BAHRIA COLLEGE ISLAMABAD GENERAL SCIENCE new lower secondary

BAHRIA COLLEGE ISLAMABAD GENERAL SCIENCE (new lower secondary science II) CLASS VIII MISS IQRA AKRAM

FIRST TERM SYLLABUS • • • Unit No. 1 ATOM AND MOLECULES PG NO 03 Unit No. 2 CHANGES IN MATTER PG NO 06 Unit No. 5 CRYSTALS PG NO 09 Unit No. 10 TRANSPORT IN LIVING ORGANISM PG NO 12 Unit No. 13 LIGHT PG NO 15

Define crystals. Pg. 44 • Crystals are solid with fixed, regular pattern • . A crystal is a solid composed of atoms, ions, or molecules arranged in a pattern that is periodic in three dimensions. • Info about crystals: • although we cannot see the individual atom inside a crystal, but we have an idea of their arrangement by looking at the crystals. • E. g. sodium chloride = common salt =
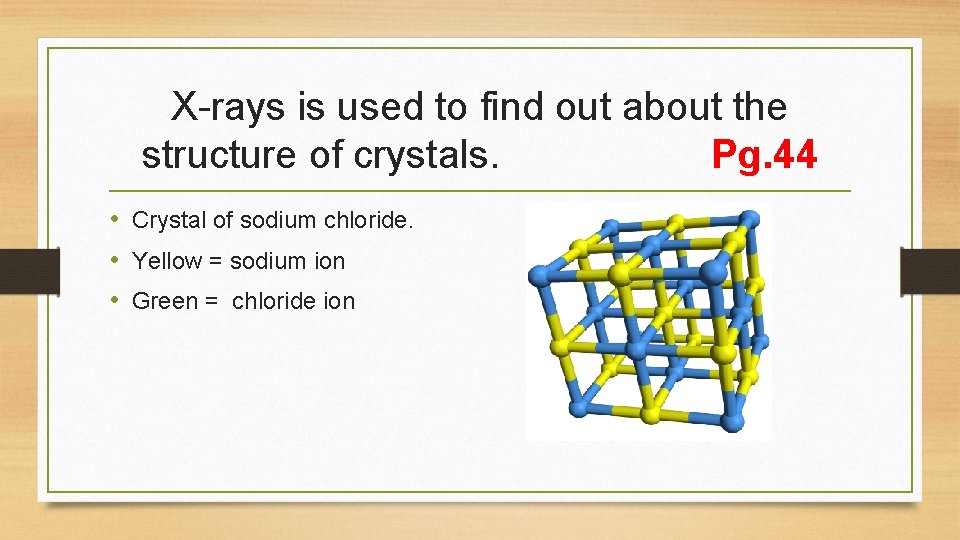
X-rays is used to find out about the structure of crystals. Pg. 44 • Crystal of sodium chloride. • Yellow = sodium ion • Green = chloride ion

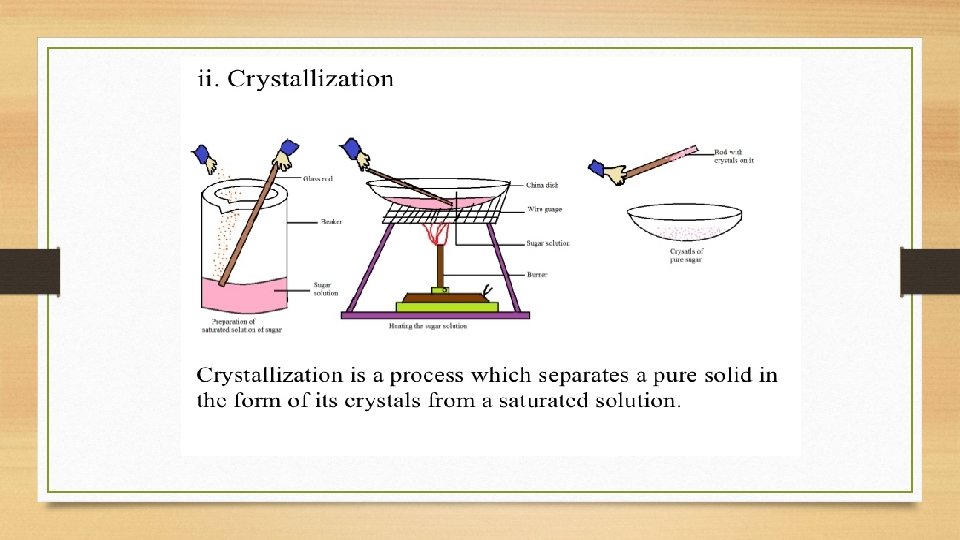
Define crystallization. • Crystallization or crystallisation is the (natural or artificial) process by which a solid forms, where the atoms or molecules are highly organized into a structure known as a crystal. • Crystallization ia a process of forming crystals.

Formation of crystals • • • Crystals are formed when: The vapour of subliming substance cool. A molten substances solidifying on cooling. A hot saturated solution is cooled. A saturated solution evaporates,

Explain method of formation of crystals by different method. • 1. The vapour of subliming substance cool: when the vapour of substance that sublimes cool, it will appear as solid crystals. For example : iodine crystals= heated =they vaporize. When vaporize iodine contact will cooler part of evaporating dish= iodine crystals.

2. A molten substance solidifies on cooling. • When a hot molten substance cools and solidifies, it turns into crystals. • Molten substance which solidify slowly produce bigger crystals. • For example: small crystals of minerals are formed by rapid cooling of molten lava.

3. A hot saturated solution is cooled: Saturated solution: A saturated solution is a chemical solution containing the maximum concentration of a solute dissolved in the solvent. The additional solute will not dissolve in a saturated solution. • Normally crystallization will only take place in solutions that have exceeded their saturation points. • A hot saturated solution cools , it will exceed its saturation point at a lower temperature. • The excess dissolve solute will then appear in the solution as a crystals.

3. A hot saturated solution is cooled • • For example: copper sulphate crystals 1. we can prepare hot saturated solution of copper sulphate. Pour some of it into an evaporating dish Allow the solution to quickly cool in to refrigerator and observe the crystals formed. • Allow the solution in another evaporation dish to cool slowly in laboratory • Compare the size and shape of crystals. • When cooling is slow, big crystals are formed.





Application of crystallization • During crystallization pure substance crystallize while other impurities remain in their original states. • Crystallization is widely used in manufacturing processes, especially those involved in producing pure substances. • E. g. production of silicon's wafers • Refining sugar

Production of silicon wafers • Seed crystals of silicon are impressed in molten silicon to obtain large silicon's crystal. • The large silicon crystals are then sliced into thin sheets called wafers. • Silicon wafers are used to make solar shell and computer microchips,

Refining Sugar • The sugar that is commonly used in homes or in the food industries is refined sugar crystals. • In the extraction of sugar from sugar syrup obtained from sugar cane , • Seed crystal of sugar are immersed in the saturated sugar syrup to form large and pure sugar crystals.

Review Exercise pg. 48 • Qno 1: clay • Qno 2: Crystals are solid with fixed, regular pattern. • A crystal is a solid composed of atoms, ions, or molecules arranged in a pattern that is periodic in three dimensions. • Qno 3: what is crystallization ? write down two useful application of crystallization See slide no 5, 12, 13, 14

Qno 4: name some ways of obtaining crystal. Ans: Crystals are formed when: • • The vapour of subliming substance cool. A molten substances solidifying on cooling. A hot saturated solution is cooled. A saturated solution evaporates

• Thank you
- Slides: 21