Balancing Chemical Equations March 2017 Balancing Chemical Equations

Balancing Chemical Equations March 2017

Balancing Chemical Equations The equation on the right is said to be “balanced” because: the number of atoms of each element on the left side of the arrow = the number of atoms of each element on the right side of the arrow

Balancing Chemical Equations The equation on the right is NOT BALANCED To balance the atoms, numbers called “coefficients” need to be added in front of the substances that contain unbalanced atoms.

Let’s start with a simpler example: Na(s) + Cl 2 (g) Na. Cl Na = Cl =

Here is another example: Na(s) + O 2 (g) Na 2 O Na = O= In this example it is easier to start by balancing the oxygen first

Here is another example with a polyatomic ion: Na 2 SO 4 (aq) + Ba. Cl 2 (aq) Na. Cl(aq) + Ba. SO 4(s) Na = SO 42 - = Cl = In this example it is easier to keep the S and O as SO 42 -

Here is another example with a polyatomic ion: Na. OH (aq) + Mg. Cl 2 (aq) Na. Cl(aq) + Mg(OH)2 (s) Na = OH- = Mg = Cl =

Here is a more challenging example: N 2 (g) + H 2 (g) NH 3 (g) N= H=

Here is a more challenging example: CH 4 + O 2 CO 2 + H 2 O C= H= O= Oxygen is difficult to balance since it appears in 2 of the products. It is easier to start balancing something other than oxygen.
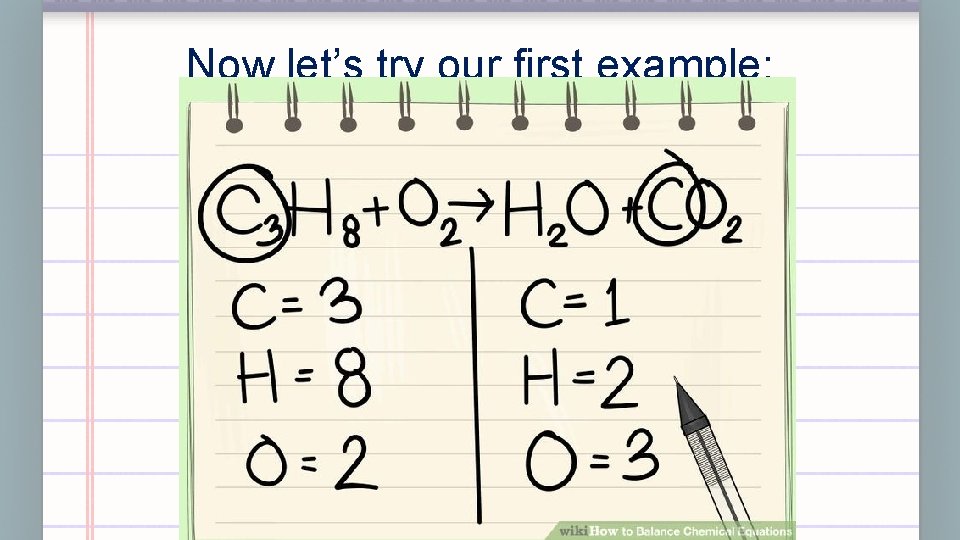
Now let’s try our first example:
- Slides: 10