BALANCING CHEMICAL EQUATIONS CHEMICAL EQUATIONS A way to

BALANCING CHEMICAL EQUATIONS

CHEMICAL EQUATIONS A way to represent what happens in a chemical reaction. In a chemical reaction, bonds are broken and new ones are formed. Ex: C+O 2 C O O YIELDS Carbon + Oxygen YIELDS CO 2 O Carbon Dioxide

WHY IS THIS IMPORTANT?

SO YOU CAN UNDERSTAND

PHOTOSYNTHESIS 6 CO 2+ 6 H 2 O + ENERGY C 6 H 12 O 6 +6 O 2
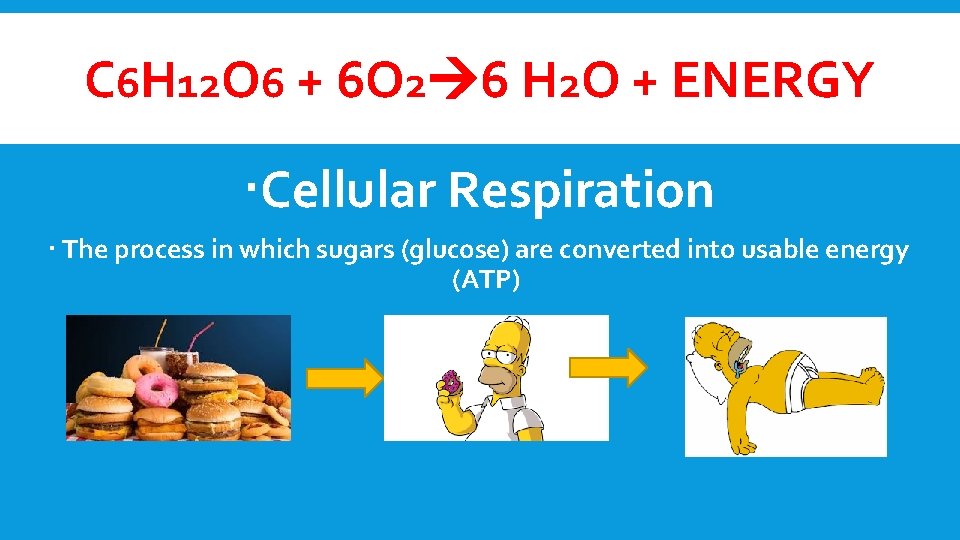
C 6 H 12 O 6 + 6 O 2 6 H 2 O + ENERGY Cellular Respiration The process in which sugars (glucose) are converted into usable energy (ATP)

RUSTING (OXIDATION)

BREATHING

DIGESTING

REACTANTS AND PRODUCTS Reactants Yields/Produces H + O H 2 O Products Reactants= Substances interacting with another Products= What is made (yielded or produced)

CHOCOLATE CAKE RECIPE REACTANTS PRODUCTS
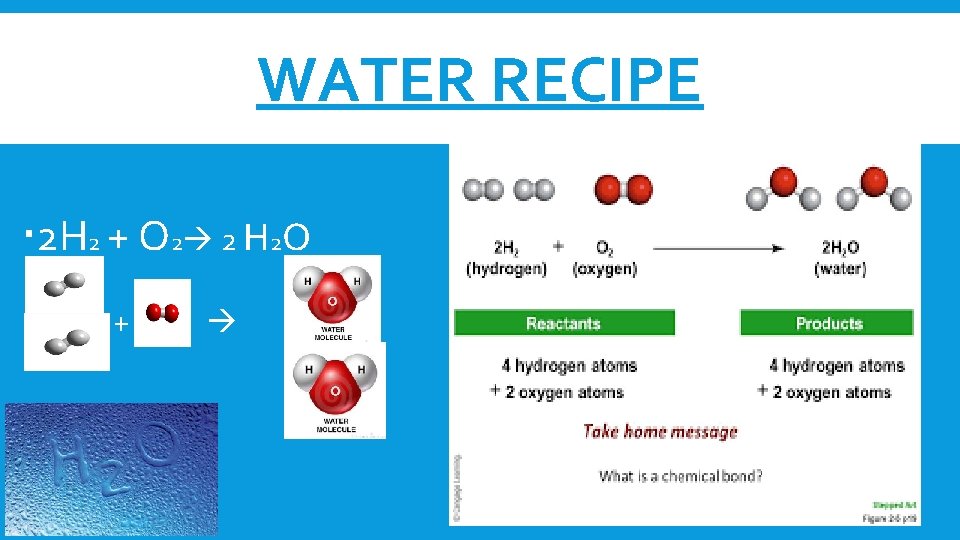
WATER RECIPE 2 H 2 + O 2 2 H 2 O +

WHY MUST CHEMICAL EQUATIONS BE BALANCED?

LAW OF CONSERVATION OF MASS Matter Cannot be created or destroyed. Matter changes form, not the amount of it after reacting.

LAW OF CONSERVATION OF MASS The number of atoms of each type of element must be the same before and after the reaction. This means that the mass before and after reacting must be the same. Sum of Reactants = Sum of Products

DEPENDS ON WHERE THE REACTION TAKES PLACE! Open System (fish bowl): Matter can enter from or escape to the surroundings. All matter is not contained (can escape) so mass can be lost to the environment.

DEPENDS ON WHERE REACTION TAKES PLACE! Closed System: Matter does not enter or leave Reactions in sealed containers are in a closed system No mass can be lost.

BALANCING EQUATIONS
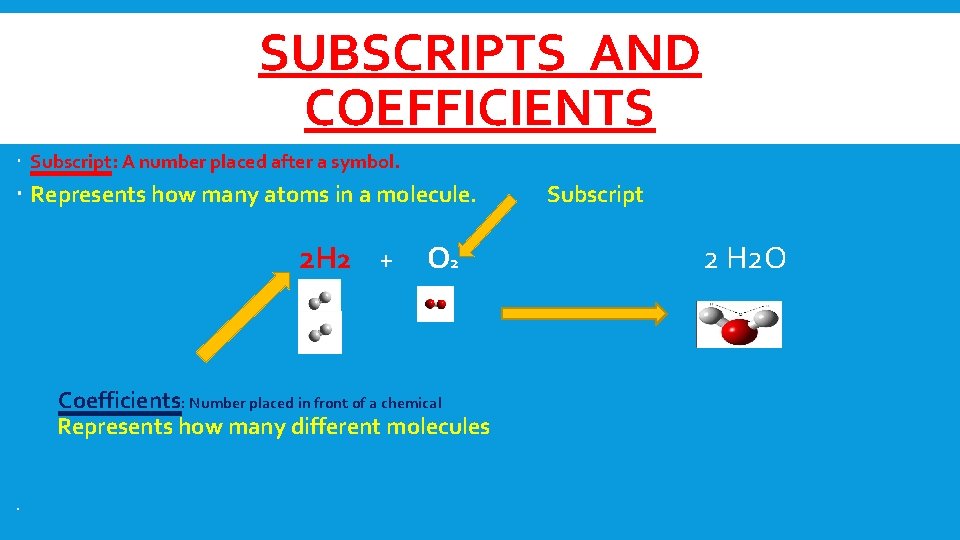
SUBSCRIPTS AND COEFFICIENTS Subscript: A number placed after a symbol. Represents how many atoms in a molecule. 2 H 2 + O 2 Coefficients: Number placed in front of a chemical Represents how many different molecules Subscript 2 H 2 O


STEPS FOR BALANCING EQUATIONS 1. Check for Diatomic Molecules (H 2, O 2, F 2, Br 2, I 2, N 2, Cl 2) ØThey need a subscript of 2 IF ALONE. ØEx: Al + O 2 = …it is alone ØEx: Al 2 O 3 = Is not alone (combined w/ aluminum) ØThis is the ONLY time you are allowed to add subscripts when balancing equations

STEPS FOR BALANCING EQUATIONS 2. Count how many of each atom there are. 3. Balance by adding coefficients to front of element symbols (metals, nonmetals, oxygen, then hydrogen) 4. Recount all atoms If the atoms are not balanced, work your way backwards until you find the mistake. 5. Reduce coefficients to simplest form (if possible) if every coefficient will reduce

REMEMBER: Balance equations by changing coefficients, NEVER by changing subscripts in a formula.
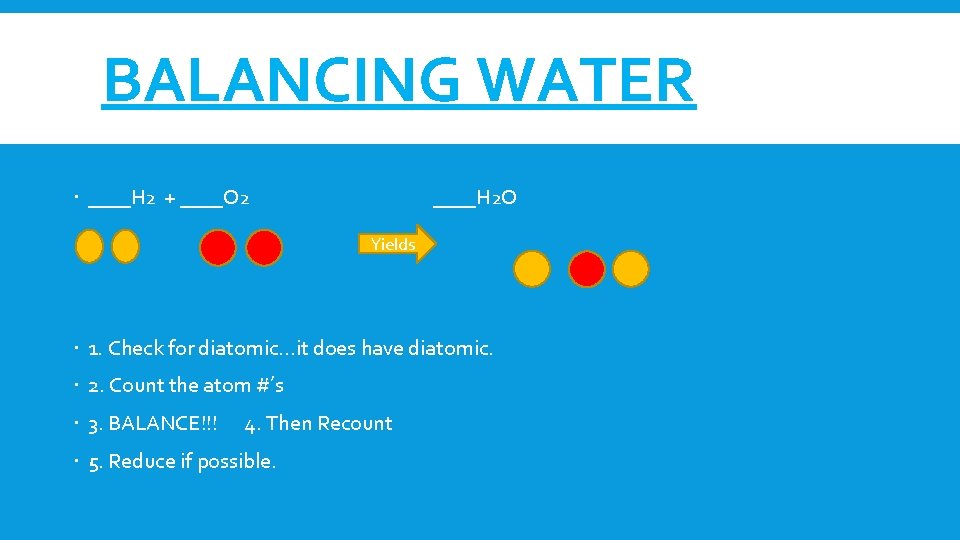
BALANCING WATER ____H 2 + ____O 2 ____H 2 O Yields 1. Check for diatomic…it does have diatomic. 2. Count the atom #’s 3. BALANCE!!! 4. Then Recount 5. Reduce if possible.

BALANCE THIS EQUATION _____Ch 4 + ____O 2 ____CO 2 + ____H 2 O
- Slides: 25