BACTERIAL CULTURE METHODS PURPOSE OF CULTURING Isolation Properties

BACTERIAL CULTURE METHODS

PURPOSE OF CULTURING § Isolation § Properties of bacteria § To create antigens for laboratory use § Typing with Bacteriophages and Bacteriocins susceptibility § To test for Antibiotic sensitivity § Estimate viable counts § Maintain stock cultures

METHODS TO ISOLATE THE BACTERIA • Streak culture • Stroke • Stab • Lawn • Pour plate • Liquid culture • Special methods for anaerobic cultures

HOW TO INOCULATE A CULTURE PLATE § Plate: provide large surface for isolation and observation of colonies § Using a sterile loop or a sterile swab streak your sample on the Petri plate § Important let your sterilized loop cool before you pick up your sample

Streaking § Streaking: The specific technique for isolating bacterium on solid agar. § Streak Plate Method: A method used to obtain colonies from a single parent species.
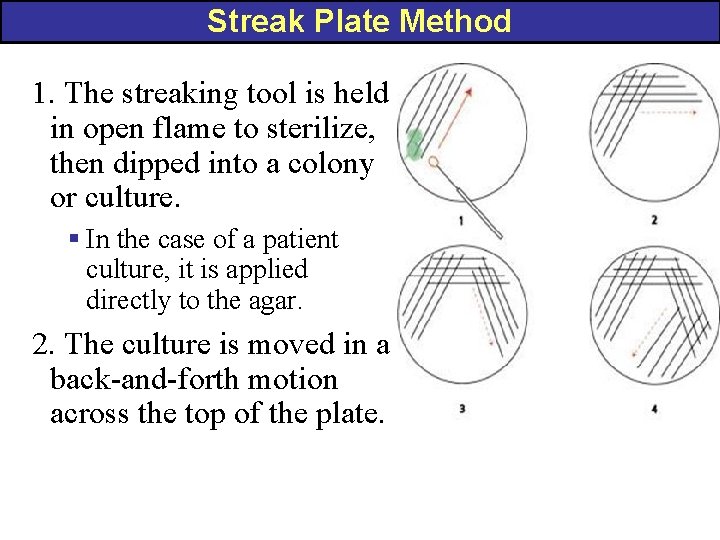
Streak Plate Method 1. The streaking tool is held in open flame to sterilize, then dipped into a colony or culture. § In the case of a patient culture, it is applied directly to the agar. 2. The culture is moved in a back-and-forth motion across the top of the plate.
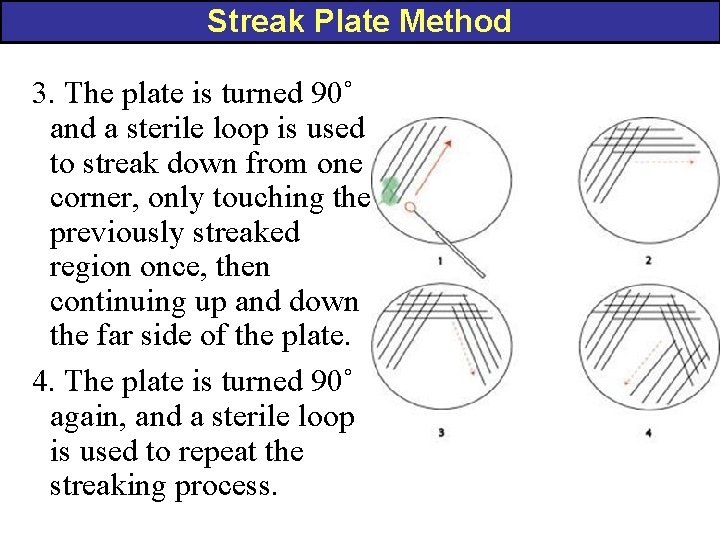
Streak Plate Method 3. The plate is turned 90˚ and a sterile loop is used to streak down from one corner, only touching the previously streaked region once, then continuing up and down the far side of the plate. 4. The plate is turned 90˚ again, and a sterile loop is used to repeat the streaking process.

STROKE CULTURE § Stroke culture is made in tubes containing agar slope / slant. § Uses § Provide a pure growth of bacterium for slide agglutination and other diagnostic tests.

STAB CULTURE § Puncturing suitable medium such as nutrient agar, gelatin, § Observe gelatin liquefaction § Preserving the stock culture.

Gelatin liquefaction Oxidation – Fermentation medium

LAWN CULTURE § Lawn or carpet culture to create uniform surface of organisms § Bacteriophages typing § To obtain large amount of antigens § To perform antibiotic sensitivity testing
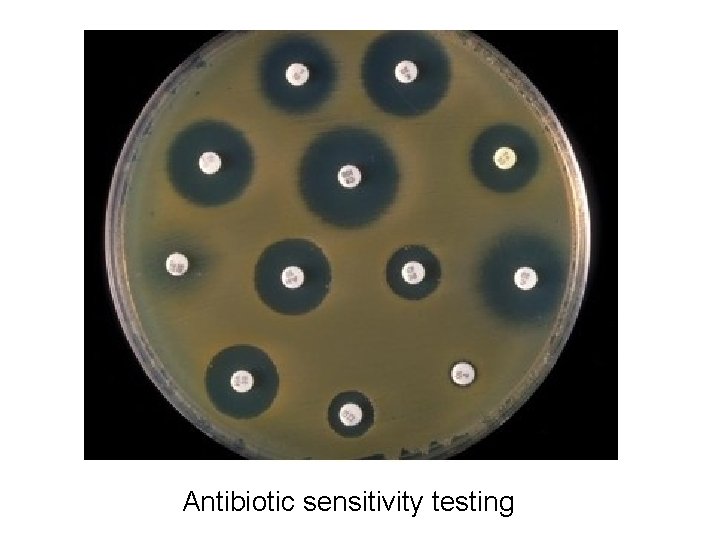
Antibiotic sensitivity testing

POUR PLATE CULTURE § Agar medium is melted (15 ml) and cooled to 45 o. C. § 1 ml of the inoculum is added to the molten agar. § Mix well and pour to a sterile petri dish. § Allow it to set. § Incubate at 37 o. C, colonies will be distributed throughout the depth of the medium. § Uses § Gives an estimate of the viable bacterial count in a suspension. § For the quantitative urine cultures.

LIQUID CULTURES § Test tubes, screw capped bottles, or flasks containing liquid media § Inoculated by pipettes, syringes or loop. § Uses: Blood culture, sterility tests; where number of bacteria is less § Disadvantage: does not provide pure culture § Identification of bacteria not possible

ANAERBIC CULTURE METHODS § Methods to produce anaerobiosis 1. Production of vacuum 2. Displacement of oxygen 3. By displacement and combustion of oxygen 4. Absorption of oxygen by chemical and biological methods 5. By reducing agents 6. Anaerobic chamber

1. Production of vacuum DESICCATOR § In Desiccator some oxygen is left § Not suitable for fluid culture § Displacement of oxygen is done with ü Hydrogen ü Nitrogen ü Helium ü Co 2

2. Displacement of oxygen CANDLE JAR § Inoculated plates are kept § Burning candle use up all oxygen § But a little O 2 is left § But presence of CO 2 stimulates the most bacterium

Mc. Intosh – Fildes’ anaerobic jar § Consists of a metal jar or glass jar with a metal lid which can be clamped air tight. § The lid has 2 tubes – gas inlet and gas outlet § The lid has two terminals – connected to electrical supply. § Under the lid – small grooved porcelain spool, wrapped with a layer of palladinised asbestos acts as catalyst.

Working: § Inoculated plates are placed inside the jar and the lid clamped air tight. § The outlet tube is connected to a vacuum pump and the air inside is evacuated. § The outlet tap is then closed and the inlet tube is connected to a hydrogen supply. § After the jar is filled with hydrogen, the electric terminals are connected to a current supply, so that the palladinised asbestos is heated. § Act as a catalyst for the combination of hydrogen with residual oxygen.

Biological method § Absorption of oxygen by incubation with aerobic bacteria, germinating seeds or chopped vegetables. Reduction of oxygen § By using reducing agents – 1% glucose, 0. 1% Thioglycolate

4. Absorption of oxygen by chemical methods 1. Pyrogallol: § A large tube containing solution of sodium hydroxide and pyrogallic acid in air tight chamber. Alkaline pyrogallol absorbs oxygen to produce anaerobiosis. 2. Chromium and Sulphuric acid: § Chromium + H 2 SO 4+O 2= Chromium sulphate 3. Gas- pak:
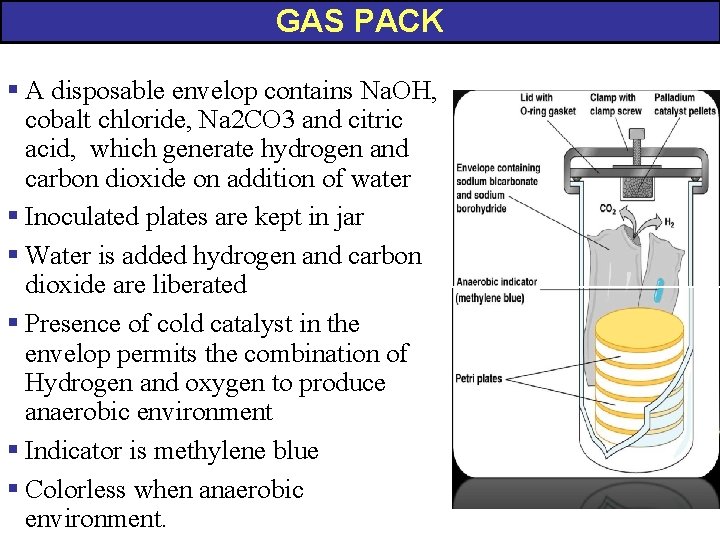
GAS PACK § A disposable envelop contains Na. OH, cobalt chloride, Na 2 CO 3 and citric acid, which generate hydrogen and carbon dioxide on addition of water § Inoculated plates are kept in jar § Water is added hydrogen and carbon dioxide are liberated § Presence of cold catalyst in the envelop permits the combination of Hydrogen and oxygen to produce anaerobic environment § Indicator is methylene blue § Colorless when anaerobic environment.

4. Absorption of oxygen by biological methods § Absorption of oxygen by incubation with aerobic bacteria, germinating seeds or chopped vegetables.

5. By reducing agents Reducing agents § O. 1% Thiglyclolate § 0. 1% Ascorbic acid § 0. 05 % cysteine

§ Robertson cooked meat medium routinely used in anaerobic spore bearing bacteria

6. Anaerobic chamber § Anaerobic inoculating and incubation system. § Fitted with air tight rubber gloves to insert hands for working.

Methods for isolation of Pure cultures § Surface plating: § Use of enriched and selective media: § Pretreatment of specimens. § By heating liquid medium.
- Slides: 28