Balancing and Classifying Chemical Equations Bell Ringer Write

Balancing and Classifying Chemical Equations

Bell Ringer • Write the name for each of the following compounds: • • • Fe 2 O 3 C 2 H 4 CO 2 HCl Al. Cl 3

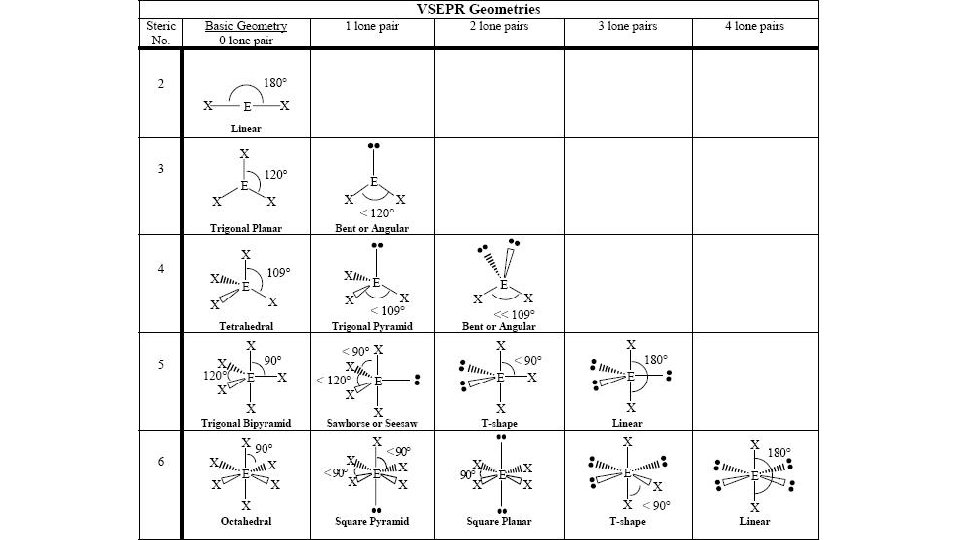
VSEPR • Valence Shell Electron Pair Repulsion • Determines the shape of compounds based on the intramolecular forces (forces that act within one compound) • • • Fe 2 O 3 C 2 H 4 CO 2 HCl Al. Cl 3

Parts of a chemical equation • Reactants (the molecules shown on the left of the arrow) • Products (the molecules shown on the right of the arrow). Example : Mg + O 2 Mg. O This reads “magnesium reacts with oxygen to yield magnesium oxide” Is the reaction balanced?

Subscripts vs. Coefficients The subscript tells you how many atoms of a particular element are in a compound. The coefficient tells you about the quantity, or number of molecules of the compound.

Why Balance? • Law of Conservation of Mass: mass is neither created nor destroyed in any ordinary chemical reaction.

Diatomic Elements • Br 2 • I 2 • N 2 • Cl 2 • H 2 • O 2 • F 2 These elements can never stand alone, they are always in pairs! (Will always have the subscript!)

Balancing Equations • Reactants “yield” Products • Balance by inspection Fe + O 2 Fe 2 O 3 4 Fe + 3 O 2 2 Fe 2 O 3 C 2 H 4 + O 2 CO 2 + H 2 O Al + HCl Al. Cl 3 + H 2 C 2 H 4 + 3 O 2 2 CO 2 + 2 H 2 O 2 Al + 6 HCl 2 Al. Cl 3 + 3 H 2

Classifying Reactions (Five Types) Combination (Synthesis) • Two or more reactants combine to form one product • General Form • A + B AB • Example: Mg + O 2 Mg. O

Decomposition • One reactant breaks down to become 2 or more products • General form • AB A + B • Examples • Na. N 3 N 2 + Na

Single Replacement • An element and a compound yield a different element and a different compound • General form • A + BC B + AC • Example • Al + Cu. Cl 2 Cu + Al. Cl 3

Double Replacement • Two compounds yield two different compounds • General form • AB + CD AD + CB • Examples • Na. OH + H 3 PO 4 Na 3 PO 4 + H 2 O

Combustion of an Organic Molecule (Hydrocarbons, alcohols, aldehydes, etc. ) • Organic molecule is burned in oxygen to yield carbon dioxide and water • General form • Cx. Hy + O 2 CO 2 + H 2 O • CH 4 + O 2 CO 2 + H 2 O

Tips for Balancing • Keep polyatomic ions together IF the same polyatomic ions are on both sides • If you get stuck, put a coefficient of 2 in front of the most complex species (if that does not work try a 3 etc. )
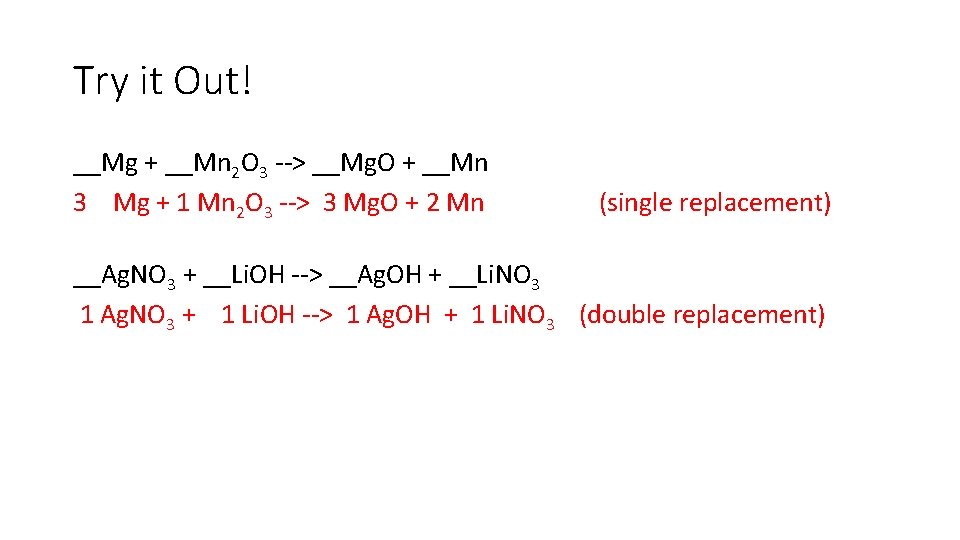
Try it Out! __Mg + __Mn 2 O 3 --> __Mg. O + __Mn 3 Mg + 1 Mn 2 O 3 --> 3 Mg. O + 2 Mn (single replacement) __Ag. NO 3 + __Li. OH --> __Ag. OH + __Li. NO 3 1 Ag. NO 3 + 1 Li. OH --> 1 Ag. OH + 1 Li. NO 3 (double replacement)
- Slides: 16