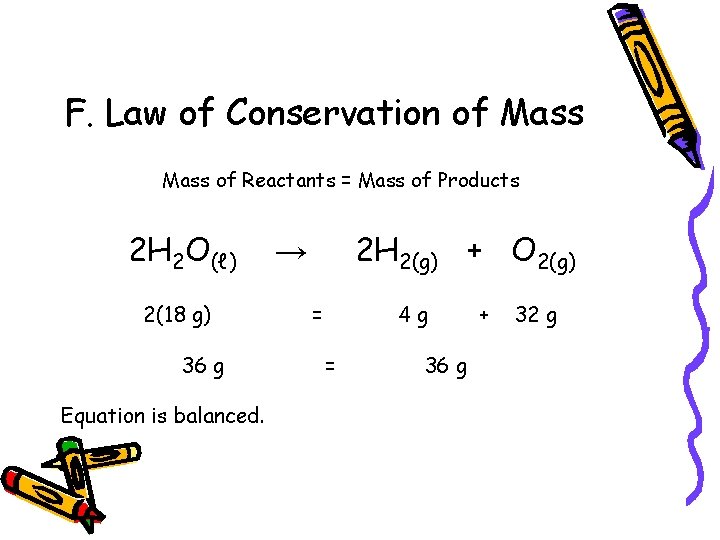
F. Law of Conservation of Mass of Reactants = Mass of Products 2 H 2 O(ℓ) 2(18 g) 36 g Equation is balanced. 2 H 2(g) + O 2(g) → = 4 g = 36 g + 32 g

Balancing A Synthesis Equation • Equation: Na + Cl → A. Diatomic Gas: Na. Cl Cl is a diatomic gas. Na isn’t a diatomic gas. Na is a metal. Na + Cl 2 → Na. Cl Formula of compound balanced? Yes. Na+Cl-

Balancing A Synthesis Equation B. Put a “ 2” in front of the Na and a “ 2” in front of Na. Cl. Check number of each element again. 2 Na + Cl 2 → Na = 2 Cl = 2 The equation is balanced. 2 Na. Cl Na = 2 Cl = 2

Balancing A Synthesis Equation C. Check number of each element on both sides. Na + Cl 2 → Na. Cl Na = 1 Cl = 2 Cl = 1 Not Balanced.
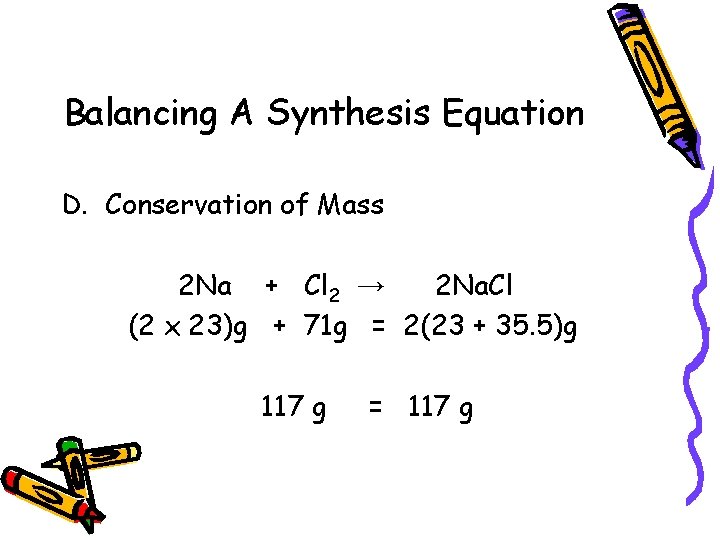
Balancing A Synthesis Equation D. Conservation of Mass 2 Na + Cl 2 → 2 Na. Cl (2 x 23)g + 71 g = 2(23 + 35. 5)g 117 g = 117 g

Balancing A Single Replacement Equation C. Same elements on both sides Na. I + F 2(g) → Na. F + I 2(g) sodium iodine fluorine

Balancing A Single Replacement Equation A. Check for diatomic gases Na. I + F → Na. F + I F and I are diatomic gases. Add “ 2” as a subscript on both. Na. I + F 2(g) → Na. F + I 2(g)

Balancing A Single Replacement Equation B. Balance the formulas of compounds + + - Na I + F 2(g) → Na F + I 2(g) The compounds, Na. I and Na. F are balanced.

Balancing A Single Replacement Equation D. Same number of atoms or ions on both sides Na. I + F 2(g) → Na. F + I 2(g) 1 – Na 1–I 2–F Equation is incorrect. 1 – Na 2–I 1–F

Balancing A Single Replacement Equation “ 2” is needed in front of Na. I and Na. F. 2 Na. I + F 2(g) → 2 Na. F + I 2(g) 2 – Na 2–I 2–F Equation is correct. 2 – Na 2–I 2–F

Balancing A Single Replacement Equation E. Same mass on both sides 2 Na. I + F 2(g) → 2 Na. F + I 2(g) 2(23 + 127)+ 2(19) = 2(23 + 19) + 2(127) 338 g = 338 g

Balancing A Double Replacement Equation A. Check for diatomic gases. Fe S(s) + H Cl(aq) → H S(g) + Fe Cl(aq) No diatomic gases. Chlorine isn’t ALONE.

B. Check The Formulas of Compounds All formulas must be balanced. Incorrect: + 2+ 22+ Fe S(s) + H Cl(aq) → H S(g) + Fe Cl(aq) Correct: 2 + 2+ 2 + 2 Fe S(s) + H Cl(aq) → H 2 S(g) + Fe Cl 2(aq)
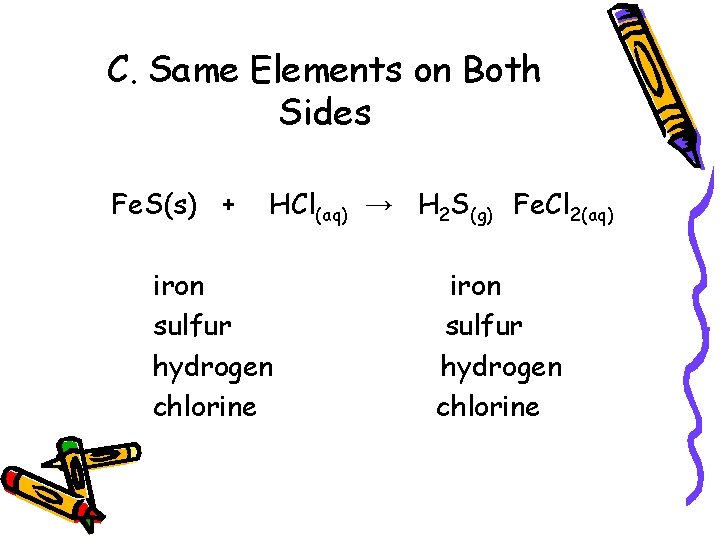
C. Same Elements on Both Sides Fe. S(s) + HCl(aq) → H 2 S(g) Fe. Cl 2(aq) iron sulfur hydrogen chlorine

D. Conservation of Atoms The number of ions or atoms each element must be the same on both sides Fe S(s) + H Cl(aq) → H 2 S(g) + Fe Cl 2(aq) Reactants Fe =1 S=1 H=1 Cl = 1 This equation is incorrect. Products Fe = 1 S=1 H=2 Cl = 2

D. What’s Needed? • We need 2 hydrogen and 2 chlorine. • You can’t write in subscripts. This is incorrect. H 2 Cl 2. This formula isn’t balanced. Hydrogen and chlorine are written with the subscript “ 2” when they are alone in an equation. • You must write the 2 in front of HCl as a coefficient. • You have 2 -H and 2 -Cl.

E. Balanced Equation Balanced Formula Fe S(s) + 2 HCl(aq) Reactants Fe =1 S=1 H=2 Cl = 2 This equation is correct. → H 2 S(g) + Fe. Cl 2(aq) Products Fe = 1 S=1 H=2 Cl = 2
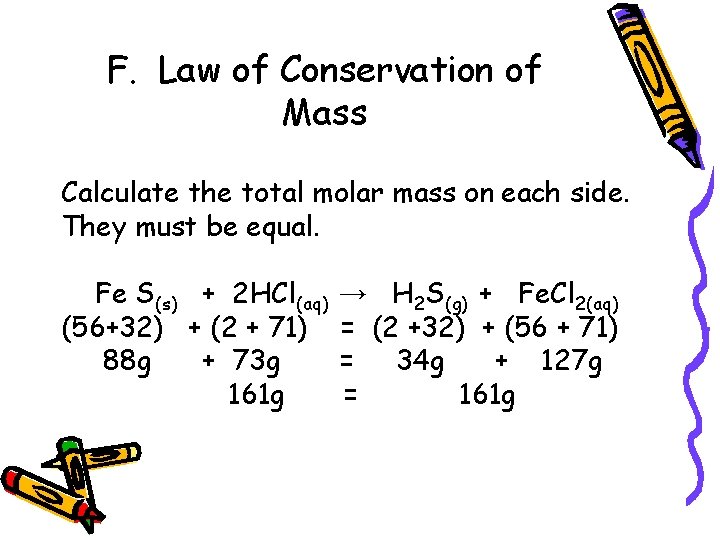
F. Law of Conservation of Mass Calculate the total molar mass on each side. They must be equal. Fe S(s) + 2 HCl(aq) (56+32) + (2 + 71) 88 g + 73 g 161 g → H 2 S(g) + Fe. Cl 2(aq) = (2 +32) + (56 + 71) = 34 g + 127 g = 161 g
- Slides: 27