Balancing Chemical Equations Notes Parts of an equation

Balancing Chemical Equations Notes

Parts of an equation • A. Reactants: the original, starting substances • B. Products: the new substances produced • C. Coefficients: whole numbers in front of a formula; indicates the number of molecules needed in the balanced equation • D. Subscripts: small numbers in formulas that tell the number of atoms of each element in the formula

• E. Correctly written formulas: chemical formulas must be written to represent 1 molecule/formula unit of a substance • F. Yields sign: the arrow; points to the products; means gives, equals, or produces • G. States of matter: shows what state of matter each substance is in (solid, liquid, gas, or aqueous)
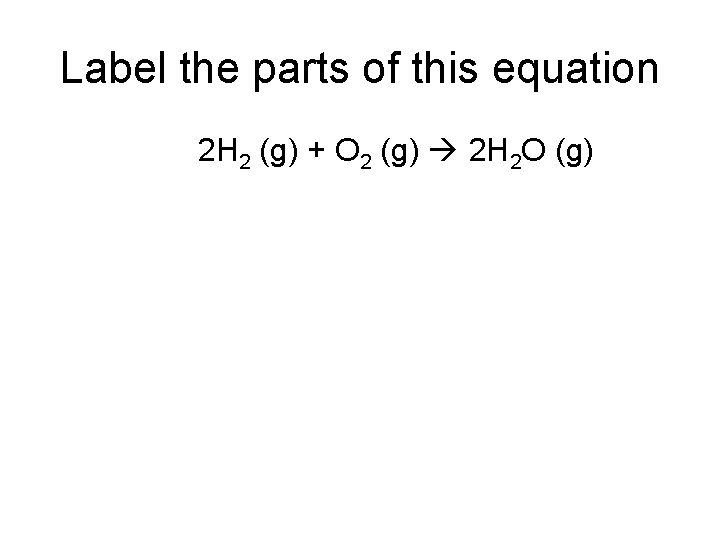
Label the parts of this equation 2 H 2 (g) + O 2 (g) 2 H 2 O (g)

Word Equations • Describe a chemical reaction with a sentence • Use correct names of reactants and products • Example – Hydrogen gas reacts with oxygen gas to produce water vapor

Formula (skeletal) Equations • Describe a reaction using the correct chemical formulas of the reactants and products • State of substance may be given • Example H 2 + O 2 H 2 O Or H 2(g)+ O 2(g) H 2 O(g)

Balanced Equations • Formula Equations that show the law of conservation of mass is obeyed • Coefficients are used to make the total number of atoms equal on each side • Example 2 H 2 + O 2 2 H 2 O *Note that a coefficient applies to the whole formula; multiply the coefficient by the subscript to get the total number of atoms of each element in the formula

Tips for balancing equations • • • Always start with correctly written formulas Keep a tally of how many atoms of each element are present on each side Example: Ca. Cl 2 + Ag. Br Ca. Br 2 + Ag. Cl 1 -Ca-1 2 -Cl-1 1 -Ag-1 1 -Br-2

• Place coefficients in front of formulas as needed to balance and change your tally numbers as you do so. • Continue until you have equal #’s of each type of atom on each side • Coefficients should be in the simplest ratio Ca. Cl 2 + Ag. Br Ca. Br 2 + 2 Ag. Cl 1 -Ca-1 2 -Cl-1 2 1 -Ag-1 2 1 -Br-2
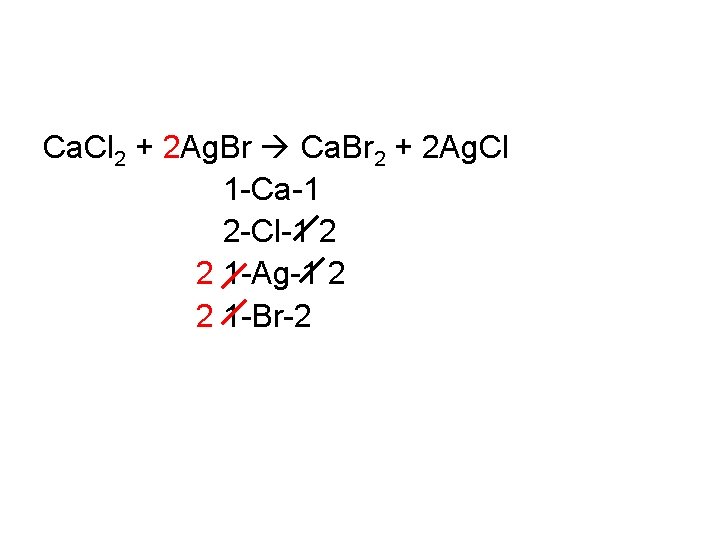
Ca. Cl 2 + 2 Ag. Br Ca. Br 2 + 2 Ag. Cl 1 -Ca-1 2 -Cl-1 2 2 1 -Ag-1 2 2 1 -Br-2

Other Tips Sometimes balancing is just trial and error, but – Sometimes you need to find the least common multiple to balance a certain element Al. Br 3 + Cl 2 Al. Cl 3 + Br 2

• Notice when you tally your atoms, you have 3 Br’s on the left side and 2 Br’s on the right side. You have to use the LCM to balance (2 x 3=6 on both sides) • 2 Al. Br 3 + 3 Cl 2 2 Al. Cl 3 + 3 Br 2 2 1 -Al-1 2 6 3 -Br-2 6 6 2 -Cl-3 6

– In ionic equations it is often helpful to keep polyatomic ions together as a unit Na. NO 3 + Ca 3(PO 4)2 Na 3 PO 4 + Ca(NO 3)2 1 -Na-3 1 -NO 3 -2 3 -Ca-1 2 -PO 4 -1 Try to finish balancing this one!

– In combustion reactions, balance C first, then H, then O – If you end up with an odd number of oxygens, double all the coefficients, then balance the oxygen C 3 H 8 + O 2 CO 2 + H 2 O Try to finish balancing this one!
- Slides: 14