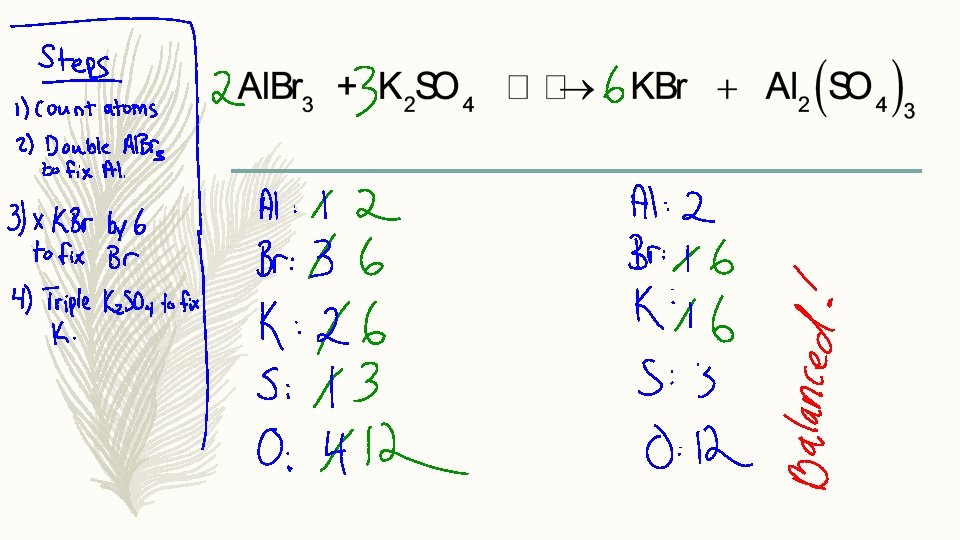
Balancing Chemical Equations Conservation of Mass Counting Atoms

Balancing Chemical Equations

Conservation of Mass (Counting Atoms) – Step 1: Count the number of atoms on both sides of the equation (for the reagents and products). If it’s balanced, you don’t have to do anything! – 2 Na. Cl + F 2 2 Na. F + Cl 2

Conservation of Mass (Counting Atoms) – Step 1: Count the number of atoms on both sides of the equation (for the reagents and products). If it’s balanced, you don’t have to do anything! – Fe. Cl 3 + Na. OH 3 Fe(OH)3 + Na. Cl

Conservation of Mass (Counting Atoms) – Step 2: Add a coefficient in front of a molecule such that one element becomes balanced. Fe. Cl 3 + Na. OH Fe(OH)3 + Na. Cl

Conservation of Mass (Counting Atoms) – Step 3: If it is not fully balanced, add another coefficient to another molecule to bring another element into balance. Repeat until the equation is fully balanced. Fe. Cl 3 + Na. OH Fe(OH)3 + Na. Cl

Important Rules – You are only allowed to add coefficients, and they must appear at the front of the molecule… not in the middle! – Ex: H 2 SO 4

Important Rules – No changing the subscript. This would change the molecule, which is not allowed. You can only change the quantity of each molecule. – Ex: H 2 SO 4

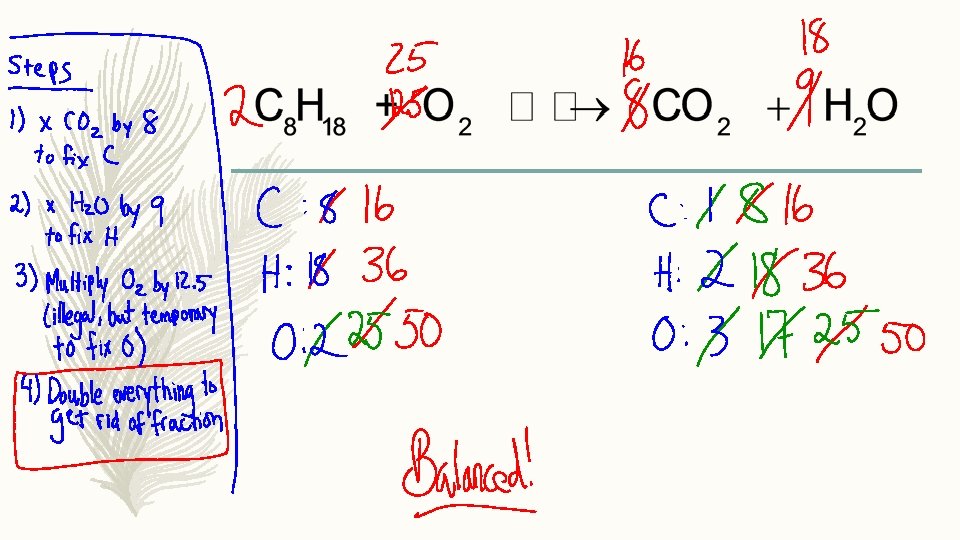
One more important rules – Hate fractions? Good news, they’re NOT allowed as coefficients. – What should you do about this? –

Tips – Sometimes you will only have to change one coefficient, and sometimes you will have to change several. This is normal. Keep going until it is perfectly balanced. – Sometimes you may have to adjust a coefficient more than once in the same equation. This is also normal. Keep going!

Workbook p. 21
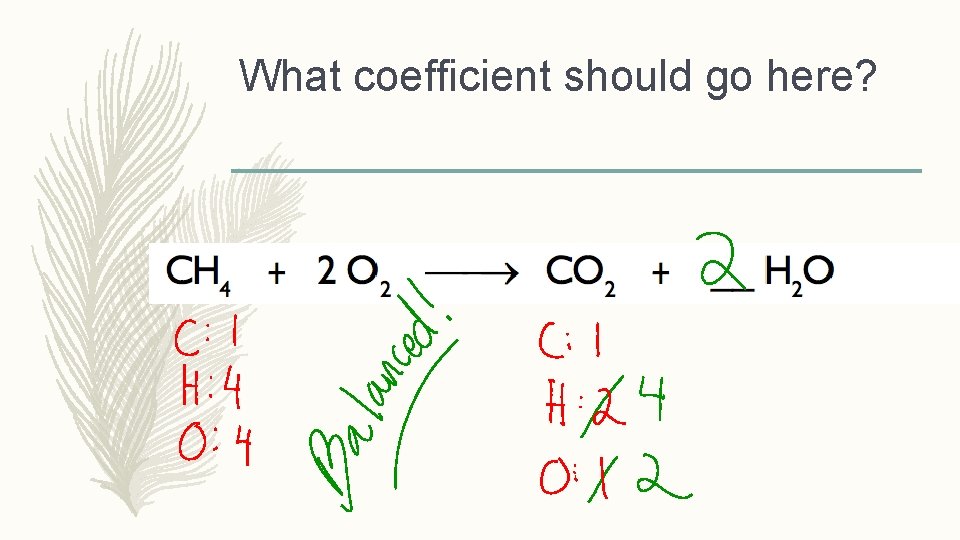
What coefficient should go here?
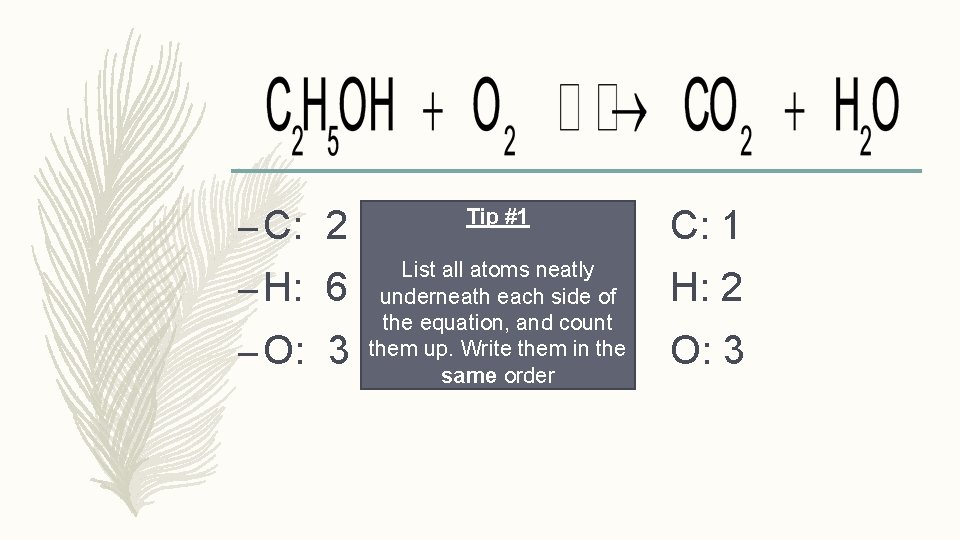
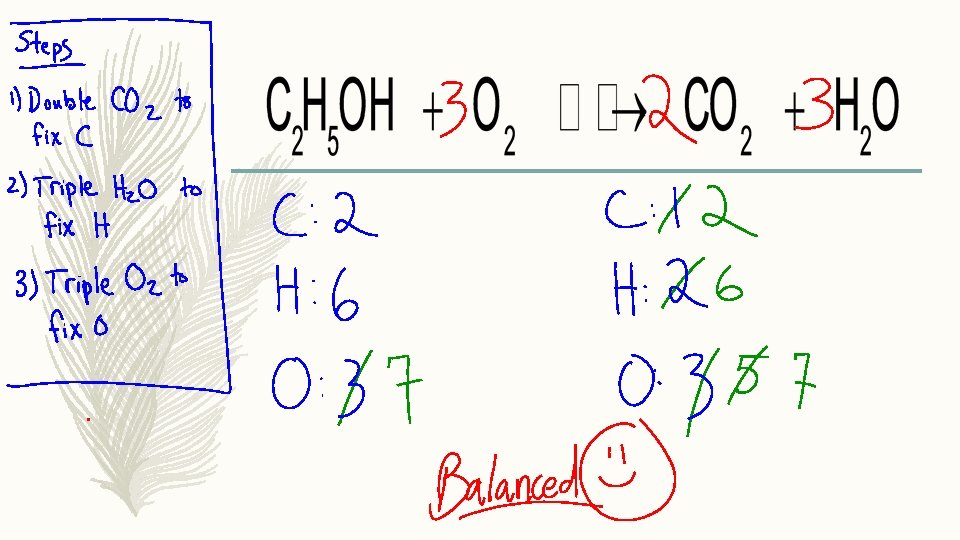
– C: 2 Tip #1 C: 1 – H: 6 List all atoms neatly underneath each side of the equation, and count them up. Write them in the same order H: 2 – O: 3
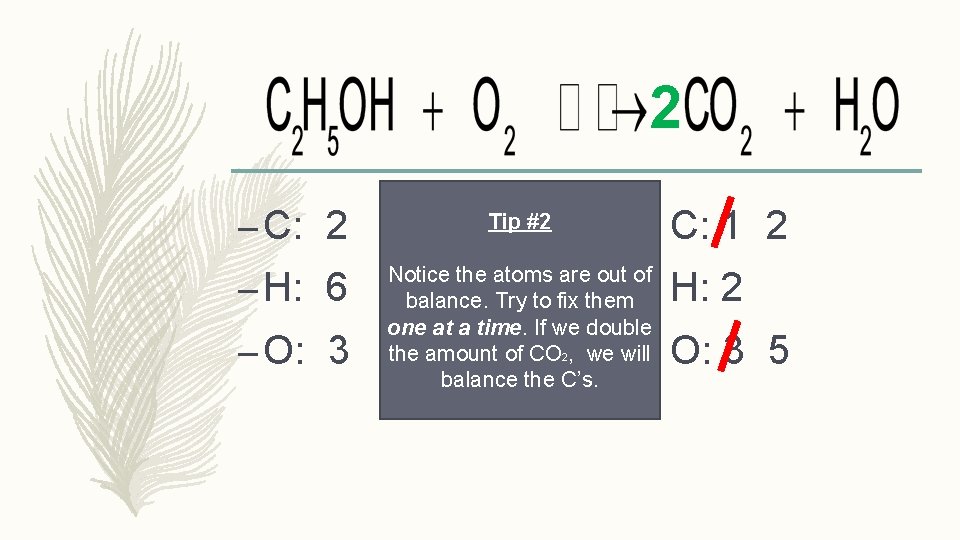
2 – C: 2 Tip #2 – H: 6 Notice the atoms are out of balance. Try to fix them one at a time. If we double the amount of CO 2, we will balance the C’s. – O: 3 C: 1 2 H: 2 O: 3 5
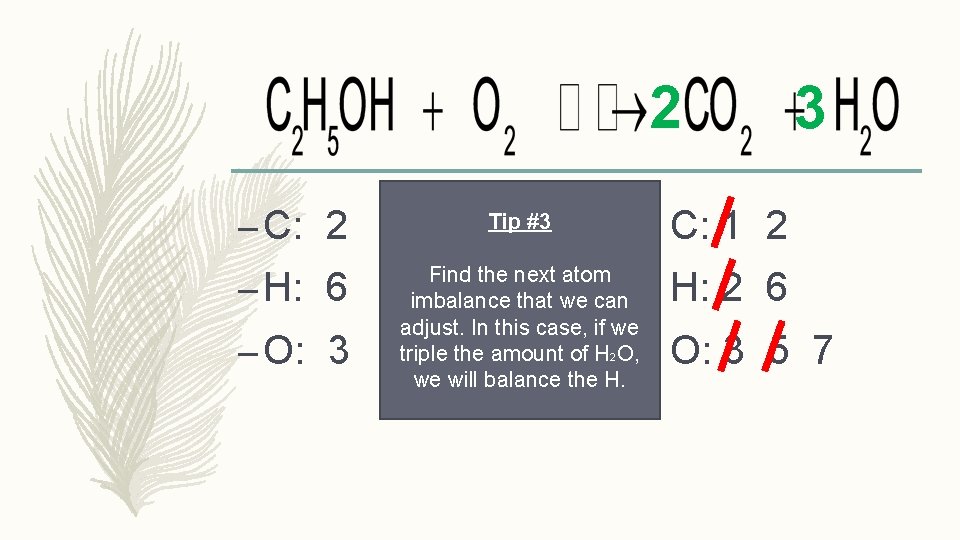
2 – C: 2 Tip #3 C: 1 2 – H: 6 Find the next atom imbalance that we can adjust. In this case, if we triple the amount of H 2 O, we will balance the H. H: 2 6 – O: 3 3 O: 3 5 7
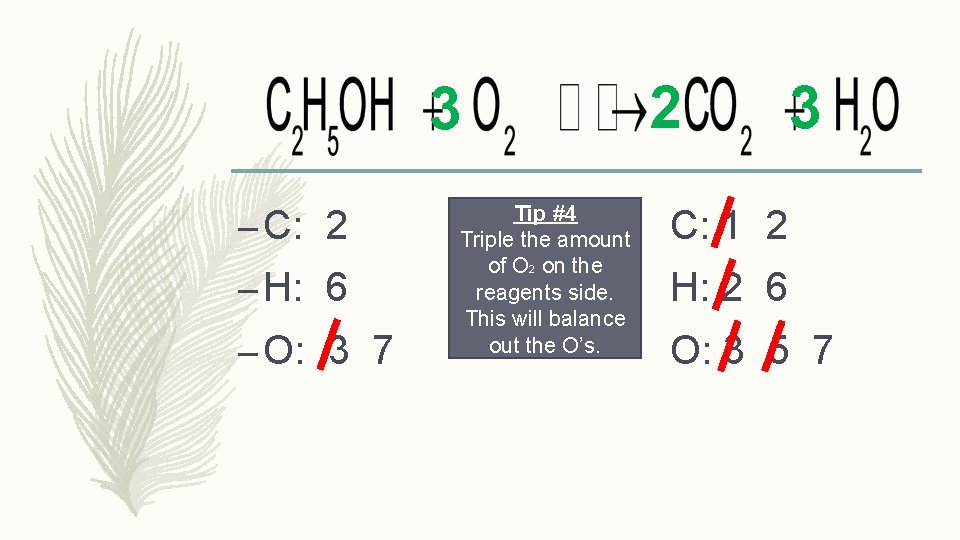
3 – C: 2 – H: 6 – O: 3 7 Tip #4 Triple the amount of O 2 on the reagents side. This will balance out the O’s. 2 3 C: 1 2 H: 2 6 O: 3 5 7
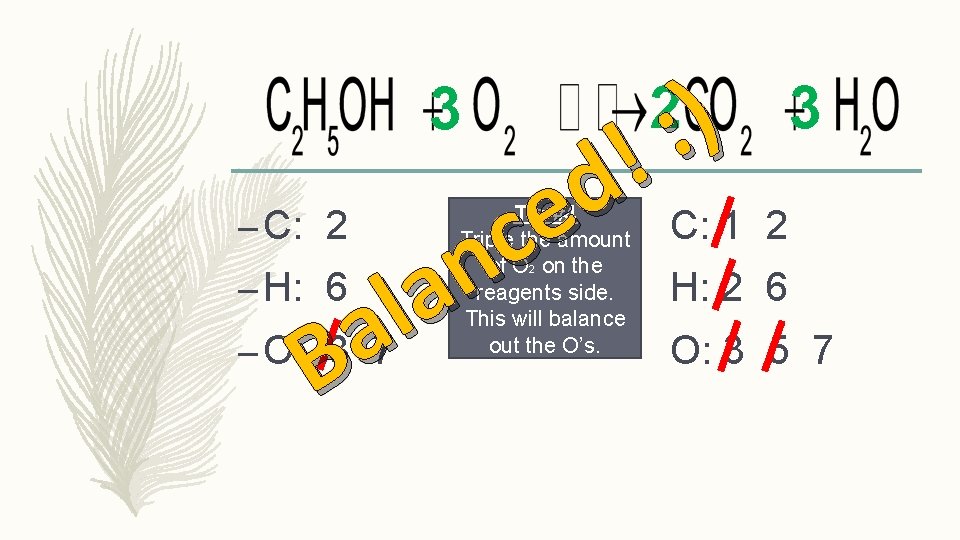
) : ! d e c n a l a B 3 – C: 2 – H: 6 – O: 3 7 Tip #4 Triple the amount of O 2 on the reagents side. This will balance out the O’s. 2 3 C: 1 2 H: 2 6 O: 3 5 7

Workbook p. 19 – https: //phet. colorado. edu/sims/html/balancing-chemicalequations/latest/balancing-chemical-equations_en. html
- Slides: 20