BALANCING CHEMICAL EQUATIONS Balancing Chemical Equations Rules 1

BALANCING CHEMICAL EQUATIONS. Balancing Chemical Equations

Rules: 1. The same number of atoms of each element appearing in reactants must appear in the products. 2. The subscripts of a formula cannot be changed to balance the equation. 3. Choose compound with greatest number of atoms first. 4. Balance by placing necessary coefficients in front of entire molecules. NO SUBSCRIPT CHANGES!

Rules cont. 5. Be certain each side of equation has same number of atoms of each type. 6. Make sure coefficients are in lowest whole number ratios. 7. Polyatomic ions usually stay intact, but multiple polyatomic ions may be separated. 8. (aq. ) stands for aqueous which is Latin for water solution: (g) gas (l) liquid (s) solid (c) crystalline solid

More Rules: means “yields” and shows direction of action ∆ above arrow shows heat has been added shows reaction is reversible MINHO: try Metals first, then Ions, then Nonmetals, and Hydrogen, then Oxygen last
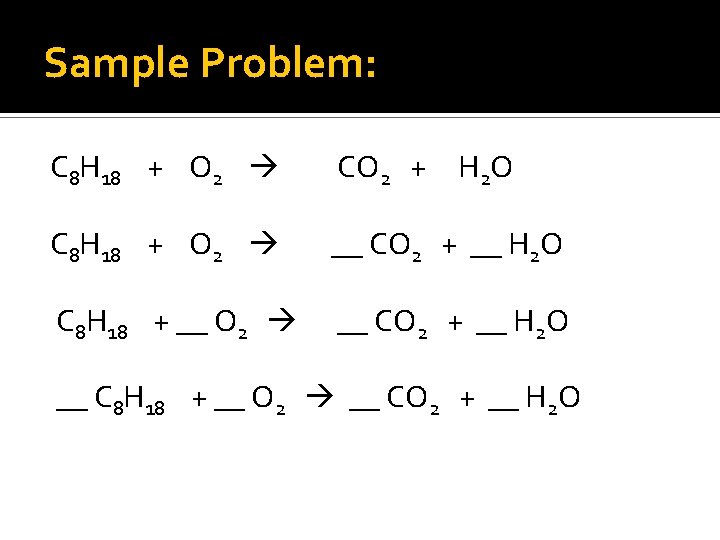
Sample Problem: C 8 H 18 + O 2 CO 2 + H 2 O C 8 H 18 + O 2 __ CO 2 + __ H 2 O C 8 H 18 + __ O 2 __ CO 2 + __ H 2 O __ C 8 H 18 + __ O 2 __ CO 2 + __ H 2 O

Practice Problems: H 2 Cr. O 4 + Ag. NO 3 Ag 2 Cr. O 4 + HNO 3 (aq) (s) (aq) Chromic Acid 1, 2, 1, 2 + Silver + Nitric Nitrate Chromate Acid Double displacement

Problem # 6 Cu(NO 3)2 (aq) Cu. O + NO 2 + O 2 (s) (g) Copper (II) + Nitrogen + Oxygen Nitrate Oxide Dioxide Gas 2, 2, 4, 1 Decomposition

Problem # 14 Al. Br 3 + Cl 2 Al. Cl 3 + Br 2 (aq) (g) Aluminum + Chlorine Aluminum + Bromine Bromide Gas Chloride Gas 2, 3, 2, 3 Single Displacement

Problem # 2 P 4 + O 2 + H 2 O H 3 PO 4 (s) (g) (aq) Phosphorus + Oxygen + Water Phosphoric (free) gas acid 1, 5, 6, 4 Synthesis

Problem # 20 sodium permanganate + sulfuric acid sodium sulfate + manganese (VII) oxide + water Na. Mn. O 4 + H 2 SO 4 Na 2 SO 4 + Mn 2 O 7 + H 2 O (aq) (s) (l) 2, 1, 1 Double Displacement

Problem # 3 Th(NO 3)4 + K 3 PO 4 (aq) Th 3(PO 4)4 + KNO 3 (s) (aq) Thorium + Potassium Nitrate Phosphate Nitrate 3, 4, 1, 12 Double Displacement

Problem # 22 sulfur trioxide + nitric acid sulfuric acid + nitrogen (V) oxide SO 3 + HNO 3 H 2 SO 4 + N 2 O 5 (g) (aq) (g) 1, 2, 1, 1 Double Displacement

Problem # 7 NH 4 NO 3 + Ca(OH)2 Ca(NO 3)2 + NH 3 + H 2 O (aq) (g) (l) Ammonium + Calcium + Ammonia + water Nitrate Hydroxide Nitrate 2, 1, 1, 2, 2 Double Displacement

Missing Products sodium chloride + potassium iodide Na. Cl + KI ____ + _____ (aq) (? ) Does a reaction take place?

Balancing Wars Problem # 1 Cu. SO 4 + H 3 PO 4 _____ + ______ (aq) A) B) C) D) E) 3, 1, 2, 1 3, 1, 1, 3 2, 3, 3, 2, 1, 3 2, 3, 3, 1 ((D)) Copper (II) + Phosphoric Copper (II) + Sulfuric Sulfate Acid Phosphate Acid

Balancing Wars Problem # 2 Mg(OH)2 + H 3 PO 4 _____ + ______ (aq) A. B. C. D. E. 1, 1, 1, 1 3, 2, 1, 3 2, 3, 1, 6 2, 3, 1, 1 3, 2, 1, 6 ((E. )) Magnesium + Phosphoric Magnesium + Water Hydroxide Acid Phosphate

Balancing Wars Problem # 3 Cu. Cl 2 + Na 3 PO 4 _____ + ______ (aq) A. B. C. D. E. 1, 2, 1, 2 2, 3, 1, 6 2, 3, 1, 1 3, 2, 1, 6 1, 1, 1, 1 ((D)) Copper (II) + Sodium Chloride Phosphate Copper (II) + Sodium Phosphate Chloride
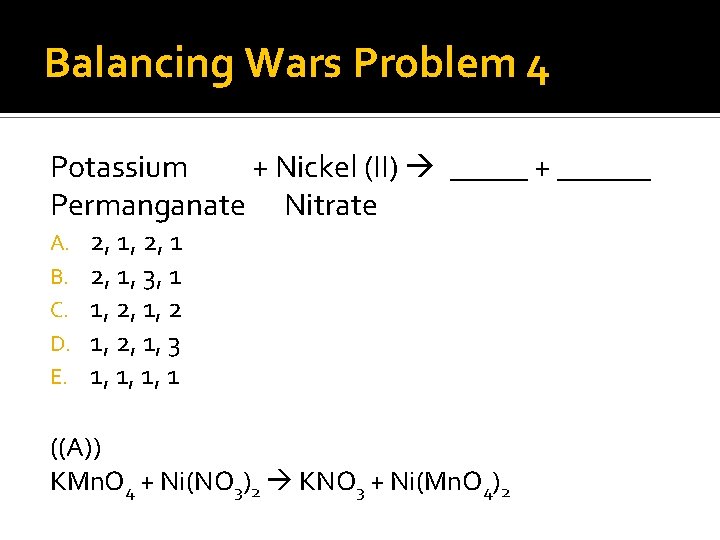
Balancing Wars Problem 4 Potassium + Nickel (II) _____ + ______ Permanganate Nitrate A. B. C. D. E. 2, 1, 3, 1 1, 2, 1, 3 1, 1, 1, 1 ((A)) KMn. O 4 + Ni(NO 3)2 KNO 3 + Ni(Mn. O 4)2
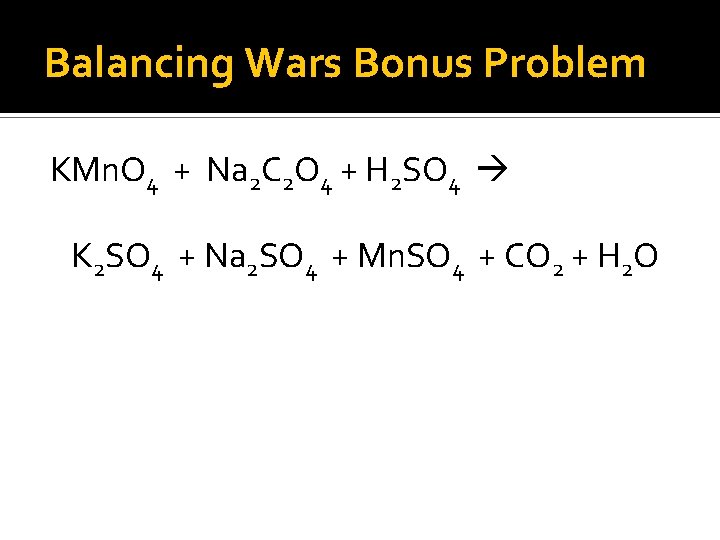
Balancing Wars Bonus Problem KMn. O 4 + Na 2 C 2 O 4 + H 2 SO 4 K 2 SO 4 + Na 2 SO 4 + Mn. SO 4 + CO 2 + H 2 O
- Slides: 19