Balanced Bone Remodeling Resorption Rate Formation Rate No

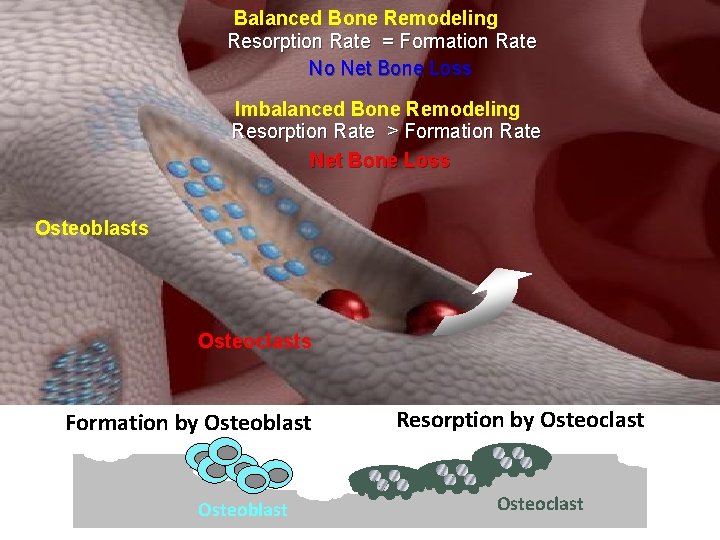
Balanced Bone Remodeling Resorption Rate = Formation Rate No Net Bone Loss Imbalanced Bone Remodeling Resorption Rate > Formation Rate Net Bone Loss Osteoblasts Osteoclasts Formation by Osteoblast Resorption by Osteoclast
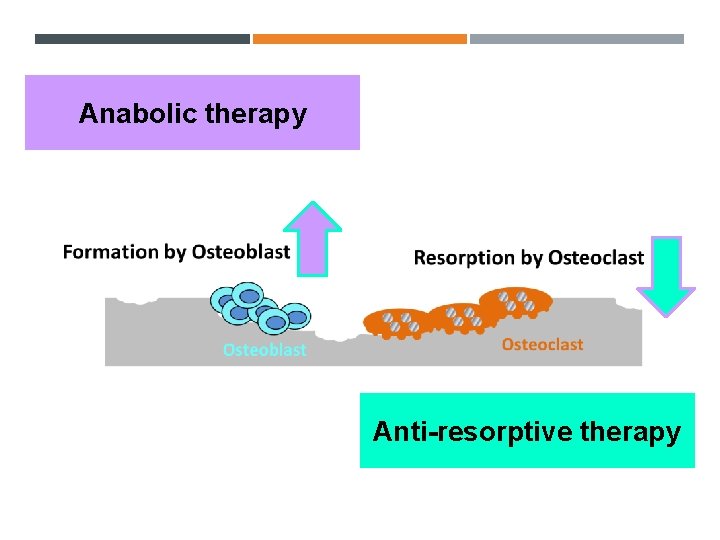
Anabolic therapy Anti-resorptive therapy
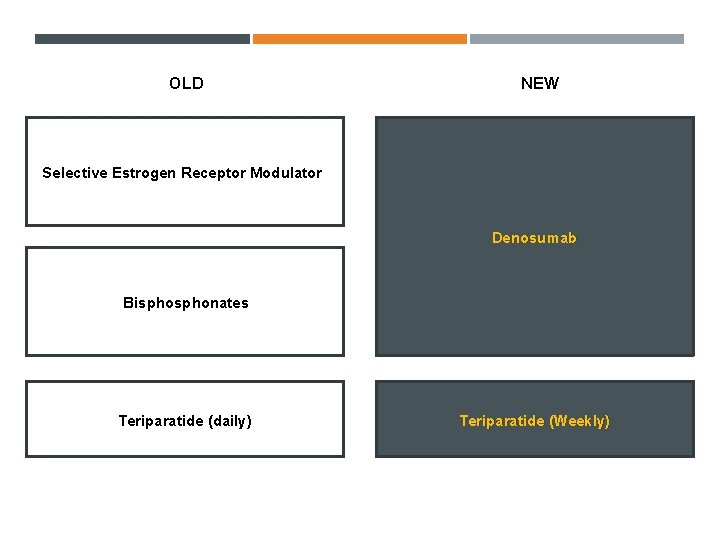
OLD NEW Selective Estrogen Receptor Modulator Denosumab Bisphonates Teriparatide (daily) Teriparatide (Weekly)
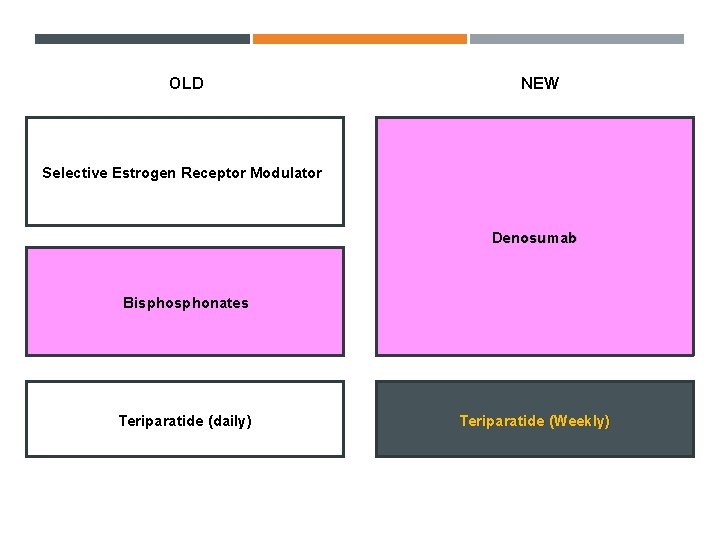
OLD NEW Selective Estrogen Receptor Modulator Denosumab Bisphonates Teriparatide (daily) Teriparatide (Weekly)

DENOSUMAB
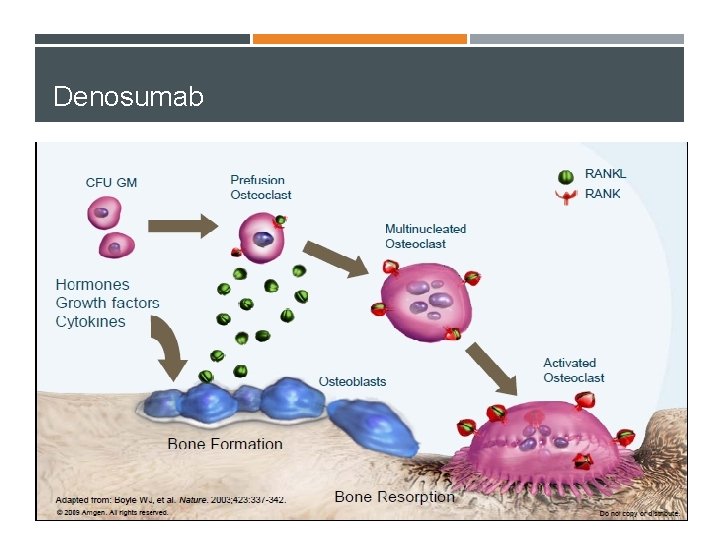
Denosumab
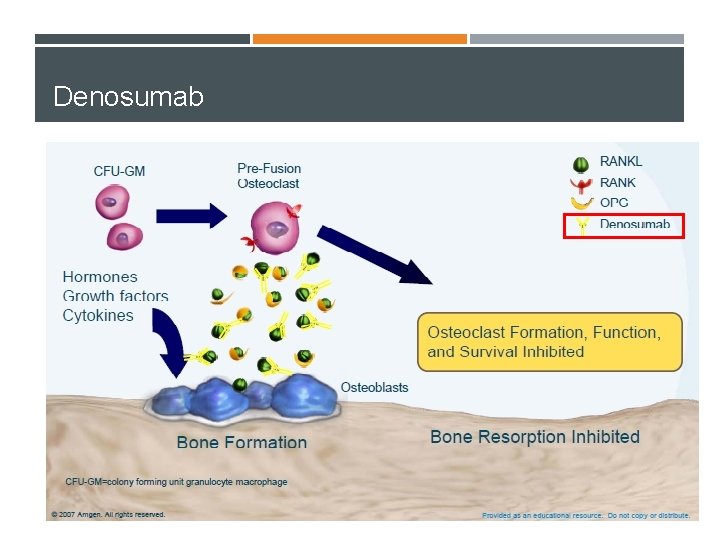
Denosumab
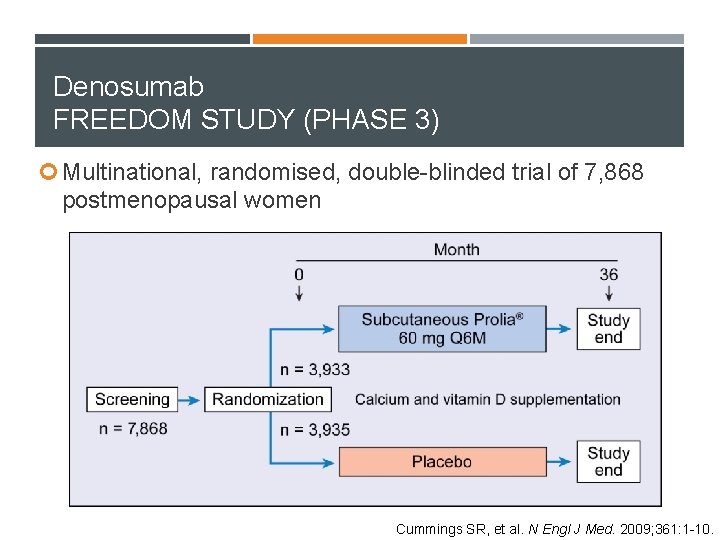
Denosumab FREEDOM STUDY (PHASE 3) Multinational, randomised, double-blinded trial of 7, 868 postmenopausal women Cummings SR, et al. N Engl J Med. 2009; 361: 1 -10.
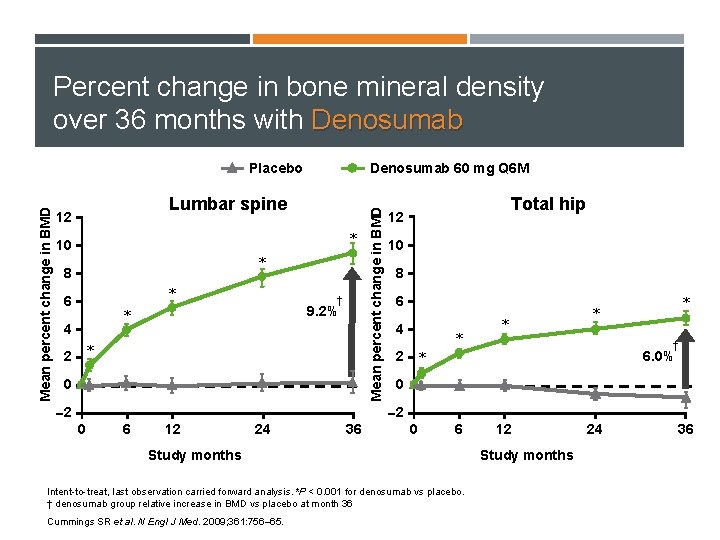
Percent change in bone mineral density over 36 months with Denosumab 60 mg Q 6 M Lumbar spine 12 * 10 * 8 * 6 † 9. 2% * 4 2 * 0 2 Mean percent change in BMD Placebo Total hip 12 10 8 6 4 * * † 6. 0% 2 * 0 2 0 6 12 24 36 0 6 Study months Intent-to-treat, last observation carried forward analysis. *P < 0. 001 for denosumab vs placebo. † denosumab group relative increase in BMD vs placebo at month 36 Cummings SR et al. N Engl J Med. 2009; 361: 756– 65. 12 Study months 24 36
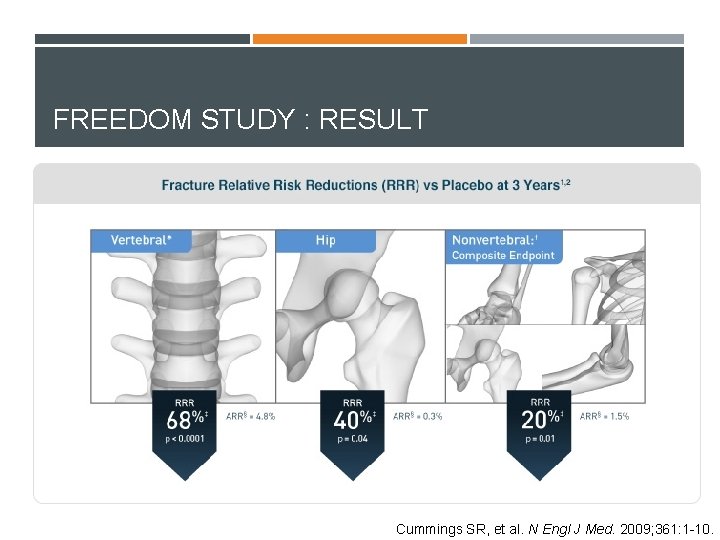
FREEDOM STUDY : RESULT Cummings SR, et al. N Engl J Med. 2009; 361: 1 -10.
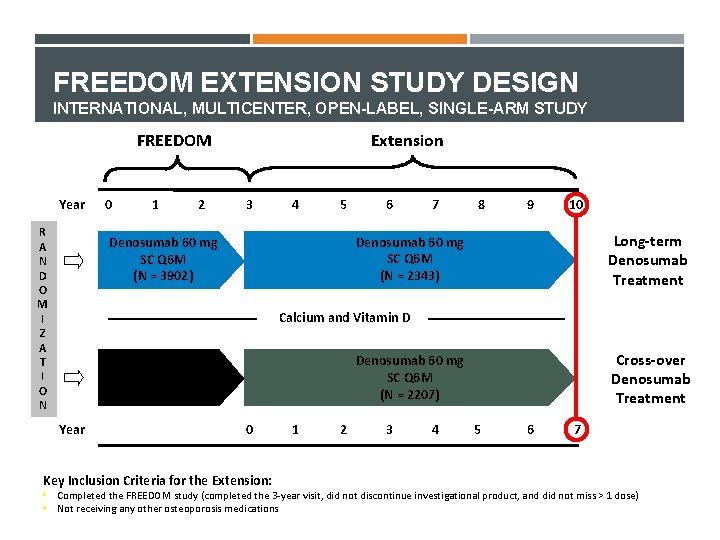
FREEDOM EXTENSION STUDY DESIGN INTERNATIONAL, MULTICENTER, OPEN-LABEL, SINGLE-ARM STUDY FREEDOM Year R A N D O M I Z A T I O N 0 1 2 Extension 3 4 5 6 7 8 9 10 Long-term Denosumab Treatment Denosumab 60 mg SC Q 6 M (N = 2343) Denosumab 60 mg SC Q 6 M (N = 3902) Calcium and Vitamin D Placebo SC Q 6 M (N = 3906) Year Cross-over Denosumab Treatment Denosumab 60 mg SC Q 6 M (N = 2207) 0 Key Inclusion Criteria for the Extension: 1 2 3 4 5 6 7 • Completed the FREEDOM study (completed the 3 -year visit, did not discontinue investigational product, and did not miss > 1 dose) • Not receiving any other osteoporosis medications
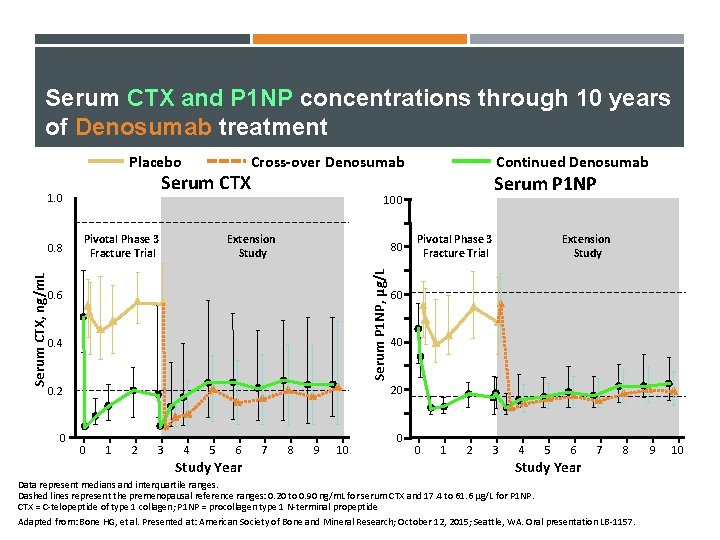
Serum CTX and P 1 NP concentrations through 10 years of Denosumab treatment Placebo Serum CTX 1. 0 Extension Study 80 Serum P 1 NP, μg/L Pivotal Phase 3 Fracture Trial 0. 6 0. 4 Serum P 1 NP Pivotal Phase 3 Fracture Trial Extension Study 60 40 20 0. 2 0 Continued Denosumab 100 Serum CTX, ng/m. L 0. 8 Cross-over Denosumab 0 1 2 3 4 5 6 Study Year 7 8 9 10 0 0 1 2 3 4 5 6 7 8 Study Year Data represent medians and interquartile ranges. Dashed lines represent the premenopausal reference ranges: 0. 20 to 0. 90 ng/m. L for serum CTX and 17. 4 to 61. 6 µg/L for P 1 NP. CTX = C-telopeptide of type 1 collagen; P 1 NP = procollagen type 1 N-terminal propeptide Adapted from: Bone HG, et al. Presented at: American Society of Bone and Mineral Research; October 12, 2015; Seattle, WA. Oral presentation LB-1157. 9 10
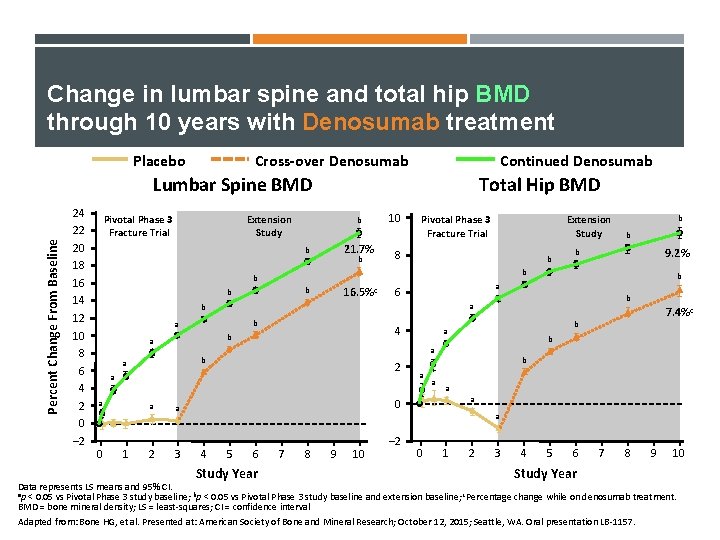
Change in lumbar spine and total hip BMD through 10 years with Denosumab treatment Placebo Cross-over Denosumab Continued Denosumab Percent Change From Baseline Lumbar Spine BMD 24 22 20 18 16 14 12 10 8 6 4 2 0 – 2 Pivotal Phase 3 Fracture Trial Total Hip BMD Extension Study b 21. 7% b b 10 Pivotal Phase 3 Fracture Trial 8 b 16. 5%c b b b 6 4 b 0 1 a a 2 3 b b 7. 4%c b a 2 a a 9. 2% b a b a b b b a Extension Study a a b a a 0 a 4 5 6 Study Year 7 8 9 10 – 2 0 1 2 3 4 5 6 Study Year 7 8 9 10 Data represents LS means and 95% CI. ap < 0. 05 vs Pivotal Phase 3 study baseline; bp < 0. 05 vs Pivotal Phase 3 study baseline and extension baseline; c. Percentage change while on denosumab treatment. BMD = bone mineral density; LS = least-squares; CI = confidence interval Adapted from: Bone HG, et al. Presented at: American Society of Bone and Mineral Research; October 12, 2015; Seattle, WA. Oral presentation LB-1157.
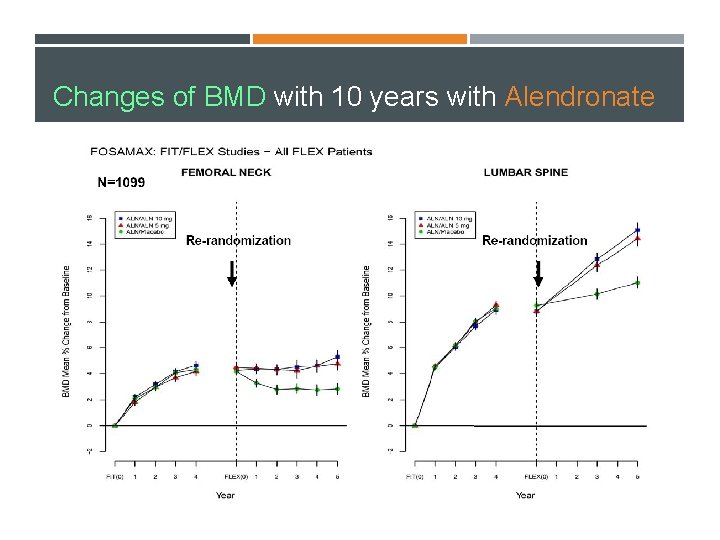
Changes of BMD with 10 years with Alendronate
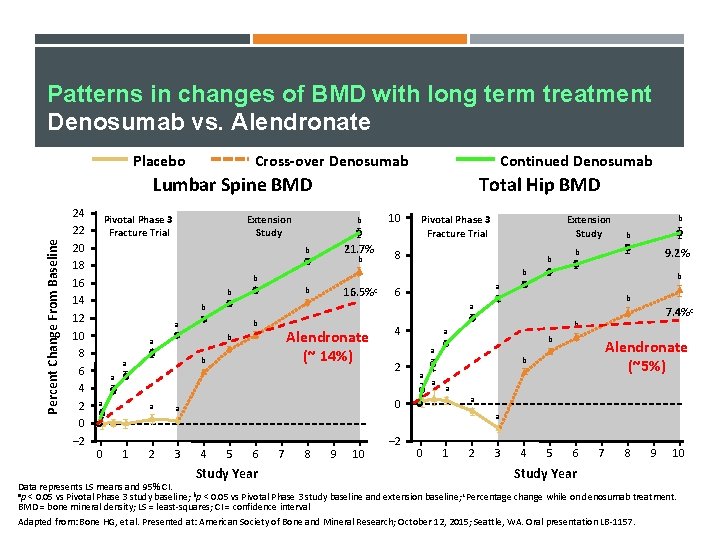
Patterns in changes of BMD with long term treatment Denosumab vs. Alendronate Placebo Cross-over Denosumab Continued Denosumab Percent Change From Baseline Lumbar Spine BMD 24 22 20 18 16 14 12 10 8 6 4 2 0 – 2 Pivotal Phase 3 Fracture Trial Total Hip BMD Extension Study b 21. 7% b b 10 Pivotal Phase 3 Fracture Trial 8 b 16. 5%c b b b Alendronate (~ 14%) b a a 0 1 a a 2 3 9. 2% b b 6 b a b a b b b a Extension Study 4 7. 4%c b a 2 a a Alendronate (~5%) b a a 0 a 4 5 6 Study Year 7 8 9 10 – 2 0 1 2 3 4 5 6 Study Year 7 8 9 10 Data represents LS means and 95% CI. ap < 0. 05 vs Pivotal Phase 3 study baseline; bp < 0. 05 vs Pivotal Phase 3 study baseline and extension baseline; c. Percentage change while on denosumab treatment. BMD = bone mineral density; LS = least-squares; CI = confidence interval Adapted from: Bone HG, et al. Presented at: American Society of Bone and Mineral Research; October 12, 2015; Seattle, WA. Oral presentation LB-1157.
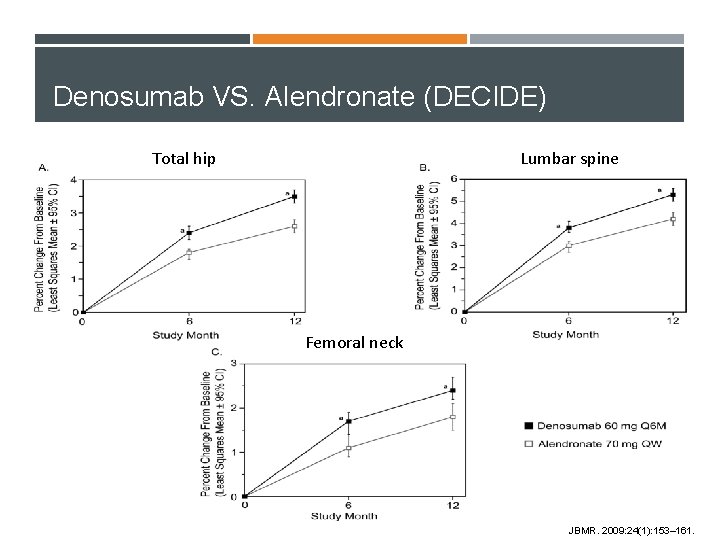
Denosumab VS. Alendronate (DECIDE) Total hip Lumbar spine Femoral neck JBMR. 2009: 24(1): 153– 161.
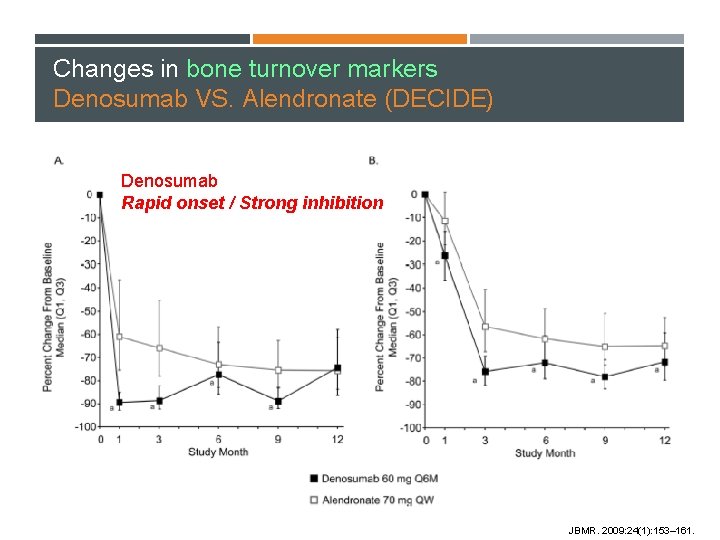
Changes in bone turnover markers Denosumab VS. Alendronate (DECIDE) Denosumab Rapid onset / Strong inhibition JBMR. 2009: 24(1): 153– 161.
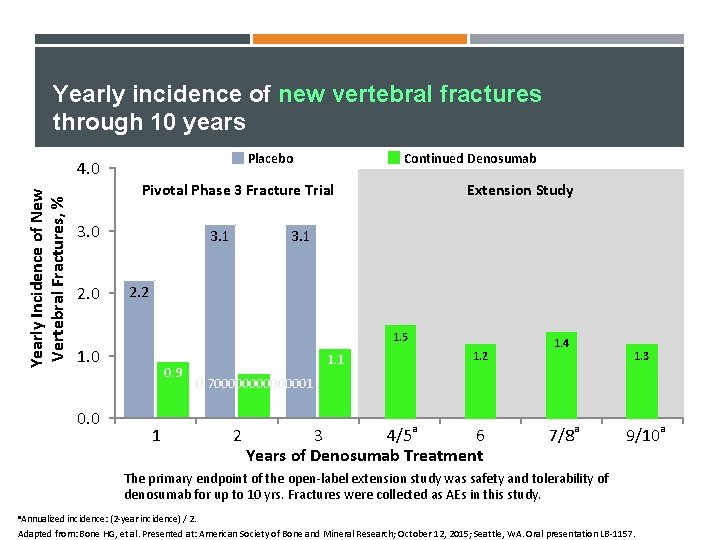
Yearly incidence of new vertebral fractures through 10 years Placebo Yearly Incidence of New Vertebral Fractures, % 4. 0 Continued Denosumab Pivotal Phase 3 Fracture Trial 3. 0 2. 0 3. 1 Extension Study 3. 1 2. 2 1. 5 1. 0 0. 9 1. 2 1. 1 1. 4 1. 3 0. 700000001 1 2 a 3 4/5 6 Years of Denosumab Treatment a 7/8 a 9/10 The primary endpoint of the open-label extension study was safety and tolerability of denosumab for up to 10 yrs. Fractures were collected as AEs in this study. a. Annualized incidence: (2 -year incidence) / 2. Adapted from: Bone HG, et al. Presented at: American Society of Bone and Mineral Research; October 12, 2015; Seattle, WA. Oral presentation LB-1157.
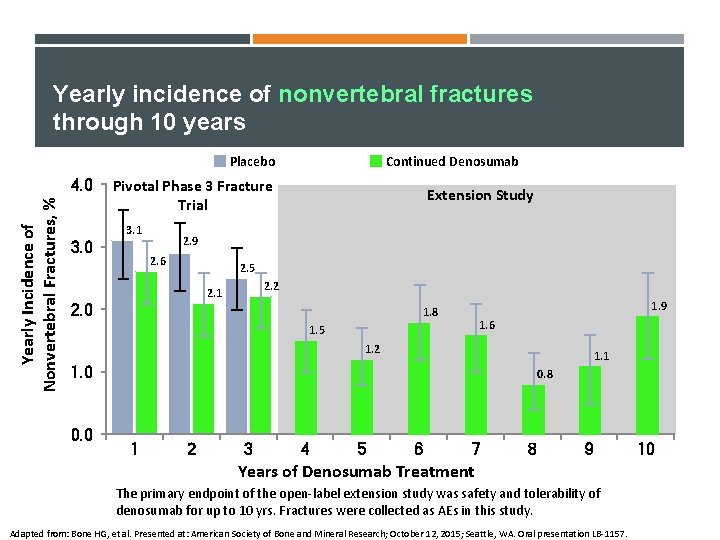
Yearly incidence of nonvertebral fractures through 10 years Placebo Yearly Incidence of Nonvertebral Fractures, % 4. 0 Continued Denosumab Pivotal Phase 3 Fracture Trial 3. 1 3. 0 Extension Study 2. 9 2. 6 2. 5 2. 2 2. 1 2. 0 1. 9 1. 8 1. 6 1. 5 1. 2 1. 1 1. 0 0. 8 1 2 3 4 5 6 7 8 9 Years of Denosumab Treatment The primary endpoint of the open-label extension study was safety and tolerability of denosumab for up to 10 yrs. Fractures were collected as AEs in this study. Adapted from: Bone HG, et al. Presented at: American Society of Bone and Mineral Research; October 12, 2015; Seattle, WA. Oral presentation LB-1157. 10
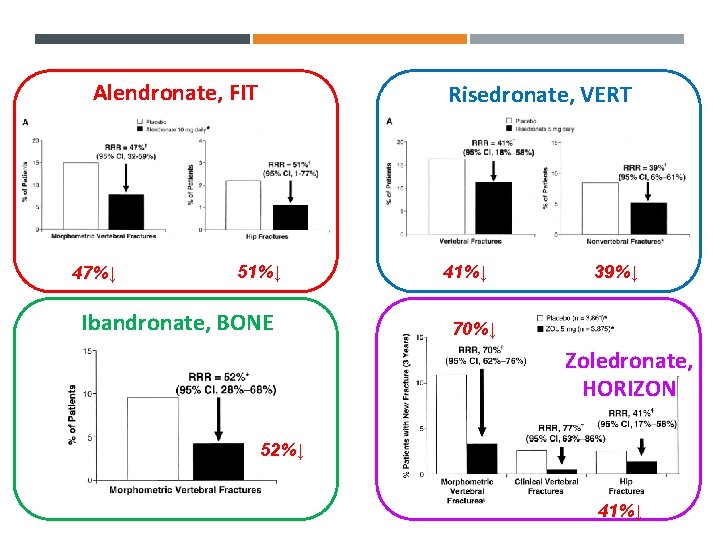
Alendronate, FIT 47%↓ Risedronate, VERT 51%↓ Ibandronate, BONE 41%↓ 39%↓ 70%↓ Zoledronate, HORIZON 52%↓ 41%↓
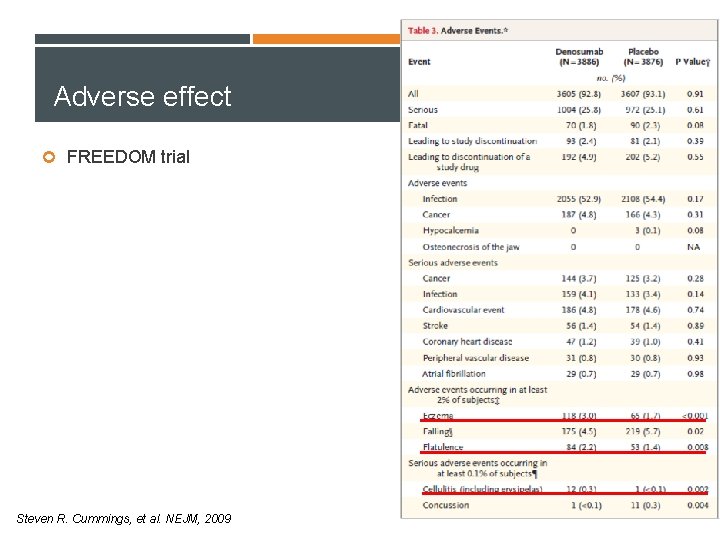
Adverse effect FREEDOM trial Steven R. Cummings, et al. NEJM, 2009 Cummings et al. , NEJM 2009
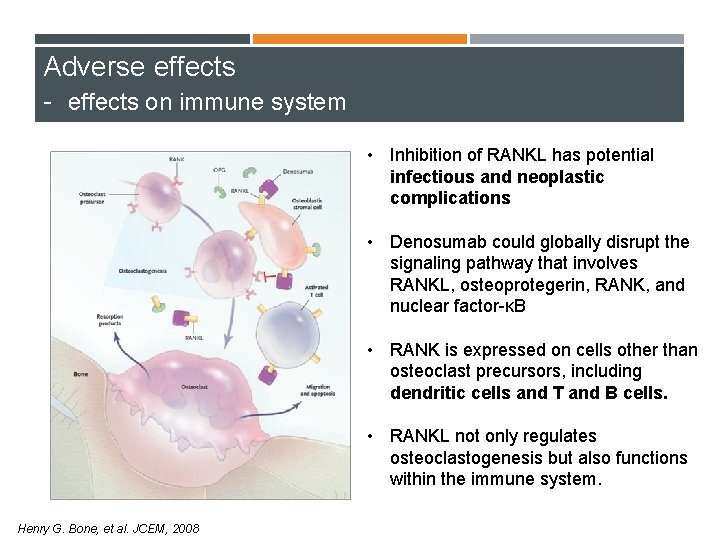
Adverse effects - effects on immune system • Inhibition of RANKL has potential infectious and neoplastic complications • Denosumab could globally disrupt the signaling pathway that involves RANKL, osteoprotegerin, RANK, and nuclear factor-κB • RANK is expressed on cells other than osteoclast precursors, including dendritic cells and T and B cells. • RANKL not only regulates osteoclastogenesis but also functions within the immune system. Henry G. Bone, et al. JCEM, 2008
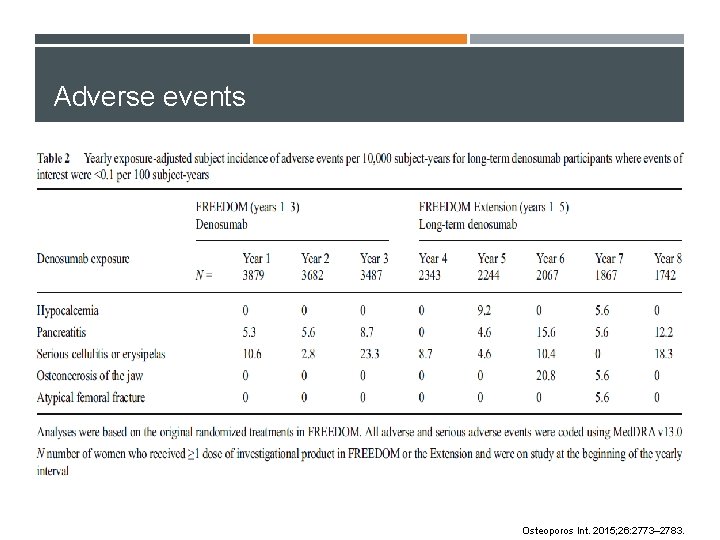
Adverse events Osteoporos Int. 2015; 26: 2773– 2783.

Adverse events Hypocalcemia Denosumab should not be given to patients with preexisting hypocalcemia until it is corrected. Predisposing to hypocalcemia (CKD, CCr <30 m. L/min) should be monitored for hypocalcemia All women were supplemented with daily calcium (1000 mg) and vitamin D (400 to 800 units) Steven R. Cummings, et al. NEJM, 2009

Adverse events Osteonecrosis of the jaw Atypical femur fractures FREEDOM extension trial 5 cases of ONJ, 1 case of AFF (after 8 years) 3 cases of ONJ, 1 case of AFF (after 5 year cross over) Henry G. Bone, et al. JCEM, 2008

Long-term treatment with anti-resorptive agents Bisphonates Denosumab Bisphonate Denosumab or Bisphonates 870 postmenopausal women aged ≥ 55 years Who transitioned from daily or weekly alendronate treatment and were considered to be suboptimally adherent to therapy Bone 58 (2014) 48– 54
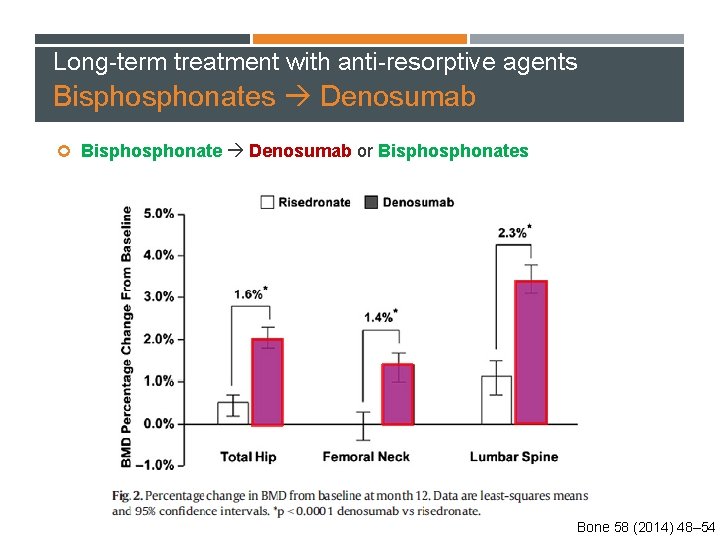
Long-term treatment with anti-resorptive agents Bisphonates Denosumab Bisphonate Denosumab or Bisphonates Bone 58 (2014) 48– 54
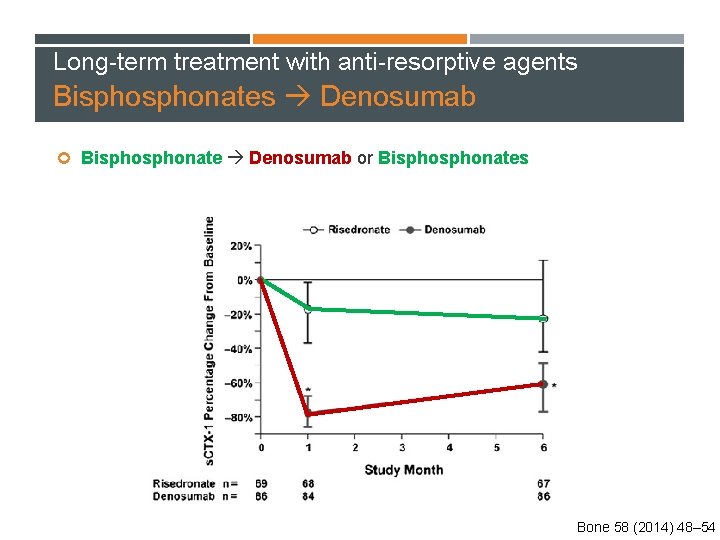
Long-term treatment with anti-resorptive agents Bisphonates Denosumab Bisphonate Denosumab or Bisphonates Bone 58 (2014) 48– 54
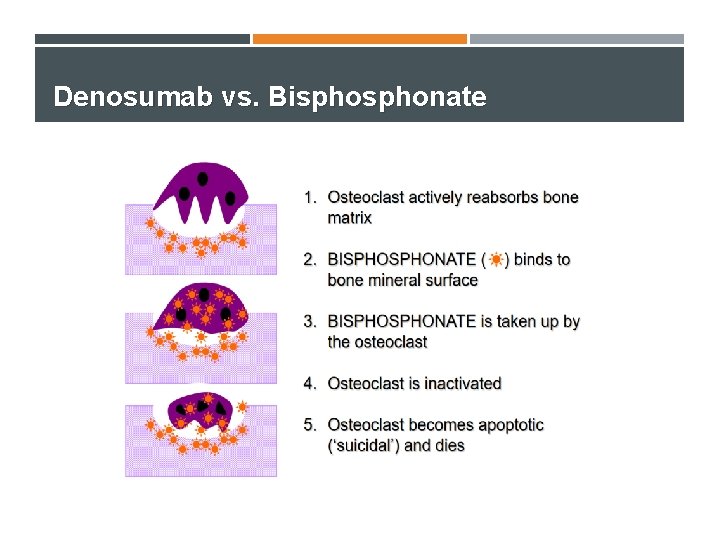
Denosumab vs. Bisphonate
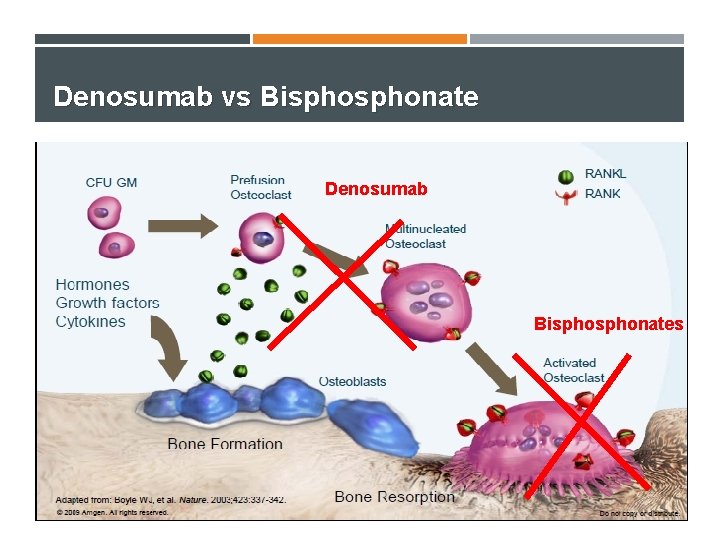
Denosumab vs Bisphonate Denosumab Bisphonates
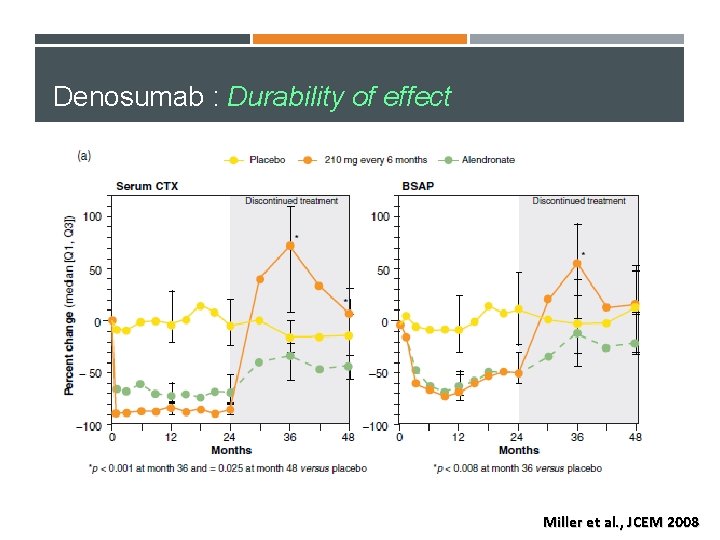
Denosumab : Durability of effect Miller et al. , JCEM 2008
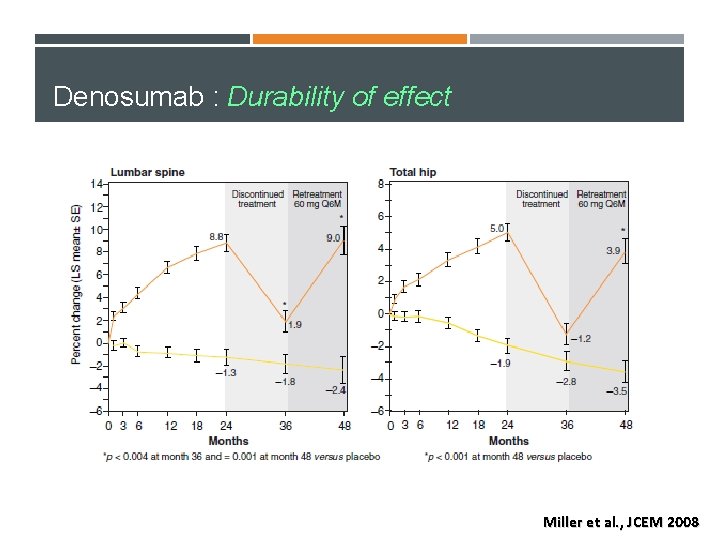
Denosumab : Durability of effect Miller et al. , JCEM 2008
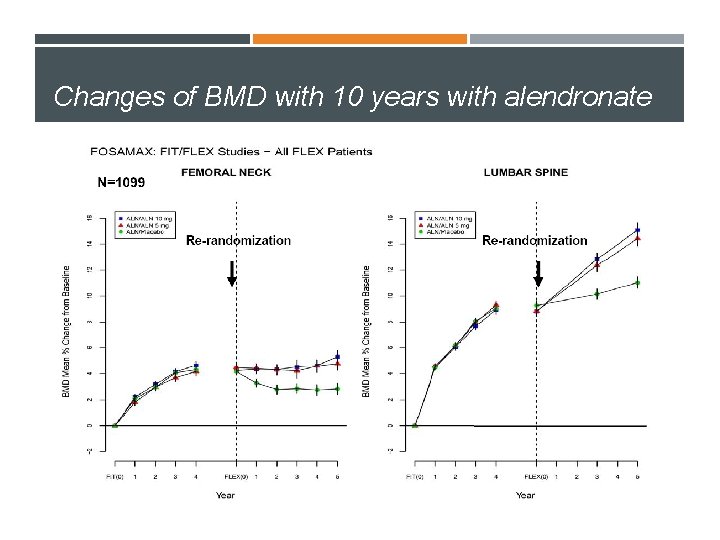
Changes of BMD with 10 years with alendronate
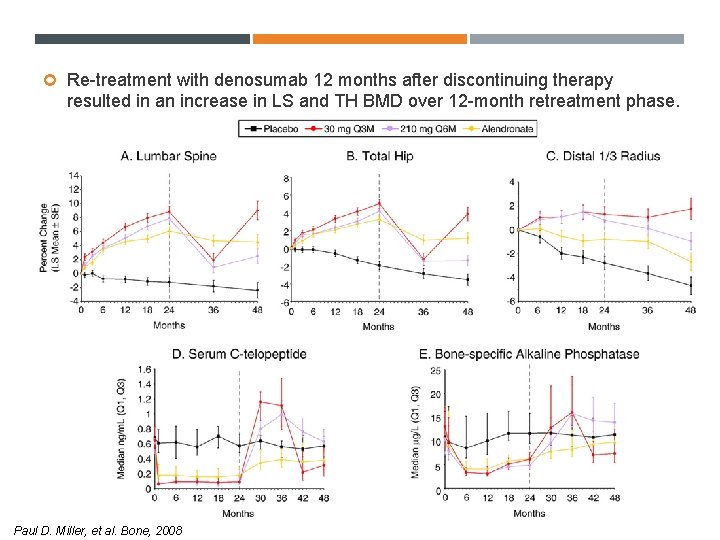
Re-treatment with denosumab 12 months after discontinuing therapy resulted in an increase in LS and TH BMD over 12 -month retreatment phase. Paul D. Miller, et al. Bone, 2008
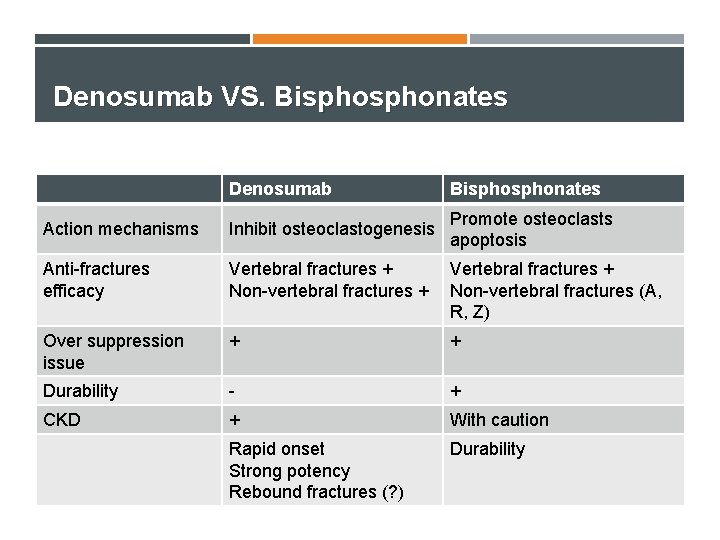
Denosumab VS. Bisphonates Denosumab Bisphonates Action mechanisms Inhibit osteoclastogenesis Promote osteoclasts apoptosis Anti-fractures efficacy Vertebral fractures + Non-vertebral fractures + Vertebral fractures + Non-vertebral fractures (A, R, Z) Over suppression issue + + Durability - + CKD + With caution Rapid onset Strong potency Rebound fractures (? ) Durability
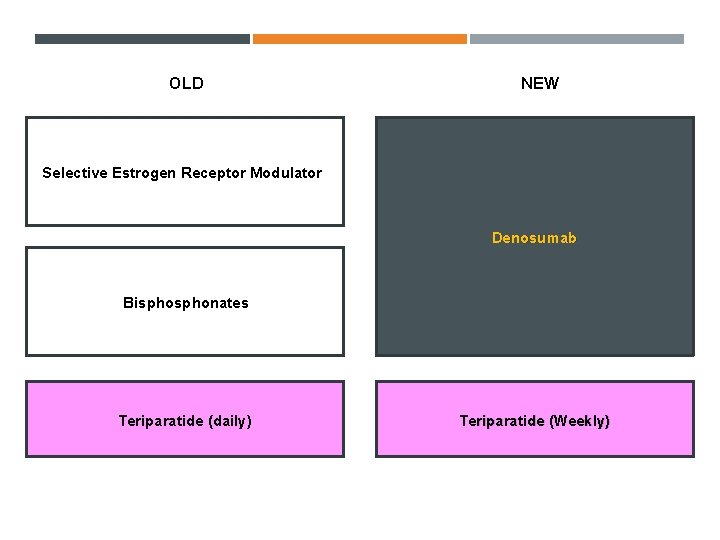
OLD NEW Selective Estrogen Receptor Modulator Denosumab Bisphonates Teriparatide (daily) Teriparatide (Weekly)

Teriparatide

Teriparatide Recombinant human PTH(1 -34) Available from 2002’ in USA, 2003’ in Europe
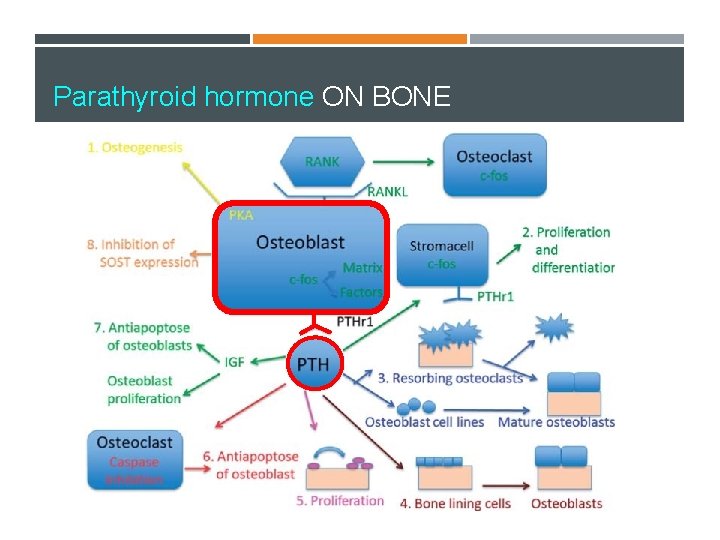
Parathyroid hormone ON BONE
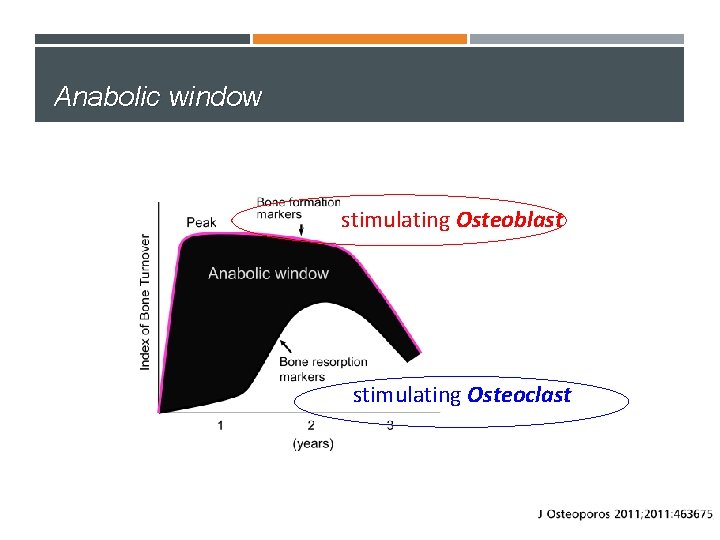
Anabolic window stimulating Osteoblast stimulating Osteoclast
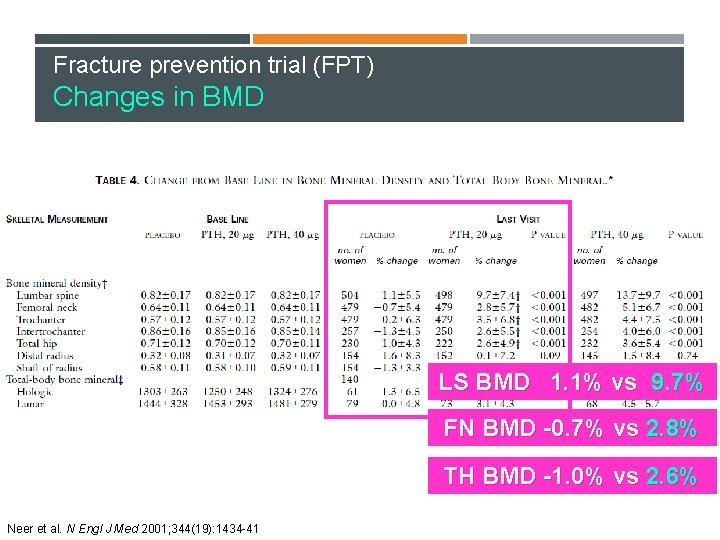
Fracture prevention trial (FPT) Changes in BMD LS BMD 1. 1% vs 9. 7% FN BMD -0. 7% vs 2. 8% TH BMD -1. 0% vs 2. 6% Neer et al. N Engl J Med 2001; 344(19): 1434 -41
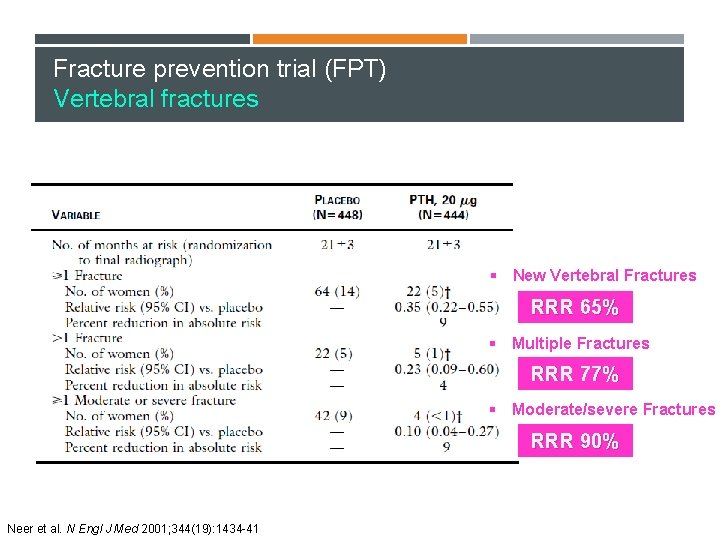
Fracture prevention trial (FPT) Vertebral fractures § New Vertebral Fractures RRR 65% § Multiple Fractures RRR 77% § Moderate/severe Fractures RRR 90% Neer et al. N Engl J Med 2001; 344(19): 1434 -41
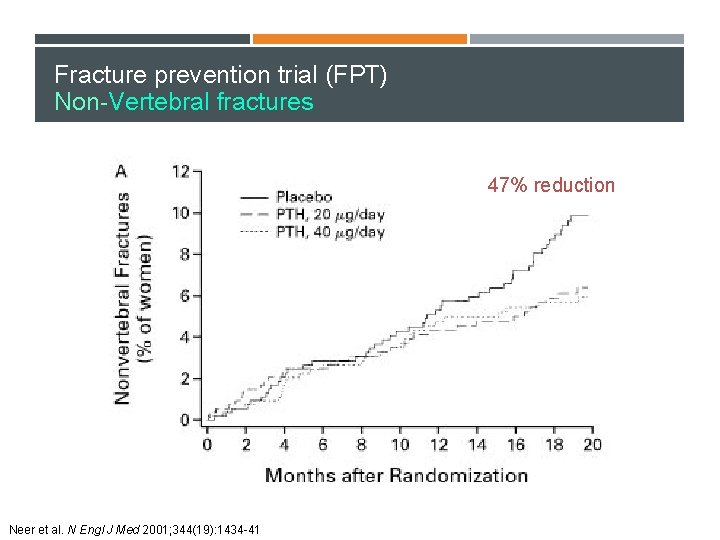
Fracture prevention trial (FPT) Non-Vertebral fractures 47% reduction Neer et al. N Engl J Med 2001; 344(19): 1434 -41

Teriparatide Daily : 20 mcg per day (Self –injection) Weekly : 56 mcg per week (Hospital-based)

Teriparatide Daily : 20 mcg per day (Self –injection) FPT study Weekly : 56. 5 mcg per week (Hospital-based) TOWER study
![Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial Lumbar Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial Lumbar](http://slidetodoc.com/presentation_image/1f933c3ecea3d7c458ad7173a467ac02/image-47.jpg)
Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial Lumbar Spine BMD Total Hip BMD 6. 4% 3. 0% [Nakamura T et al. , J Clin Endocrinol Metab 97: 3097, 2012]
![Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial [Nakamura Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial [Nakamura](http://slidetodoc.com/presentation_image/1f933c3ecea3d7c458ad7173a467ac02/image-48.jpg)
Once-weekly Teriparatide [human parathyroid hormone (PTH) 1 -34] Once-Weekly Efficacy Research (TOWER) trial [Nakamura T et al. , J Clin Endocrinol Metab 97: 3097, 2012]
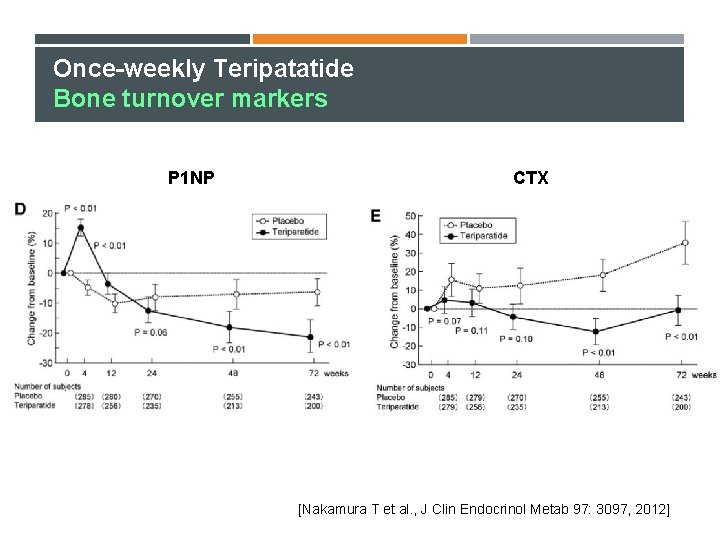
Once-weekly Teripatatide Bone turnover markers P 1 NP CTX [Nakamura T et al. , J Clin Endocrinol Metab 97: 3097, 2012]
![BTM changes with once-daily teriparatide [M. Tsujimoto et al. Bone 48 (2011) 798] BTM changes with once-daily teriparatide [M. Tsujimoto et al. Bone 48 (2011) 798]](http://slidetodoc.com/presentation_image/1f933c3ecea3d7c458ad7173a467ac02/image-50.jpg)
BTM changes with once-daily teriparatide [M. Tsujimoto et al. Bone 48 (2011) 798]
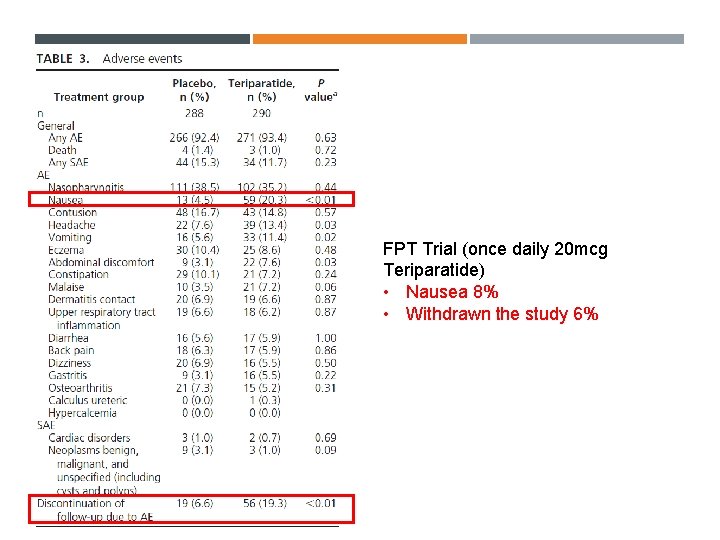
FPT Trial (once daily 20 mcg Teriparatide) • Nausea 8% • Withdrawn the study 6%

Conclusion OLD NEW Selective Estrogen Receptor Modulator Denosumab Individualized treatment Goal-directed therapy Bisphonates Teriparatide (daily) Teriparatide (Weekly)
- Slides: 52