Balances on Nonreactive Processes Energy Balances on Nonreactive

Balances on Nonreactive Processes

Energy Balances on Nonreactive Processes Objectives: Ø Calculate DH (and DU ) from heat capacity equations, graphs, charts, tables associated with the change in T, P, phase and mixing process (given the initial and final states of materials). Ø Become familiar with steam tables and psychometric charts Ø Understand the concept of reference state for enthalpy values in the data source, identify the reference states. Ø Convert the expression for heat capacity from one unit to another.
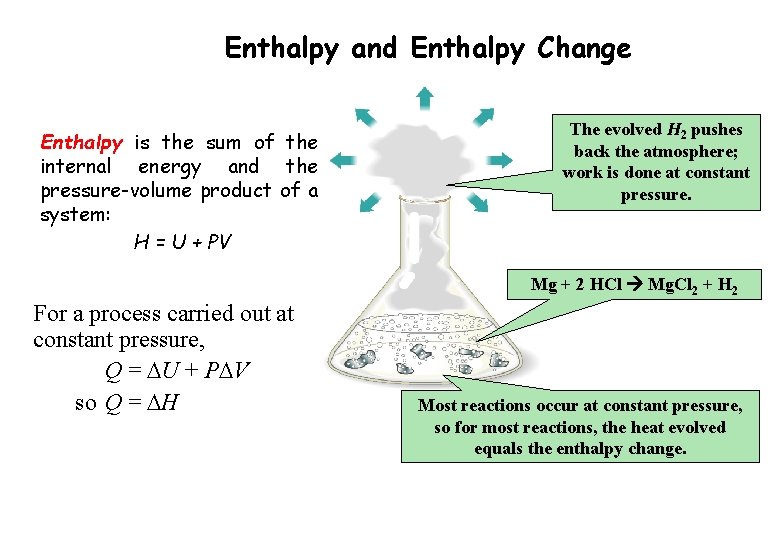
Enthalpy and Enthalpy Change Enthalpy is the sum of the internal energy and the pressure-volume product of a system: H = U + PV The evolved H 2 pushes back the atmosphere; work is done at constant pressure. Mg + 2 HCl Mg. Cl 2 + H 2 For a process carried out at constant pressure, Q = DU + PDV so Q = DH Most reactions occur at constant pressure, so for most reactions, the heat evolved equals the enthalpy change.

Properties of Enthalpy Ø Enthalpy is an extensive property. v It depends on how much of the substance is present. • Since U, P, and V are all state functions, enthalpy H must be a state function also. • Enthalpy changes have unique values. DH = Q Two logs on a fire give off twice as much heat as does one log. Enthalpy change depends only on the initial and final states.

Reference State • Enthalpy (H) and internal energy (U) are not absolute values …. . But values relative to a value at the reference state (T&P) • Energy balance of a control volume between 2 states requires only the energy difference (DU or DH) …. So Uref or Href can be at any reference state
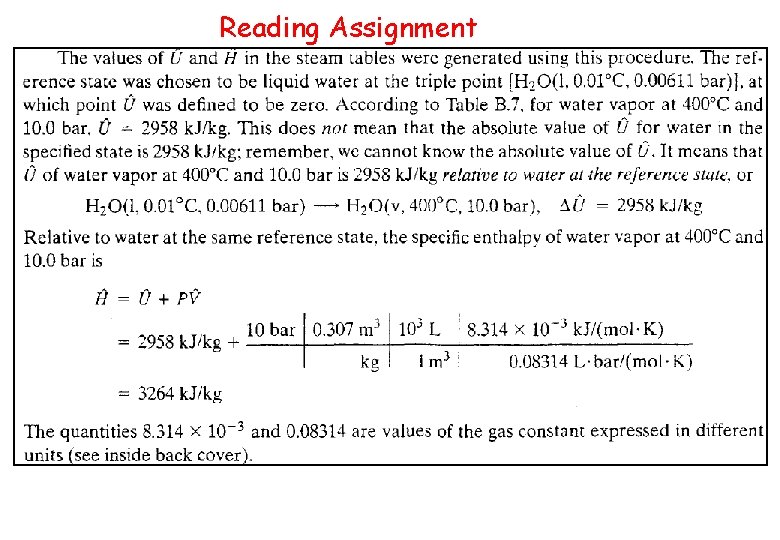
Reading Assignment

Example 1 • What reference state was used to generate the specific internal energies and enthalpies of steam? • Suppose water vapor at 300 o. C and 5 bar is chosen as a reference state at which Ĥ is defined to be zero. Relative to this state, what is the specific enthalpy of liquid at 75 o. C and 1 bar. What is the specific internal energy of liquid at 75 o. C and 1 bar? (Use Table B. 7) Ĥ=-2751 k. J/kg and specific internal energy =-2489 k. J/kg

Hypothetical Process Path Ø We will learn how to calculate DH and DU changes associated with certain process specifically: v v v Changes in P at constant T and state of aggregation Changes in T at constant P and state of aggregation Phase changes at constant T and P – melting, solidifying, vaporizing, condensing, and sublimation Mixing of two liquids or dissolving of a gas or a solid in a liquid at constant T and P. Chemical reaction at constant T and P Ø Calculate the overall DH using the summation of each DHi steps in system.

Hypothetical Process Path Suppose that we wish to calculate DĤ for a process in which solid ice at -5 o. C and 1 atm is converted to water vapor at 300 o. C and 5 atm. Since Ĥ is a state property, we could simply subtract DĤ at the initial state from DĤ at the final state DĤ = Ĥ (vapor, 300 o. C, 5 atm) - Ĥ (solid, -5 o. C, 1 atm) Suppose that we do not have such a table, our task is then to construct a hypothetical process path from the solid ice at -5 o. C and 1 atm to the water vapor at 300 o. C and 5 atm
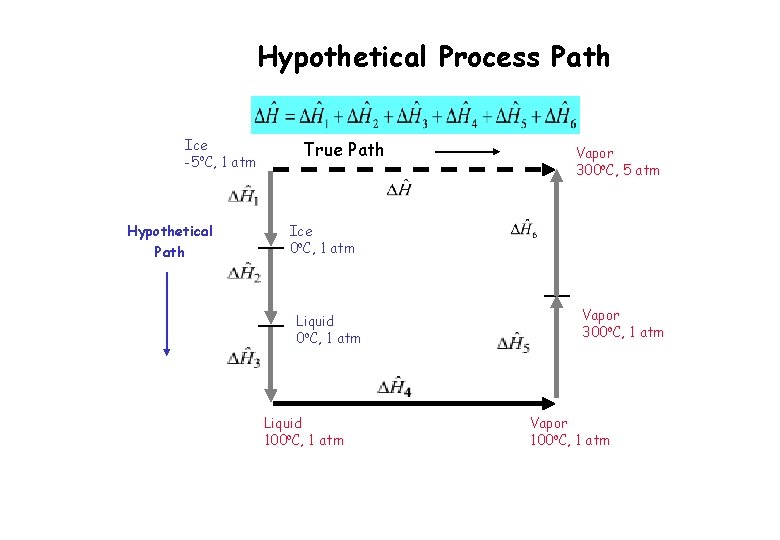
Hypothetical Process Path Ice -5 o. C, 1 atm Hypothetical Path True Path Vapor 300 o. C, 5 atm Ice 0 o. C, 1 atm Liquid 100 o. C, 1 atm Vapor 300 o. C, 1 atm Vapor 100 o. C, 1 atm

Reading Assignment

Reading Assignment

Example 2 Calculate the specific enthalpy requirement to transform water liquid at 0 o. C and 0. 01 bar to water vapor at 300 o. C and 5 bar DĤ = Ĥ (vapor, 300 o. C, 5 bar) - Ĥ (liquid, 0 o. C, 0. 01 bar) Table B. 7, Ĥ (300 o. C, 5 bar) = 3065 k. J/kg Table B. 5, Ĥ (0 o. C, 0. 01 bar) ~ 0 k. J/kg Hence, DĤ ~ 3065 k. J/kg Suppose that we do not have such a table, consruct a hypothetical process from water liquid at 0 o. C and 0. 01 bar to water vapor at 300 o. C and 5 atm

Example 2 - Hypothetical Process Path of enthalpy requirement to generate steam at 300 o. C, 5 bar

Working session 1 a. Cyclohexane vapor at 180 o. C and 5 atm is cooled and condensed to liquid cyclohexane at 25 o. C and 5 atm. The change enthalpy for the condensation of cyclohexane at 80. 7 o. C and 1 atm is known. Construct a hypothetical process path of the above process b. O 2 at 170 o. C and 1 atm, and CH 4 at 25 o. C and 1 atam are mixed and react completely to form CO 2 and H 2 O at 300 o. C and 1 atm. The enthalpy change for the reaction occuring at 25 o. C and 1 atm is known.

Solution Working session 1 a. Lower P isothermally to 1 atm, cool at 1 atm to 80. 7°C, condense at 80. 7°C and 1 atm, cool liquid at 1 atm to 25°C, raise pressure to 5 atm. b. Keeping pressure constant at 1 atm. cool O 2 to 25°C, mix O 2 and CH 4 at 25°C, carry out reaction at 25°C, raise product gas to 300°C.

Procedure for Energy Balance Calculations Perform all required material balance calculations Write the appropriate form of energy balance (closed or open system) and delete any of the terms that are either zero or negligible for the given process systems Choose a reference state – phase, temperature, and pressure – for each species involved in the process v v v For water look at the steam tables Choose the reference state used to generate the table Choose the inlet or outlet states as the reference state of the species (so that at least one or may be set to zero)
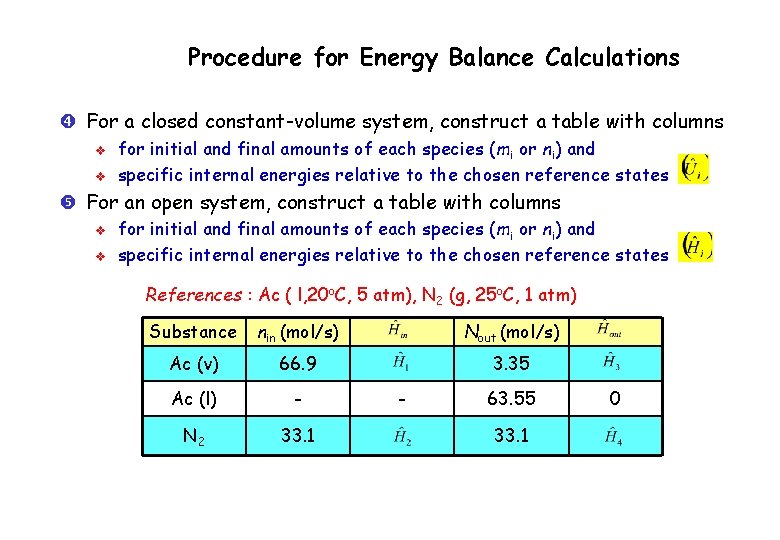
Procedure for Energy Balance Calculations For a closed constant-volume system, construct a table with columns v v for initial and final amounts of each species (mi or ni) and specific internal energies relative to the chosen reference states For an open system, construct a table with columns v v for initial and final amounts of each species (mi or ni) and specific internal energies relative to the chosen reference states References : Ac ( l, 20 o. C, 5 atm), N 2 (g, 25 o. C, 1 atm) Substance nin (mol/s) Nout (mol/s) Ac (v) 66. 9 3. 35 Ac (l) - N 2 33. 1 - 63. 55 33. 1 0

Procedure for Energy Balance Calculations Calculate all required values of internal energy and enthalpies and insert in the appropriate places in the table. Calculate

Procedure for Energy Balance Calculations Calculate any work, kinetic energy, or potential energy terms that you have not dropped from the energy balance. Solve the energy balance for whichever variable is unknown from the equation given below (often Q )

Changes in Pressure at Constant Temperature Ø Internal energy is nearly independent of pressure on solid and liquid at fixed temperature, as is specific volume. Ø If the pressure of liquid and solid change at constant temperature, you may write Ø Both and are independent of pressure for ideal gas. Ø Consequently, you may generally assume and for a gas undergoing isothermal pressure changes unless gas at temperature well below 0 o. C or well above 1 atm are involved

Sensible Heat and Heat Capacities Ø Sensible heat signifies heat that must be transferred to raise or lower the temperature of a substance or mixture of substances. Ø The quantity of heat required to produce a temperature change in a system is given by the first law of thermodynamics, neglecting the changes in kinetic and potential energies and as well as work.
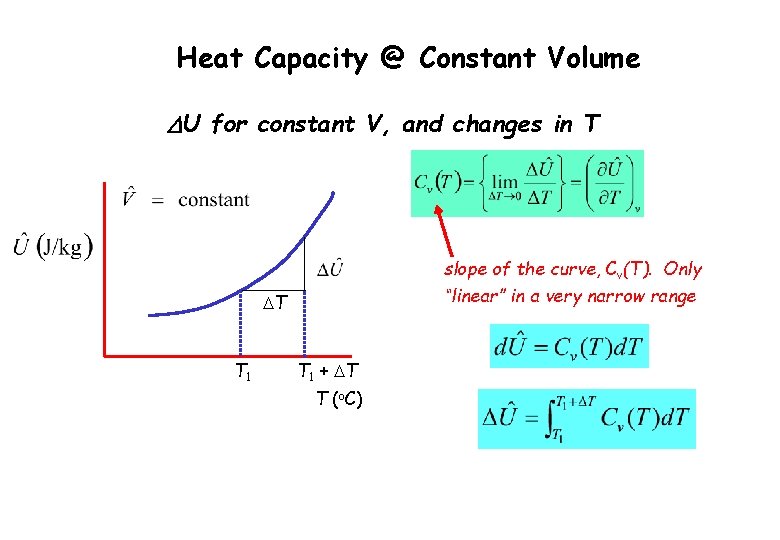
Heat Capacity @ Constant Volume DU for constant V, and changes in T slope of the curve, Cv(T). Only “linear” in a very narrow range DT T 1 + DT T (o. C)
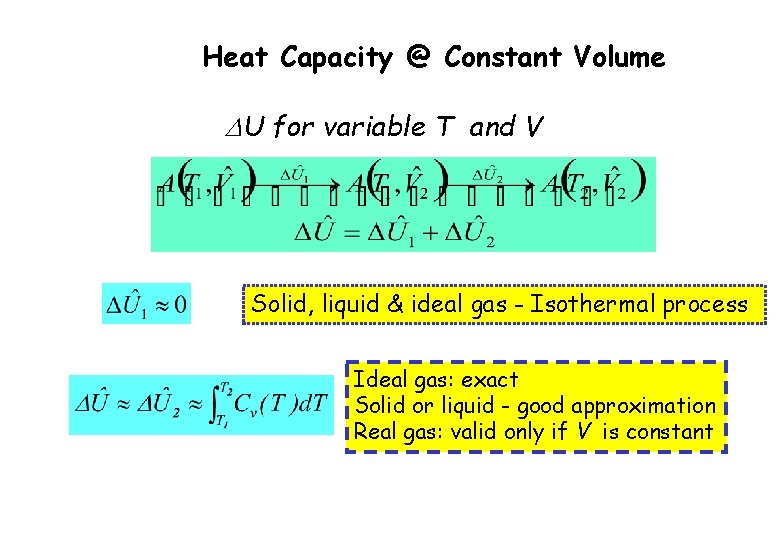
Heat Capacity @ Constant Volume DU for variable T and V Solid, liquid & ideal gas - Isothermal process Ideal gas: exact Solid or liquid - good approximation Real gas: valid only if V is constant

Example 3 : Evaluation of an Internal Energy Change from Tabulated Heat Capacity Calculate the heat required to raise 200 kg of nitrous oxide from 20 o. C to 150 o. C in a constant-volume vessel. The constant-volume heat capacity of N 2 O in this temperature range is given by the equation where T is in o. C.

Example 3 : Evaluation of an Internal Energy Change from Tabulated Heat Capacity Calculate the heat required to raise 200 kg of nitrous oxide from 20 o. C to 150 o. C in a constant-volume vessel. The constant-volume heat capacity of N 2 O in this temperature range is given by the equation where T is in o. C. The energy balance for this closed system
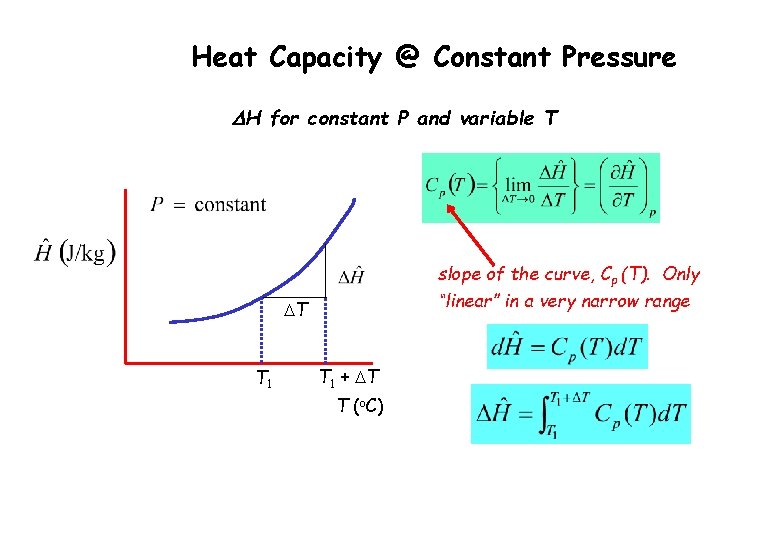
Heat Capacity @ Constant Pressure DH for constant P and variable T slope of the curve, Cp (T). Only “linear” in a very narrow range DT T 1 + DT T (o. C)

Heat Capacity @ Constant Pressure DH for variable T and P Ø For a process A(T 1, P 1) A(T 2, P 2) we may construct two-step process path Ø For the first step change (DĤ 1) in pressure at constant temperature

Heat Capacity @ Constant Pressure Ø For the second step change (DĤ 2) in temperature at constant pressure Solid, liquid & ideal gas Ø Hence, DĤ = DĤ 1 + DĤ 2 Ideal gas: Exact Nonideal gas : Exact only if P constant Solid or Liquid

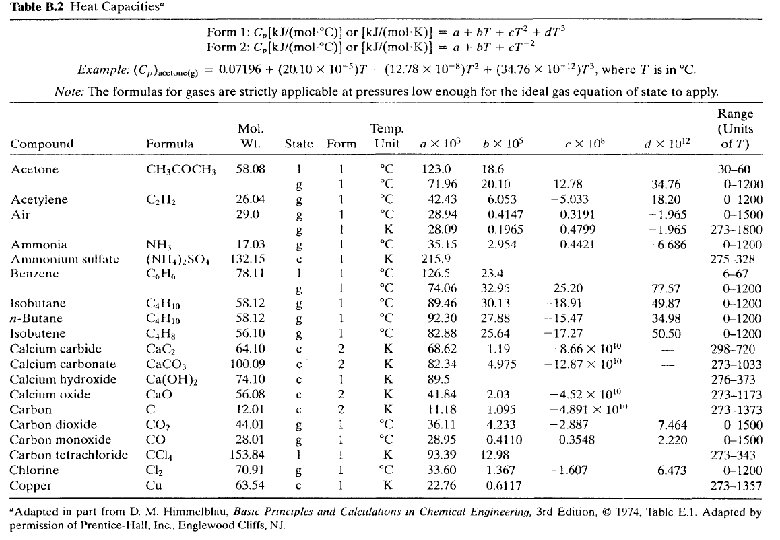
Specific Heat Capacity Formulas Cp = a + b. T + c. T 2 + d. T 3 Ø values for a, b, c and d are obtain from v v Table B. 2 Perry’s Handbook (section 3) Ø When reading the coefficients from Table B. 2 do not mistake their orders of magnitude v 72. 4 is read from column labeled b. 105 means that b = 72. 4 x 10 -5 Ø Simple relationship exist between Cp and Cv for two cases: For liquids and solids: Cp ~ Cv Ideal gases: Cp = Cv + R (R = gas constant)

Specific Heat Capacity Formula Ø The heat required to raise the temperature of m (kg) of a liquid from T 1 to T 2 at constant pressure is (1) The above formula is sometimes simplified as (2)

Example 4: Evaluation of Cp & Cv The constant-pressure heat capacity of carbon monoxide (CO) is given by the expression a. b. Write an expression for the heat capacity at constant volume for CO, assuming ideal gas behaviour Derive an expression for Cp(BTU/Ib-mole. o. F) as a function of T(o. F). (Remember that the temperature unit in the denominator refers to a temperature interval)

Example 4: Evaluation of Cp & Cv a. Write an expression for the heat capacity at constant volume for CO, assuming ideal gas behaviour Gas constant R=1. 987 cal/mol-o. C b. Derive an expression for Cp(BTU/Ib-mole. o. F) as a function of T(o. F). (Remember that the temperature unit in the denominator refers to a temperature interval)

Working session 2 Estimate the specific enthalpy of steam at 350 o. C and 100 bar relative to steam at 100 o. C and 1 atm using a. b. c. The steam tables Table B. 2 and assuming ideal gas behaviour Table B. 8 What is the physical significance of the difference between the values of Ĥ calculated by the two methods?

Example 5 : Evaluation of DH using Heat Capacity Ø Fifteen kmol/min of air is cooled from 430 o. C to 100 o. C. Calculate the required heat removal rate using v v Heat Capacity from Table B. 2 Specific Enthalpies from Table B. 8 Air (g, 430 o. C) Air (g, 100 o. C) With DEk, DEp, and Ws deleted, the energy balance is Ø Assuming ideal gas behavior, so that the pressure changes do not effect specific enthalpy

Solution : The Hard Way (using Heat Capacity from Table B. 2) Ø Integrate the heat capacity formula in Table B. 2

Solution : The Easy Way (using Specific Enthalpies from Table B. 8) Ø Use Tabulated enthalpies form Table B. 8 ( …. What is reference temp. ? ) Ø Table B. 8 and B. 9 apply strictly for heating and cooling at a constant pressure of 1 atm and may also be used for nonisobaric heating and cooling of ideal gas

Example 6 : Evaluation of DU using Heat Capacity relationship Ø Fifteen kmol of air is cooled form 430 o. C to 100 o. C in a constant-volume vessel. Calculate the required heat removal rate using v Cv = Cp - R & Heat Capacity from Table B. 2 Air (g, 430 o. C) Air (g, 100 o. C) With DEk, DEp, and Ws deleted, the energy balance is Ø Assuming ideal gas behavior

Solution : The Hard Way (using CV & Cp relationship and Heat Capacity from Table B. 2) Ø substitute Cv by Cp - R & apply the heat capacity formula in Table B. 2 R (k. J/mol-C)

Solution : The Easy Way (using CV & Cp relationship and Heat Capacities from Table B. 8) Ø Use H = U + PV correlation & Table B. 8

Working session 3 (similar Problem 8. 3) The constant-volume heat capacity of hydrogen sulfide (H 2 S) at low pressures is given by the expression where T is in o. C. A quantity of H 2 S is kept in a piston-fitted cylinder with initial temperature, pressure and volume equal to 25 o. C, 2 atm and 3 liters, respectively. a. b. c. Calculate the heat (k. J) required to raise the gas temperature from 25 o. C to 1000 o. C if the heating takes place at constant volume. Calculate the heat (k. J) required to raise the gas temperature from 25 o. C to 1000 o. C if the heating takes place at constant pressure. What would piston do during the heating process in part (b). What is the physical significance of the difference between the Values of Q calculated in part (a) and part (b)? Home work

Working session 4 - Problem 8. 9 Chlorine gas is to be heated from 100 o. C and 1 atm to 200 o. C a. b. c. Calculate the heat input (k. W) required to heat a stream of the gas flowing at 5 kmol/s at constant pressure Calculate the heat input (k. J) required to raise the temperature of 5 kmol of chlorine in a closed rigid vessel from 100 o. C and 1 atm to 200 o. C What is the physical significance of the numerical difference between tha values calculated in part (a) and part (b)? Home work

Working session 5 - Problem 8. 15 A stream of water vapor flowing at a rate of 250 mol/h is brought from 600 o. C and 10 bar to 100 o. C and 1 atm a. b. c. Estimate the cooling rate (k. W) using (i) steam tables, (ii) Table B. 2 and (iii) Table B. 8 Which of the answers is part (a) is most accurate? Why? What is the physical significance of the numerical difference between the values calculated with methods (i) and (ii)? Home work

Estimation of Heat Capacities Ø You may use the following approximation to find heat capacity if you don’t have enough data: v v v For a mixture of gases and liquids, calculate the total enthalpy change as the sum of the enthalpy changes for the pure mixture components. For a highly dilute solutions of solids or gases in liquids, neglect the enthalpy change of the solute. The more dilute the solution, the better this approximation. The calculation of enthalpy changes for the heating or cooling of a mixture of known composition can be simplified using this formula:

Estimation of Heat Capacities Ø Kopp’s rule is an empirical method for estimating heat capacity of a solid or liquid at or near 20 o. C. Ø Cp of a molecular compound is the sum of contributions of each element in the compound. Ø The values for the coefficients are given in Table B. 10 Ø Heat capacity of solid calcium hydroxide, Ca(OH)2 is estimated as

Example 7 - Heat Capacity of a Mixture Ø Calculate the heat required to bring 150 mol/h of a stream containing 60% C 2 H 6 and 40% C 3 H 8 by mole from 0 o. C to 400 o. C. Determine the heat capacity for the mixture as part of the problem solution. Ø The polynomial heat capacity formula for ethane and propane given in Table B. 2 are substituted:

Ø Alternatively …. calculate DH of each component, multiply by its mole fraction, respectively & then sum them up, i. e Ø If the potential and kinetic energy changes and shaft work are neglected, the energy balance becomes Ø As usual, we assumed that the gases are sufficiently close to ideal for the formula for Cp at 1 atm is valid.
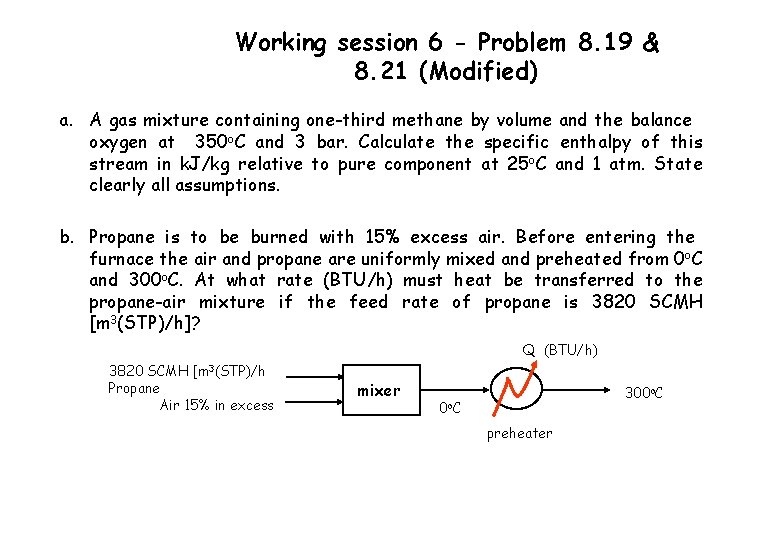
Working session 6 - Problem 8. 19 & 8. 21 (Modified) a. A gas mixture containing one-third methane by volume and the balance oxygen at 350 o. C and 3 bar. Calculate the specific enthalpy of this stream in k. J/kg relative to pure component at 25 o. C and 1 atm. State clearly all assumptions. b. Propane is to be burned with 15% excess air. Before entering the furnace the air and propane are uniformly mixed and preheated from 0 o. C and 300 o. C. At what rate (BTU/h) must heat be transferred to the propane-air mixture if the feed rate of propane is 3820 SCMH [m 3(STP)/h]? Q (BTU/h) 3820 SCMH [m 3(STP)/h Propane Air 15% in excess mixer 300 o. C 0 o C preheater

Working session 6 - 8. 21 (Modified) Molar flow rate of propane , Stoichiometric combustion of propane, C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O Molar flow rate of 15% excess air, Simplified energy bal. around preheater What are reference states for propane & air? How do you calculate the value of Ĥ?
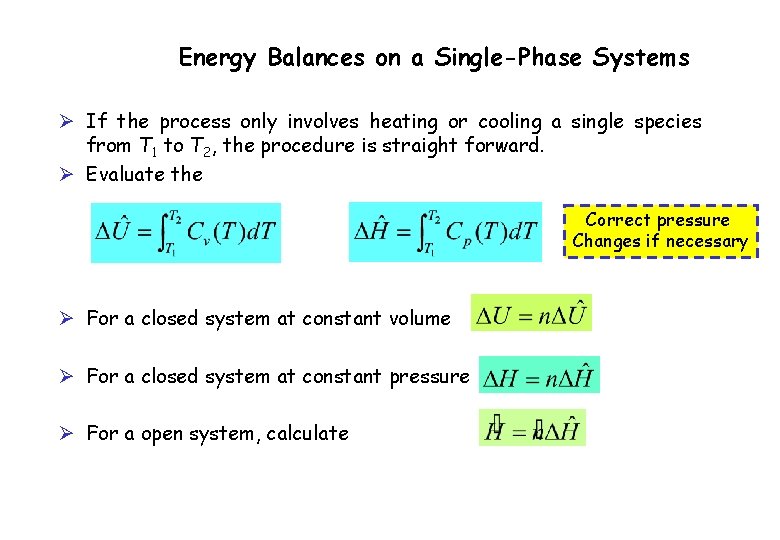
Energy Balances on a Single-Phase Systems Ø If the process only involves heating or cooling a single species from T 1 to T 2, the procedure is straight forward. Ø Evaluate the Correct pressure Changes if necessary Ø For a closed system at constant volume Ø For a closed system at constant pressure Ø For a open system, calculate
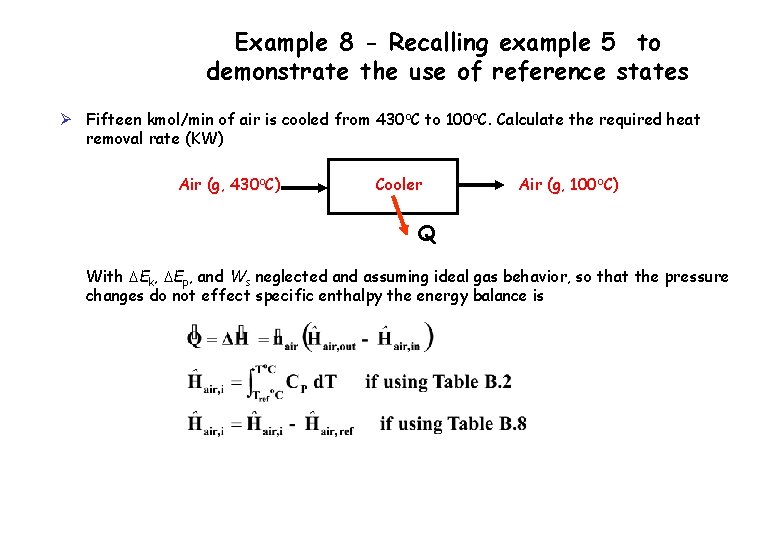
Example 8 - Recalling example 5 to demonstrate the use of reference states Ø Fifteen kmol/min of air is cooled from 430 o. C to 100 o. C. Calculate the required heat removal rate (KW) Air (g, 430 o. C) Cooler Air (g, 100 o. C) Q With DEk, DEp, and Ws neglected and assuming ideal gas behavior, so that the pressure changes do not effect specific enthalpy the energy balance is

Recalling example 5 to demonstrate the use of reference states Reference : Air (g, 25 o. C, 1 atm) Substance nin (kmol/min) Ĥin nout (kmol/min) Ĥout Air 15 Ĥ 1 15 Ĥ 2
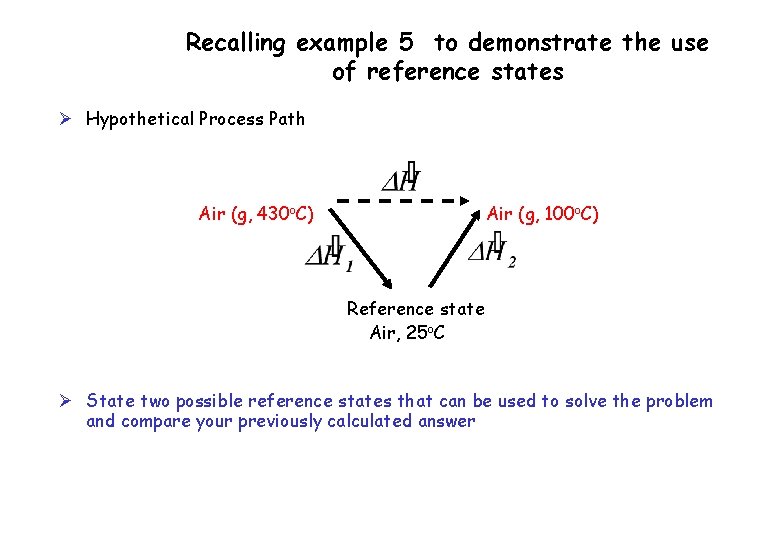
Recalling example 5 to demonstrate the use of reference states Ø Hypothetical Process Path Air (g, 430 o. C) Air (g, 100 o. C) Reference state Air, 25 o. C Ø State two possible reference states that can be used to solve the problem and compare your previously calculated answer
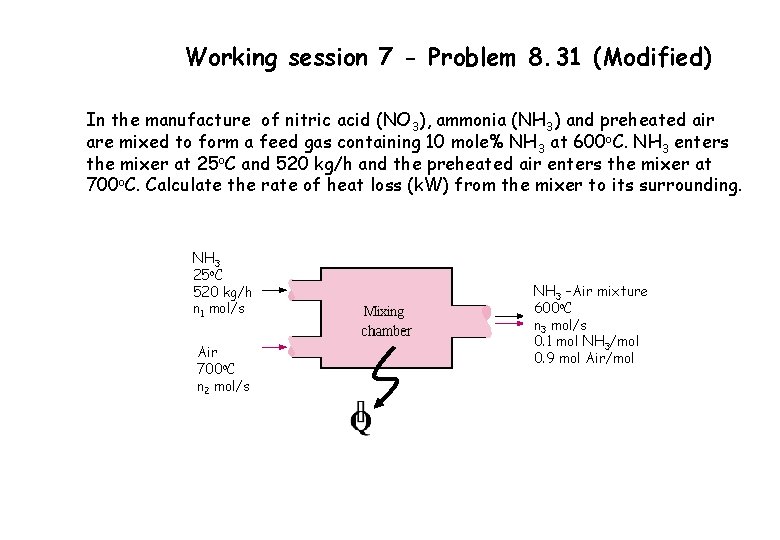
Working session 7 - Problem 8. 31 (Modified) In the manufacture of nitric acid (NO 3), ammonia (NH 3) and preheated air are mixed to form a feed gas containing 10 mole% NH 3 at 600 o. C. NH 3 enters the mixer at 25 o. C and 520 kg/h and the preheated air enters the mixer at 700 o. C. Calculate the rate of heat loss (k. W) from the mixer to its surrounding. NH 3 25 o. C 520 kg/h n 1 mol/s Air 700 o. C n 2 mol/s NH 3 –Air mixture 600 o. C n 3 mol/s 0. 1 mol NH 3/mol 0. 9 mol Air/mol
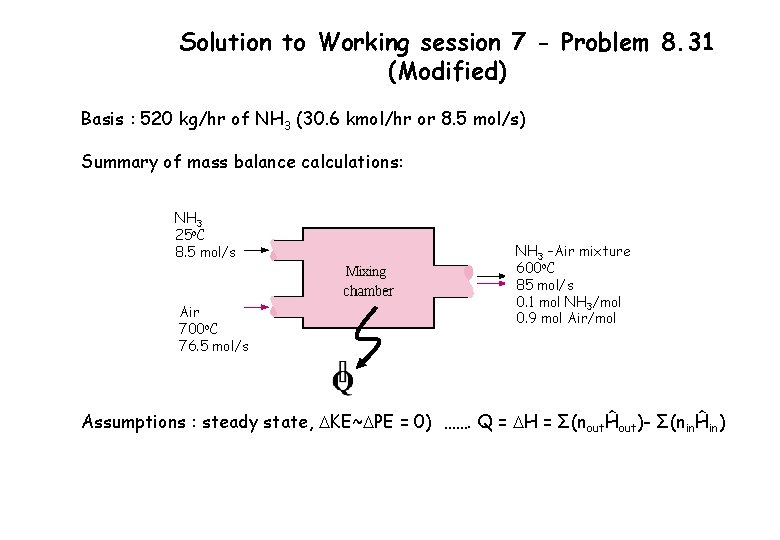
Solution to Working session 7 - Problem 8. 31 (Modified) Basis : 520 kg/hr of NH 3 (30. 6 kmol/hr or 8. 5 mol/s) Summary of mass balance calculations: NH 3 25 o. C 8. 5 mol/s Air 700 o. C 76. 5 mol/s NH 3 –Air mixture 600 o. C 85 mol/s 0. 1 mol NH 3/mol 0. 9 mol Air/mol Assumptions : steady state, DKE~DPE = 0) ……. Q = DH = Σ(noutĤout)- Σ(ninĤin)

Solution to Working session 7 - Problem 8. 31 (Modified) Reference : (Air & NH 3 @25 o. C) Substance nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) NH 3 8. 5 Ĥ 1 8. 5 Ĥ 3 Air 76. 5 Ĥ 2 76. 5 Ĥ 4

Solution to Working session 7 - Problem 8. 31 (Modified)

Solution to Working session 7 - Problem 8. 31 (Modified) Reference : (Air @ 700 o. C & NH 3 @ 25 o. C) Substance nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) NH 3 8. 5 Ĥ 1 8. 5 Ĥ 3 Air 76. 5 Ĥ 2 76. 5 Ĥ 4

Solution to Working session 7 - Problem 8. 31 (Modified)
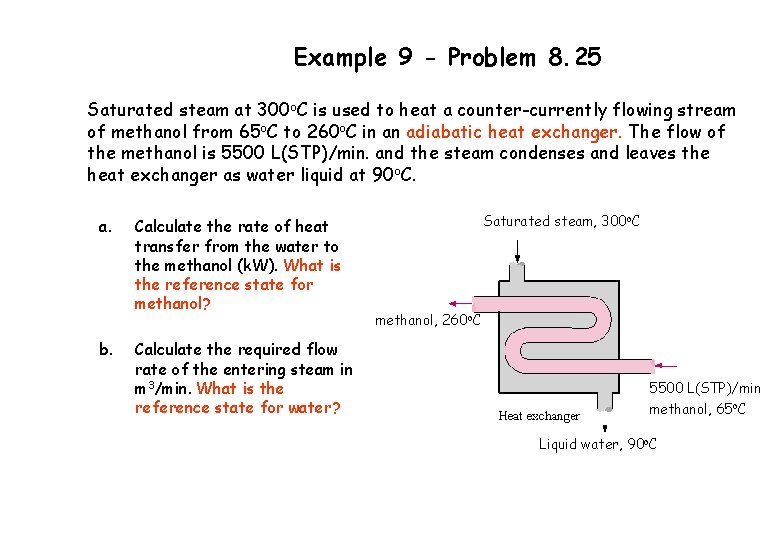
Example 9 - Problem 8. 25 Saturated steam at 300 o. C is used to heat a counter-currently flowing stream of methanol from 65 o. C to 260 o. C in an adiabatic heat exchanger. The flow of the methanol is 5500 L(STP)/min. and the steam condenses and leaves the heat exchanger as water liquid at 90 o. C. a. b. Calculate the rate of heat transfer from the water to the methanol (k. W). What is the reference state for methanol? Calculate the required flow rate of the entering steam in m 3/min. What is the reference state for water? Saturated steam, 300 o. C methanol, 260 o. C 5500 L(STP)/min methanol, 65 o. C Liquid water, 90 o. C
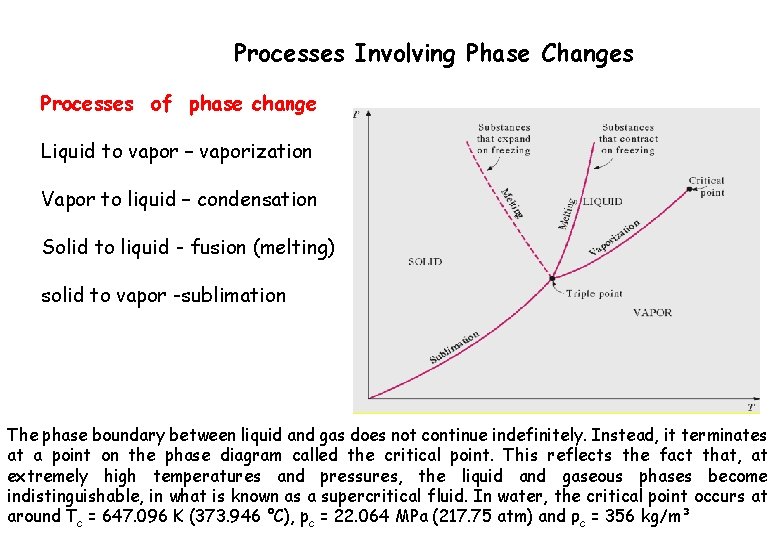
Processes Involving Phase Changes Processes of phase change Liquid to vapor – vaporization Vapor to liquid – condensation Solid to liquid - fusion (melting) solid to vapor -sublimation The phase boundary between liquid and gas does not continue indefinitely. Instead, it terminates at a point on the phase diagram called the critical point. This reflects the fact that, at extremely high temperatures and pressures, the liquid and gaseous phases become indistinguishable, in what is known as a supercritical fluid. In water, the critical point occurs at around Tc = 647. 096 K (373. 946 °C), pc = 22. 064 MPa (217. 75 atm) and ρc = 356 kg/m³

Latent heat § Heat required to transform a substance from one phase to another phase at constant temperature or pressure § Latent heat of 1. vaporization (liquid vapor), DĤv 2. fusion or melting (solid liquid), DĤm 3. sublimation (solid vapor), DĤs Table B. 1 has some values at normal conditions (1 atm) & Perry’s Chemical Handbook
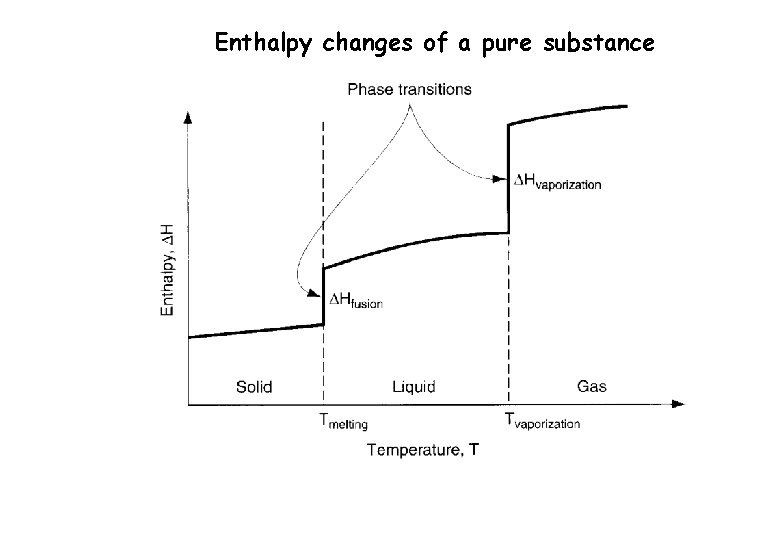
Enthalpy changes of a pure substance
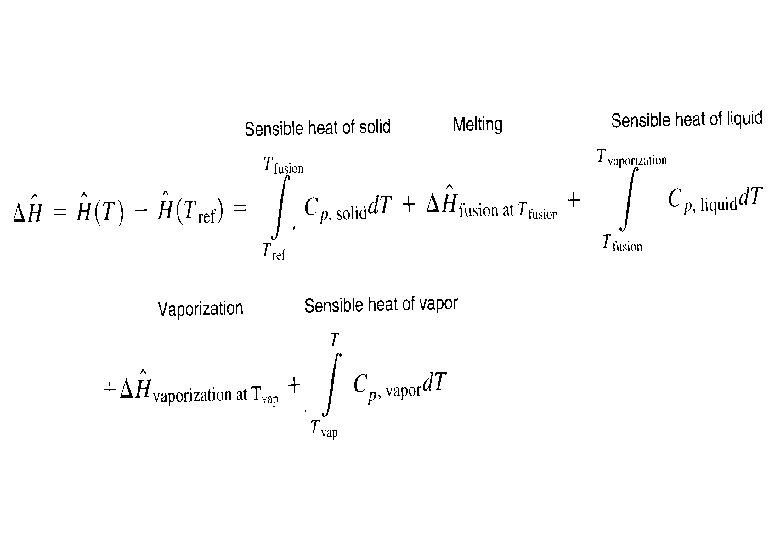

§ If a phase change takes place in an open system, the heat of vaporization (DĤvap) of a substance may be determined from Table B. 1 (for normal boilng point) or estimated using Trouton’s rule, Chen’s equation, Clausius-Clapeyron equation or Watson’s correlation § If a phase change takes place in a closed system, DÛ will be used to substitute into the energy balance equation assuming ideal gas behaviour § For phase change such as fusion involving only solids or liquids, changes in is usually negligible, so DÛ ≈ DĤ
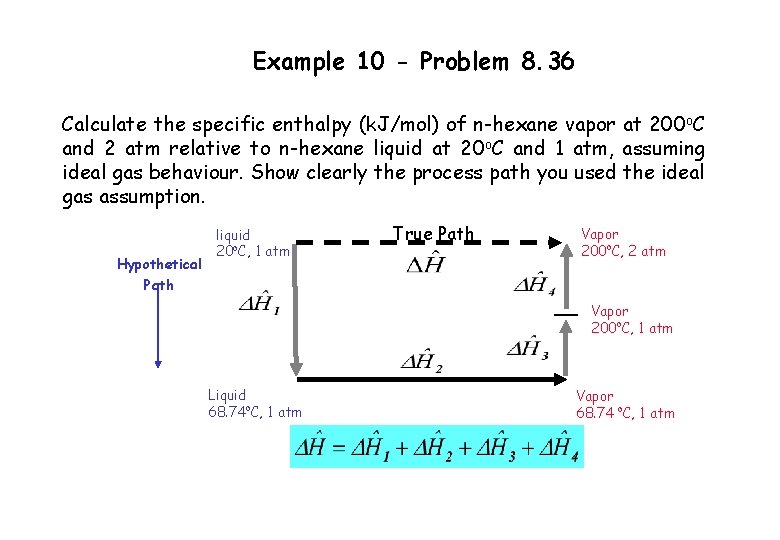
Example 10 - Problem 8. 36 Calculate the specific enthalpy (k. J/mol) of n-hexane vapor at 200 o. C and 2 atm relative to n-hexane liquid at 20 o. C and 1 atm, assuming ideal gas behaviour. Show clearly the process path you used the ideal gas assumption. Hypothetical Path liquid 20 o. C, 1 atm True Path Vapor 200 o. C, 2 atm Vapor 200 o. C, 1 atm Liquid 68. 74 o. C, 1 atm Vapor 68. 74 o. C, 1 atm
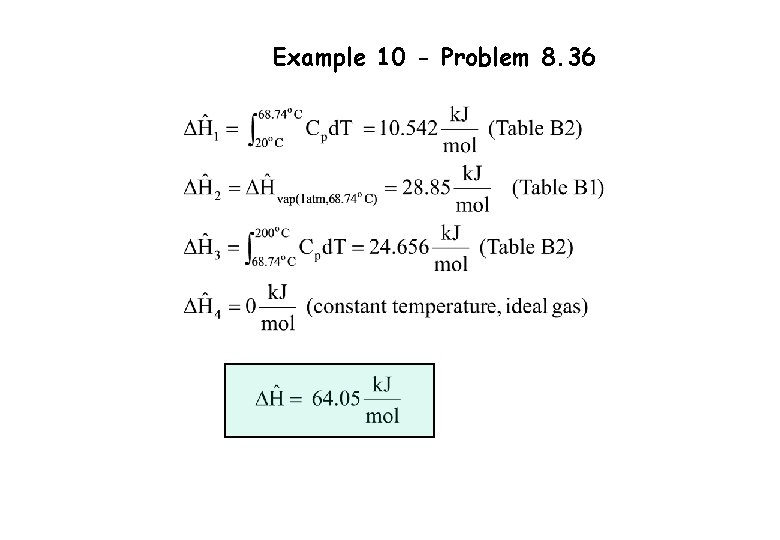
Example 10 - Problem 8. 36
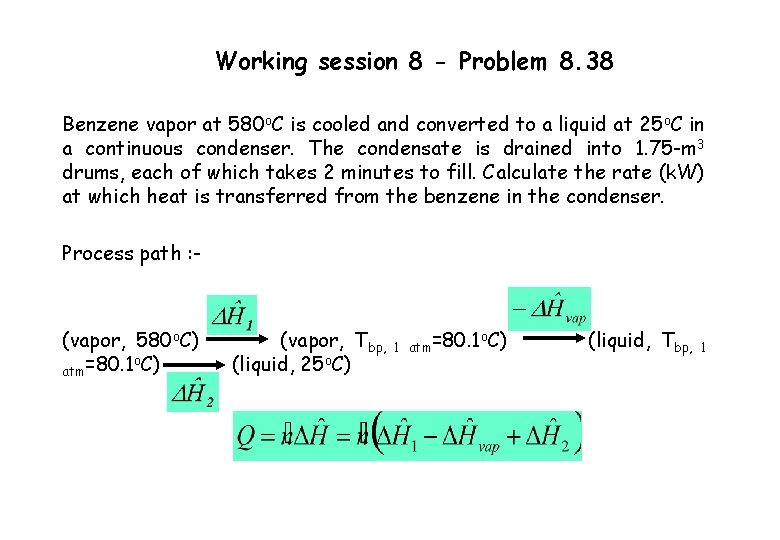
Working session 8 - Problem 8. 38 Benzene vapor at 580 o. C is cooled and converted to a liquid at 25 o. C in a continuous condenser. The condensate is drained into 1. 75 -m 3 drums, each of which takes 2 minutes to fill. Calculate the rate (k. W) at which heat is transferred from the benzene in the condenser. Process path : - (vapor, 580 o. C) o atm=80. 1 C) (vapor, Tbp, (liquid, 25 o. C) 1 atm=80. 1 o. C) (liquid, Tbp, 1

Solution to Working session 8 - Problem 8. 38

Estimation & correlation of latent heats • There are some estimation methods on pp. 381 -2 1. Trouton’s rule – estimate a standard latent heat of vaporization at Accurate to within 30% the normal boiling point Chen’s equation where Tb is the normal boiling point of the liquid provides roughly 2% accuracy where Tb and Tc are the normal boiling point and critical temperature in kelvin and Pc is the critical pressure in atmospheres.

2. Clausius-Clapeyron equation 3. Watson’s correlation

Example 11 - Problem 8. 44 Estimate the heat of vaporization (k. J/mol) of benzene at a pressure of 100 mm Hg, using a. The normal boiling point given in Table B. 1, the boiling point at 100 mm Hg as determined by the Antoine equation and Watson’s correlation

Example 11 - Problem 8. 44 Estimate the heat of vaporization (k. J/mol) of benzene at a pressure of 100 mm Hg, using b. The Clausius-Clayperon equation and the boiling points at 50 mm Hg and 150 mm Hg as determined from the Antoine equation

Example 11 - Problem 8. 44 Estimate the heat of vaporization (k. J/mol) of benzene at a pressure of 100 mm Hg, using c. The normal boiling point given in Table B. 1, the boiling point at 100 mm Hg and the heat capacity data given in Table B. 2 liquid 26. 1 o. C, 100 mm Hg liquid 26. 1 o. C, 760 mm Hg liquid 80. 1 o. C, 760 mm Hg True Path Vapor 26. 1 o. C, 100 mm Hg Vapor 26. 1 o. C, 760 mm Hg Vapor 80. 1 o. C, 760 mm Hg

Example 11 - Problem 8. 44

Energy Balances on Process involving Phase Changes § An energy balance on a process in which a component exists in two phases § Choose a reference state for that component by specifying both a phase and a temperature § Calculate the specific enthalpy of the component in all process streams relative to a reference state § If the substance is a liquid at its reference state a vapor in a process stream, Ĥ may be calculated using process path calculations

Example 12 - Problem 8. 50 (modified) A mixture of n-hexane vapor and air leaves a solvent recovery unit and flows through a 70 cm-diameter duct at a velocity of 3 m/s. At sampling point in the duct the temperature is 40 o. C, the pressure is 850 mm Hg, and the dew point of the gas sample is 25 o. C. The gas is fed to a condenser in which it is cooled at constant pressure, condensing 60% of the hexane in the feed. Calculate the required condenser outlet temperature and cooling rate (KW). 70 cm-ID duct V=3 m/s T= 40 o. C P= 850 mm Hg Tdp= 25 o. C nf = ? mol/s Yh, f=? mol hexane/mol Yair, f=? mol air/mol condenser 60% of Hexane feed nhex = ? mol/s P= 850 mm Hg Tc = ? o. C nc = ? mol/s Yh, c=? mol hexane/mol Yair, c=? Mol air/mol

Problem 8. 50 (modified) Assuming an ideal gas air-hexane mixture entering the condenser, nf = PV/RT = 50. 3 mol/s Tdp= 25 o. C , thus the partial pressure of condensable species (i. e. hexane), using Antoine equation, p*h = 151. 3 mm Hg hence, p*h = Yh, f P …… Yh, f= 0. 178 mol hexane/mol and Yair, f= 0. 822 mol air/mol then, nhex = (0. 6)(0. 178)(50. 3) = 5. 372 mol/s hexane condensed ! and nc = 50. 3 – 5. 372 = 44. 928 mol/s Yair, c= (0. 822)(50. 3)/44. 928 = 0. 92 mol air/mol Yh, c= 0. 08 mol hexane/mol p*h = Yh, f P …… Yh, f= (0. 08)(850) = 68 mm Hg from Antoine equation, Tc(p*h = 68 mm Hg ) = 7. 83 o. C

Problem 8. 50 (modified) Energy balance for open system, neglecting changes in kinetic and potential energy and enthalpy is independent of pressure change, References : Hexane ( liquid, 7. 8 o. C), Air (gas, 25 o. C) Substance nin (mol/s) Hexane (v) 8. 95 Hexane (l) - Air 41. 35 Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) 3. 58 - 5. 37 41. 35 0

Problem 8. 50 (modified) Inlet stream Ĥhexane (v) = = 13. 181 + 28. 85 – 4. 568 = 37. 463 k. J/mol Ĥair = = 0. 436 k. J/mol (Table B. 2) or alternatively Table B. 8 Outlet stream Ĥair = = - 0. 499 k. J/mol Ĥhexane (v) = = 13. 181 + 28. 85 – 9. 303 = 32. 728 k. J/mol Ĥhexane (l) = 0 k. J/mol
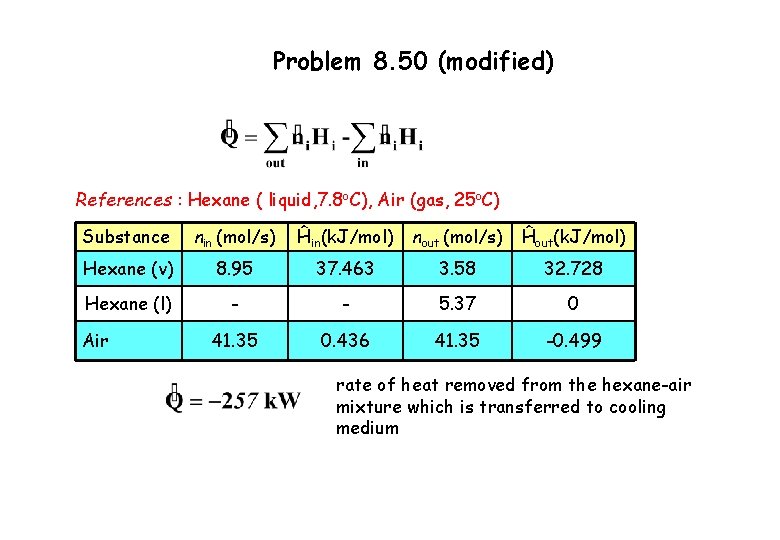
Problem 8. 50 (modified) References : Hexane ( liquid, 7. 8 o. C), Air (gas, 25 o. C) Substance nin (mol/s) Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) Hexane (v) 8. 95 37. 463 3. 58 32. 728 Hexane (l) - - 5. 37 0 41. 35 0. 436 41. 35 -0. 499 Air rate of heat removed from the hexane-air mixture which is transferred to cooling medium

Problem 8. 50 (modified) – using different reference states Recalculate the required cooling rate using different reference state, e. g. 40 o. C and gas phase for both substances References : Hexane (gas, 40 o. C), Air (gas, 40 o. C) Substance nin (mol/s) Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) Hexane (v) 8. 95 ? 3. 58 ? Hexane (l) - - 5. 37 ? 41. 35 ? Air

Problem 8. 50 (modified) Inlet stream Ĥhexane (v) = 0 k. J/mol Ĥair = 0 k. J/mol Outlet stream Ĥair = = - 0. 935 k. J/mol Ĥhexane (l) = = 4. 568 - 28. 85 – 13. 181 = -37. 463 k. J/mol Ĥhexane (v) = = -4. 735 k. J/mol

Problem 8. 50 (modified) – using different reference states Recalculate the required cooling rate using different reference state, e. g. 40 o. C and gas phase for both substances References : Hexane (gas, 40 o. C), Air (gas, 40 o. C) Substance nin (mol/s) Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) Hexane (v) 8. 95 0 3. 58 -4. 735 Hexane (l) - - 5. 37 -37. 463 41. 35 0 41. 35 -0. 935 Air
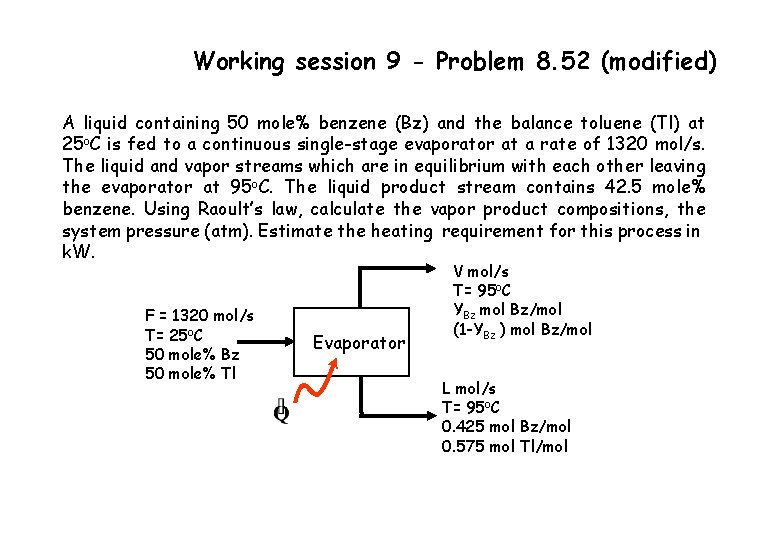
Working session 9 - Problem 8. 52 (modified) A liquid containing 50 mole% benzene (Bz) and the balance toluene (Tl) at 25 o. C is fed to a continuous single-stage evaporator at a rate of 1320 mol/s. The liquid and vapor streams which are in equilibrium with each other leaving the evaporator at 95 o. C. The liquid product stream contains 42. 5 mole% benzene. Using Raoult’s law, calculate the vapor product compositions, the system pressure (atm). Estimate the heating requirement for this process in k. W. F = 1320 mol/s T= 25 o. C 50 mole% Bz 50 mole% Tl Evaporator V mol/s T= 95 o. C YBz mol Bz/mol (1 -YBz ) mol Bz/mol L mol/s T= 95 o. C 0. 425 mol Bz/mol 0. 575 mol Tl/mol

Working session 9 - Problem 8. 52 (modified) Raoult’s law, Yi P = Xi p*i (T) Using Antoine eq. to estimate the partial pressure of ; p*Bz(95 o. C)= 1176. 6 mm Hg Benzene ; Toluene; and p*Tl(95 o. C)= 476. 9 mm Hg YBz P = 0. 425 (1176. 6) ……. . (1) YTl P = 0. 575 (476. 9) …………(2) YBz + YTl = 1 ……………(3) Solve eqs. (1)/(2) & (3) , YBz = 0. 646 YTl = 0. 354 Hence, P = 774 mm Hg (1. 018 atm), V = 448 mol/s , L=872 mol/s
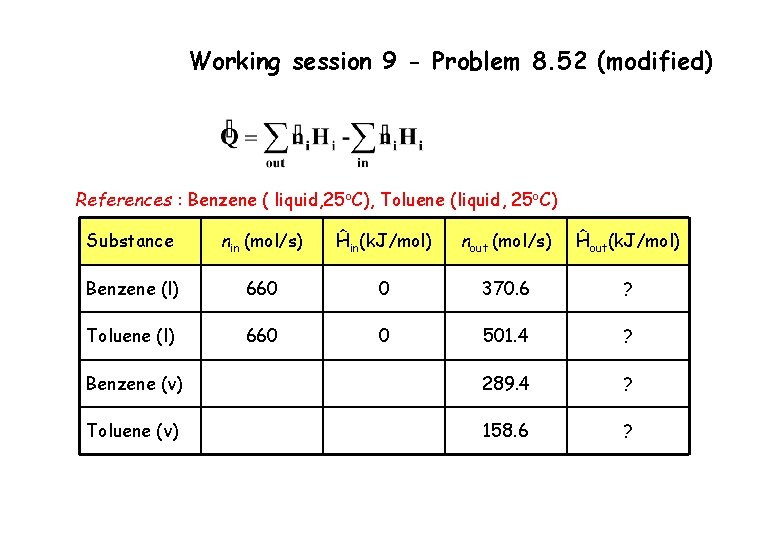
Working session 9 - Problem 8. 52 (modified) References : Benzene ( liquid, 25 o. C), Toluene (liquid, 25 o. C) Substance nin (mol/s) Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) Benzene (l) 660 0 370. 6 ? Toluene (l) 660 0 501. 4 ? Benzene (v) 289. 4 ? Toluene (v) 158. 6 ?

Working session 9 - Problem 8. 52 (modified) Outlet liquid stream ĤBenzene (l) = ĤToluene (l) = = 9. 838 k. J/mol = 11. 777 k. J/mol Outlet vapor stream ĤBenzene (v) = = 7. 633 + 30. 765 + 1. 515 = 39. 913 k. J/mol ĤToluene (v) = = 14. 618 + 33. 47 – 2. 034 = 46. 054 k. J/mol
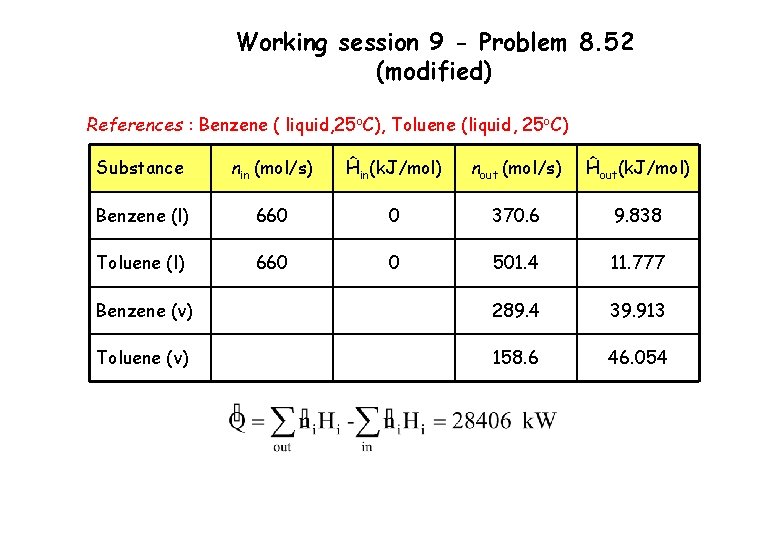
Working session 9 - Problem 8. 52 (modified) References : Benzene ( liquid, 25 o. C), Toluene (liquid, 25 o. C) Substance nin (mol/s) Ĥin(k. J/mol) nout (mol/s) Ĥout(k. J/mol) Benzene (l) 660 0 370. 6 9. 838 Toluene (l) 660 0 501. 4 11. 777 Benzene (v) 289. 4 39. 913 Toluene (v) 158. 6 46. 054
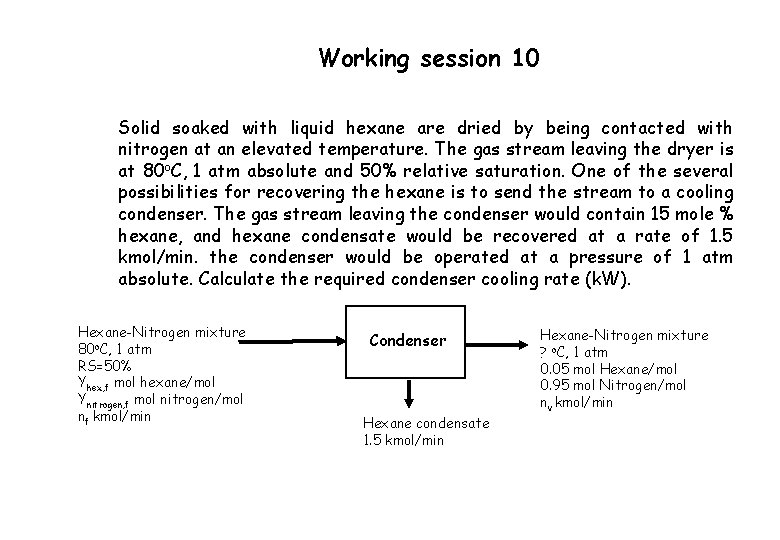
Working session 10 Solid soaked with liquid hexane are dried by being contacted with nitrogen at an elevated temperature. The gas stream leaving the dryer is at 80 o. C, 1 atm absolute and 50% relative saturation. One of the several possibilities for recovering the hexane is to send the stream to a cooling condenser. The gas stream leaving the condenser would contain 15 mole % hexane, and hexane condensate would be recovered at a rate of 1. 5 kmol/min. the condenser would be operated at a pressure of 1 atm absolute. Calculate the required condenser cooling rate (k. W). Hexane-Nitrogen mixture 80 o. C, 1 atm RS=50% Yhex, f mol hexane/mol Ynitrogen, f mol nitrogen/mol nf kmol/min Condenser Hexane condensate 1. 5 kmol/min Hexane-Nitrogen mixture ? o. C, 1 atm 0. 05 mol Hexane/mol 0. 95 mol Nitrogen/mol nv kmol/min

Working session 10 Relative saturation, RS= pi/pi*(T) Inlet stream The only condensable species is hexane, thus RS = 0. 5 = p hex/phex*(80 o. C) Using Antoine equation, p*hex = 1068. 4 mm Hg, and hence, phex = 534. 2 mm Hg Yhex, f= 0. 703 mol hexane/mol and Ynitrogen, f= 0. 297 mol nitrogen/mol Outlet stream Assuming the condenser is in thermal equilibrium, p*hex = Yhex P …… Yh, f= (0. 15)(760) = 114 mm Hg from Antoine equation, Tc(p*h = 114 mm Hg ) = 18. 64 o. C
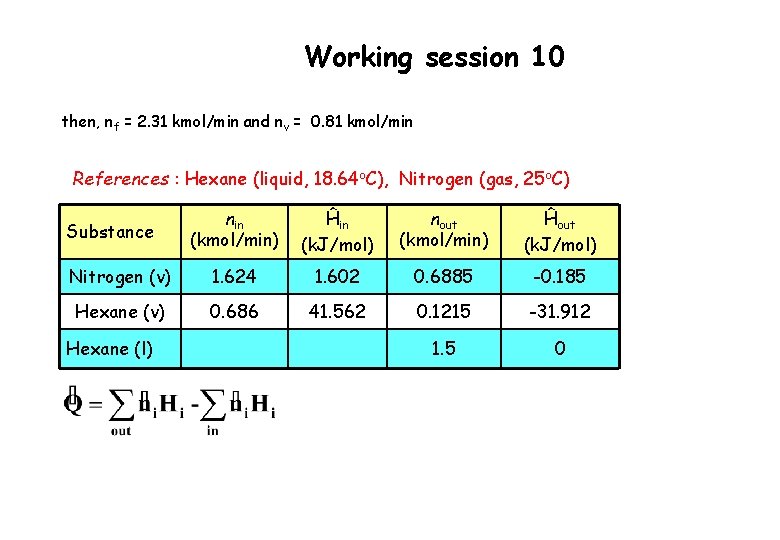
Working session 10 then, nf = 2. 31 kmol/min and nv = 0. 81 kmol/min References : Hexane (liquid, 18. 64 o. C), Nitrogen (gas, 25 o. C) nin (kmol/min) Ĥin (k. J/mol) nout (kmol/min) Ĥout (k. J/mol) Nitrogen (v) 1. 624 1. 602 0. 6885 -0. 185 Hexane (v) 0. 686 41. 562 0. 1215 -31. 912 1. 5 0 Substance Hexane (l)

Working session 10 Inlet stream Ĥhexane(v) = = 10. 837 + 28. 85 1. 875 = 41. 562 k. J/mol Ĥnitrogen = = 1. 602 k. J/mol (Table B. 2) or alternatively Table B. 8 Outlet stream Ĥnitrogen = = - 0. 185 k. J/mol Ĥhexane (v) = = 10. 837 + 28. 85 – 7. 7755 = 31. 912 k. J/mol Ĥhexane (l) = 0 k. J/mol
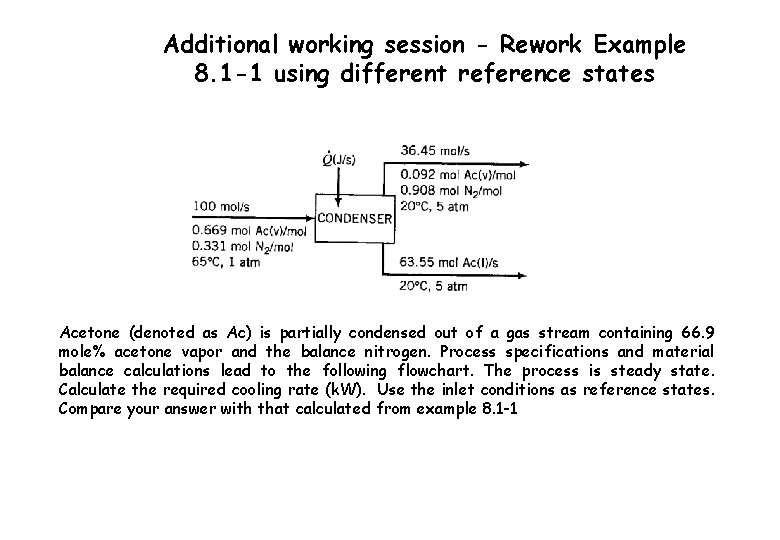
Additional working session - Rework Example 8. 1 -1 using different reference states Acetone (denoted as Ac) is partially condensed out of a gas stream containing 66. 9 mole% acetone vapor and the balance nitrogen. Process specifications and material balance calculations lead to the following flowchart. The process is steady state. Calculate the required cooling rate (k. W). Use the inlet conditions as reference states. Compare your answer with that calculated from example 8. 1 -1
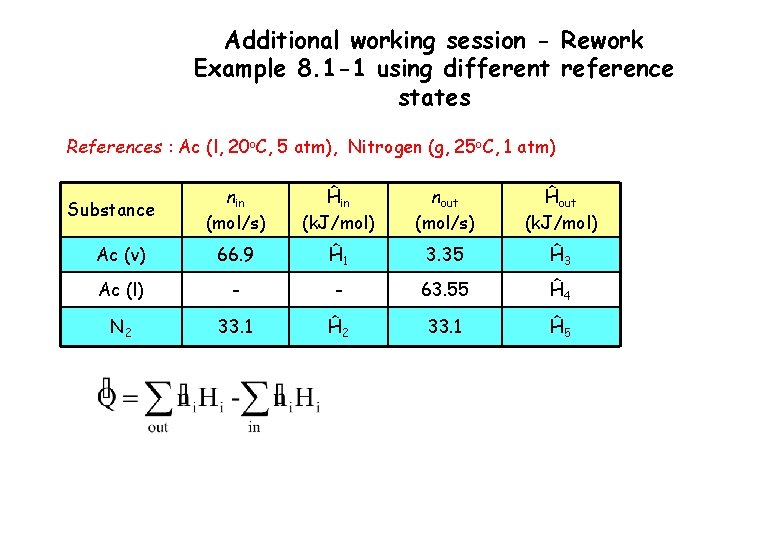
Additional working session - Rework Example 8. 1 -1 using different reference states References : Ac (l, 20 o. C, 5 atm), Nitrogen (g, 25 o. C, 1 atm) nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) Ac (v) 66. 9 Ĥ 1 3. 35 Ĥ 3 Ac (l) - - 63. 55 Ĥ 4 N 2 33. 1 Ĥ 5 Substance

Additional working session - Rework Example 8. 1 -1 using different reference states
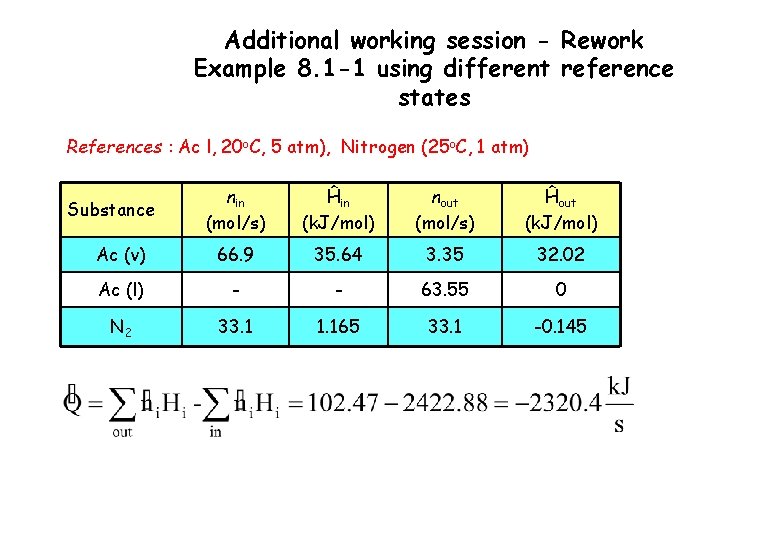
Additional working session - Rework Example 8. 1 -1 using different reference states References : Ac l, 20 o. C, 5 atm), Nitrogen (25 o. C, 1 atm) nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) Ac (v) 66. 9 35. 64 3. 35 32. 02 Ac (l) - - 63. 55 0 N 2 33. 1 1. 165 33. 1 -0. 145 Substance
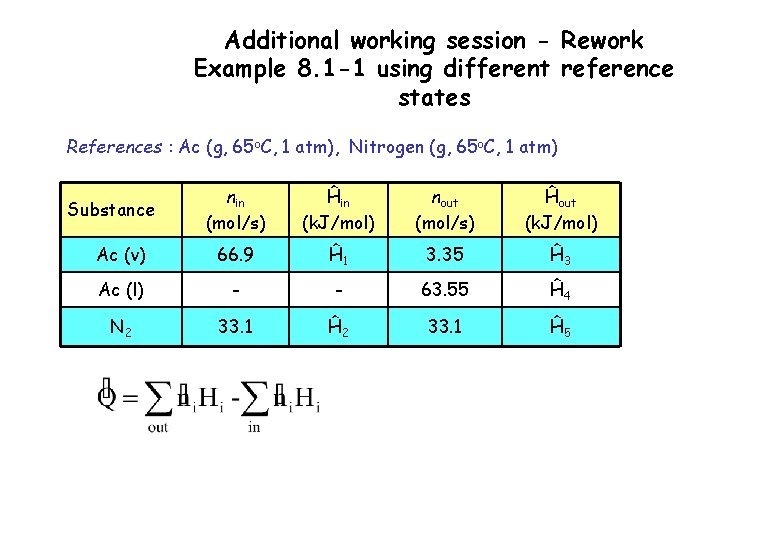
Additional working session - Rework Example 8. 1 -1 using different reference states References : Ac (g, 65 o. C, 1 atm), Nitrogen (g, 65 o. C, 1 atm) nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) Ac (v) 66. 9 Ĥ 1 3. 35 Ĥ 3 Ac (l) - - 63. 55 Ĥ 4 N 2 33. 1 Ĥ 5 Substance

Additional working session - Rework Example 8. 1 -1 using different reference states
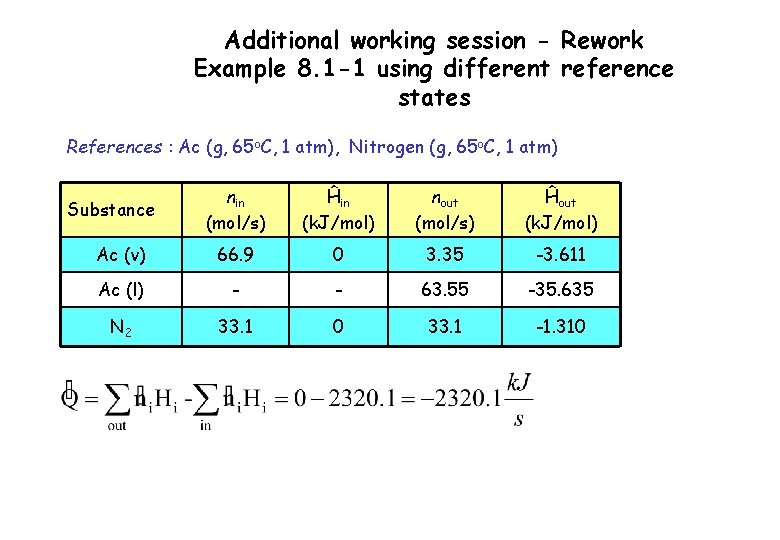
Additional working session - Rework Example 8. 1 -1 using different reference states References : Ac (g, 65 o. C, 1 atm), Nitrogen (g, 65 o. C, 1 atm) nin (mol/s) Ĥin (k. J/mol) nout (mol/s) Ĥout (k. J/mol) Ac (v) 66. 9 0 3. 35 -3. 611 Ac (l) - - 63. 55 -35. 635 N 2 33. 1 0 33. 1 -1. 310 Substance
- Slides: 102