Bacteria and the cytoskeleton The human body is
Bacteria and the cytoskeleton
The human body is a dangerous place for a bacteria to be! Antibodies Neutrophils Complement Innate response - lysozymes

Many bacteria find it much more comfortable inside the cells of its host.

Some bacteria gain entry to cells by forcing them to phagocytose them. Bacteria secrete Once engulfed Factors that stimulate Bacteria digest the Macrophagocytosis phagolysosome Now cells can grow Within the cell

Fibroblast protrusion, Louise Cramer University College London

GFP-actin. Stimulated Macropinocytosis

Killing phagocytosis tight compartment Stimulated phagocytosis loose compartment
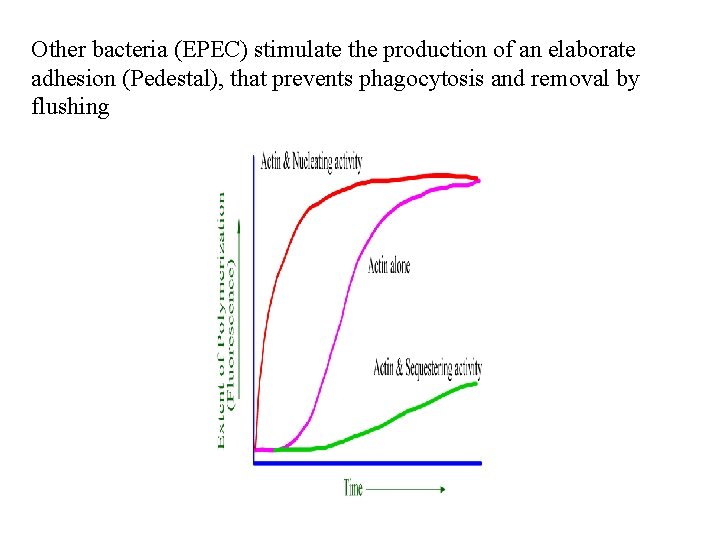
Other bacteria (EPEC) stimulate the production of an elaborate adhesion (Pedestal), that prevents phagocytosis and removal by flushing

Many bacteria subvert normal cytoskeletal function in order to parasitize their eukaryotic host through either adhesive complexes or inducing macro-pinocytosis Salmonella Escherichia Yersinia Shigella Staphylococcus (Most nasty bacteria are named After people!!) Legionella Listeria

Yersinia pestis was responsible for the Great Plagues. During the 6 th century AD, the plague ravaged the known world over a 50 year period causing 100 million deaths. The "black death" again devastated Europe during the 14 th century over a 5 year period causing 25 million deaths (25% of the European population). The bacterium was named after Yersin who identified it as being the causative agent of an outbreak of plague in Hong Kong
Xenopsylla cheopis Dirty Rat Homo sapiens Dirty Rattus norvegicus

WHO reports 1, 000 to 3, 000 cases of plague every year!

Other Yersinia cause disease. Yersinia enterocolitica Typically, only a small number of human cases of Yersiniosis are recognized. Symptoms are like that of appendicitis and out breaks are often detected by a sudden increase in appendectomies in a particular region. The Center for Disease Control & Prevention estimates that about 17, 000 cases occur each year in the United States.

Bacteria inject toxins into cells to subvert their activities The hypodermic syringes that they use are modified flagella
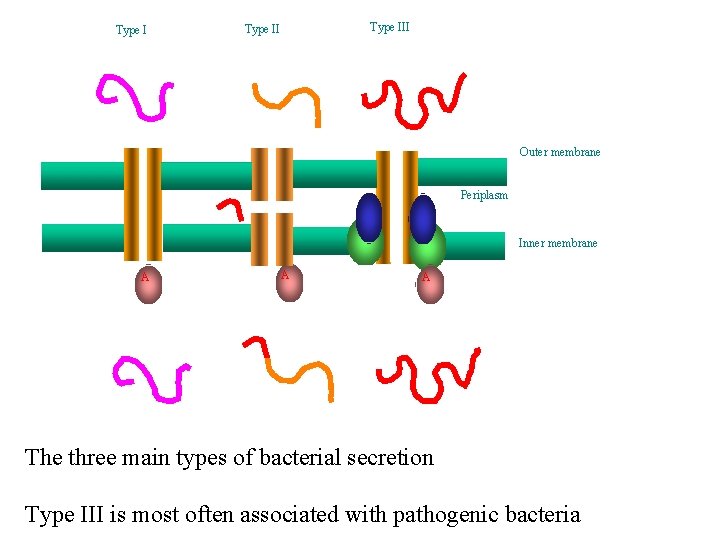
Type II Outer membrane Periplasm Inner membrane A A A The three main types of bacterial secretion Type III is most often associated with pathogenic bacteria

The most common pathogenic E. coli Abbrev. ETEC EIEC EPEC EHEC Full name Enterotoxin E. coli Common name and features inocolum Source Faecal Montizuma’s revenge, traveller’s tummy 108 usually comparatively mild, (Diacalm grade) contamination Enteroinvasive Invades, Shigella pathogenicity island high Food & waterborne 8 - 10 Enteropathogenic Pedestal formation, infant diarrhoea 10 Nosocomial community Enterohaemorrhagic (O 157)“Hamburger disease” Shiga toxin 3 Cattle faeces, meat
Interactions of the common pathogenic E. coli with epithelial cells
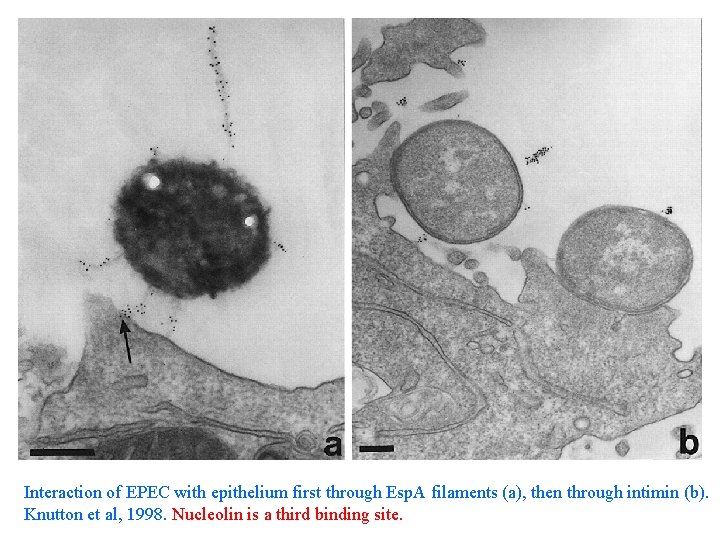
Interaction of EPEC with epithelium first through Esp. A filaments (a), then through intimin (b). Knutton et al, 1998. Nucleolin is a third binding site.

Scanning E. M. of EPEC and epithelium. Esp. A filaments appear to insert into cell (arrows in A), possibly to deliver Esp. B. Esp. A may be part of the Type III secretion pathway, it is needed for Esp. B delivery. Note the pedestals are all of equal length.

Some pathogenic E. coli (EPEC, EHEC) put down their own “Welcome mat” Tir (translocated intimin receptor) is injected into host by Type III secretion Tir binds to host a-actinin, talin and vinculin all components of the focal contact. Nucleolin is a bacterial binding site for EPEC.
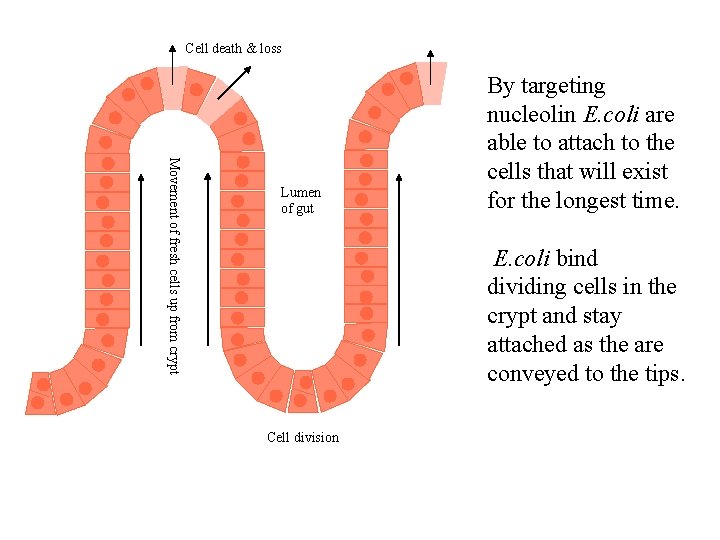
Cell death & loss Movement of fresh cells up from crypt Lumen of gut By targeting nucleolin E. coli are able to attach to the cells that will exist for the longest time. E. coli bind dividing cells in the crypt and stay attached as the are conveyed to the tips. Cell division
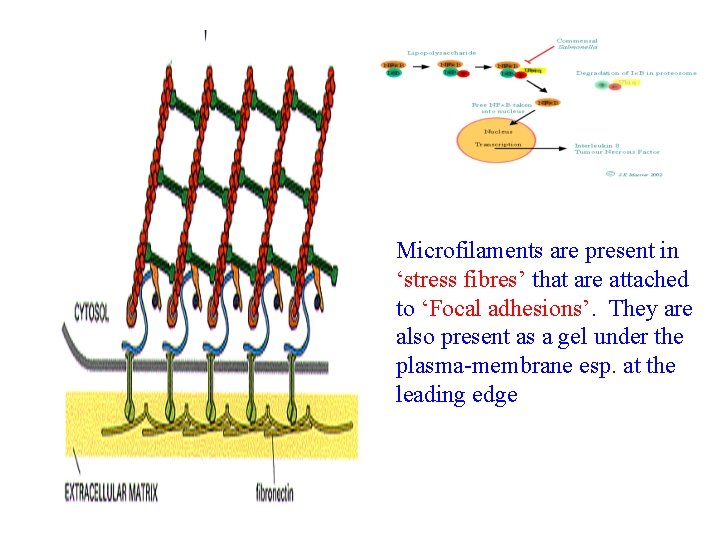
Microfilaments are present in ‘stress fibres’ that are attached to ‘Focal adhesions’. They are also present as a gel under the plasma-membrane esp. at the leading edge
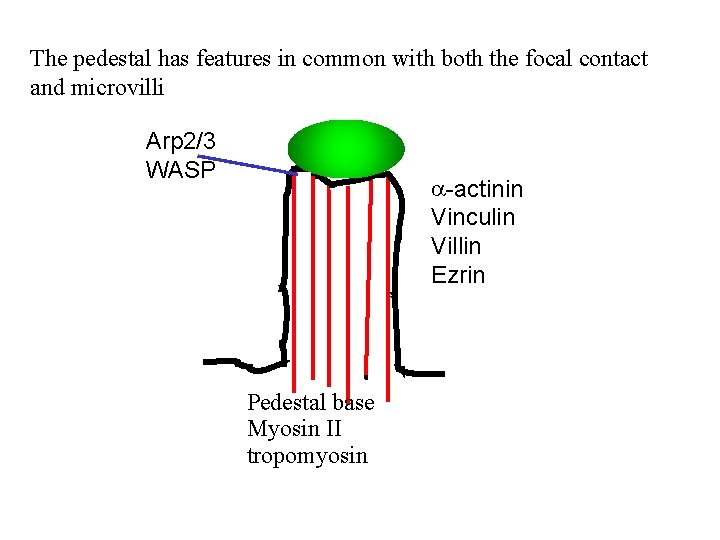
The pedestal has features in common with both the focal contact and microvilli Arp 2/3 WASP a-actinin Vinculin Villin Ezrin Pedestal base Myosin II tropomyosin

Pyrene-actin method to measure polymerisation kinetics O II NH-C-CH 2 I + H-S-H 2 C-Actin NH-C-CH 2 -S-H 2 C-Actin Pyrene Excited at 366 nm Light emitted at 384 nm measured Pyrene-actin in quartz cuvette
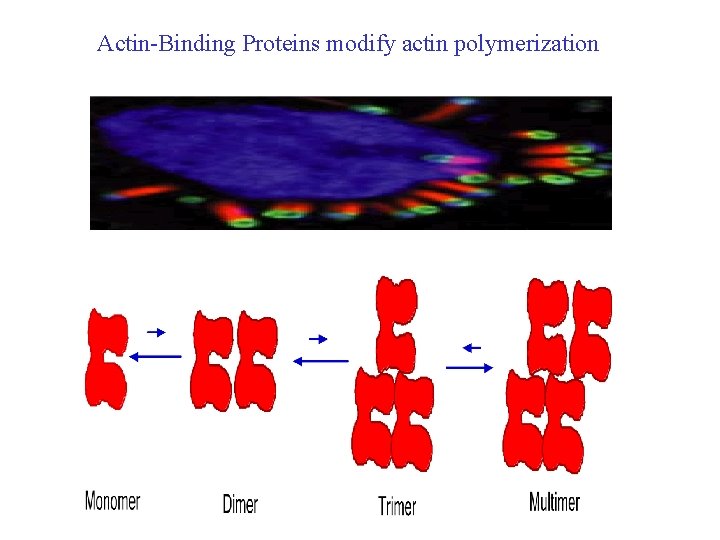
Actin-Binding Proteins modify actin polymerization
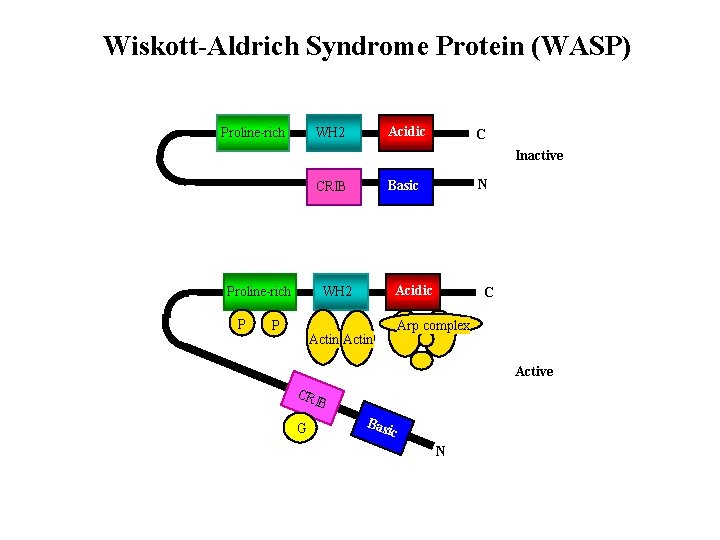
Wiskott-Aldrich Syndrome Protein (WASP) Proline-rich Acidic WH 2 C Inactive Proline-rich P Acidic WH 2 P N Basic CRIB Actin C Arp complex Active CRI B G Basi c N

The Arp 2/3 complex An actin-binding group of proteins pivotally involved in the regulation of actin polymerisation. Zero length crosslink Arp 3 Arp 2 Non-zero length crosslink Yeast two hybrid screen p 18 p 19 p 14 p 35 p 40 Microfilament binding

Analysis of the WASP domains required for Pedestal formation WASP-WT WASP-DC WASP-DGBD Kalman et al, 1999 Nature Cell Biol. 1; 389 -391.
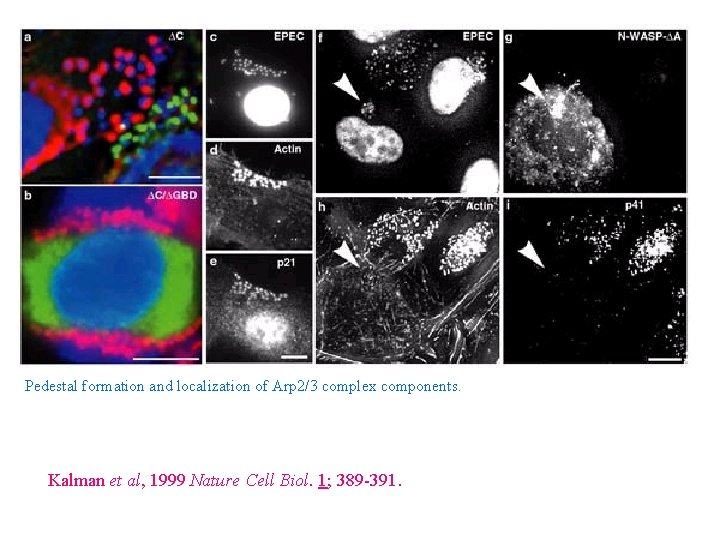
Pedestal formation and localization of Arp 2/3 complex components. Kalman et al, 1999 Nature Cell Biol. 1; 389 -391.

Pedestal formation by EPEC
Salmonella Edwina Currie Eggs Salmonella

Commensal Salmonella calm the Immune system
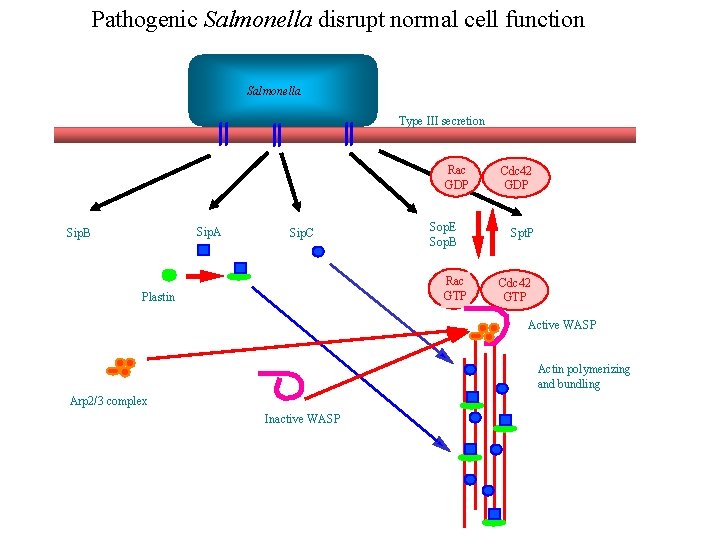
Pathogenic Salmonella disrupt normal cell function Salmonella Type III secretion Rac GDP Sip. A Sip. B Sip. C Sop. E Sop. B Rac GTP Plastin Cdc 42 GDP Spt. P Cdc 42 GTP Active WASP Actin polymerizing and bundling Arp 2/3 complex Inactive WASP
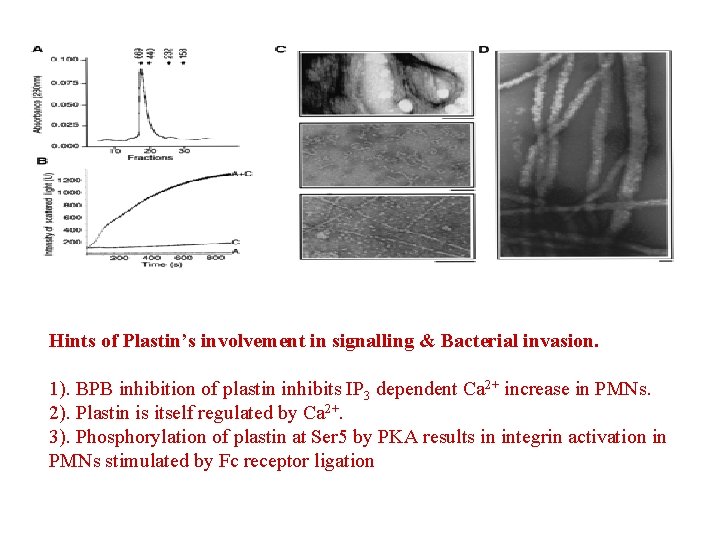
Hints of Plastin’s involvement in signalling & Bacterial invasion. 1). BPB inhibition of plastin inhibits IP 3 dependent Ca 2+ increase in PMNs. 2). Plastin is itself regulated by Ca 2+. 3). Phosphorylation of plastin at Ser 5 by PKA results in integrin activation in PMNs stimulated by Fc receptor ligation
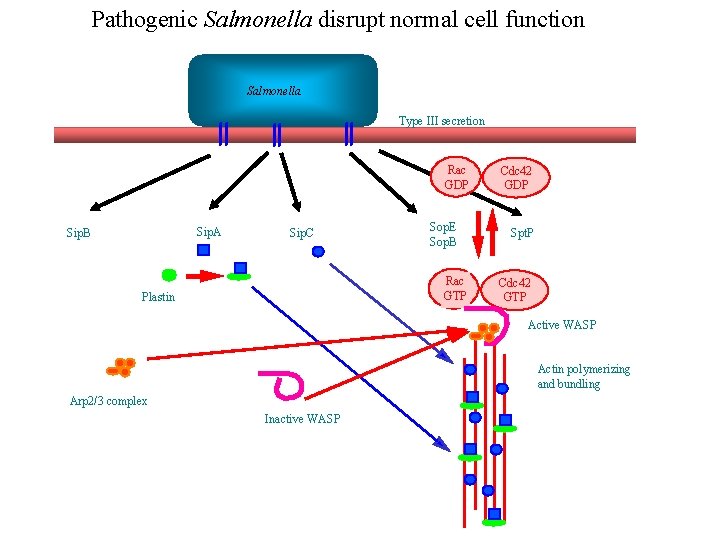
Pathogenic Salmonella disrupt normal cell function Salmonella Type III secretion Rac GDP Sip. A Sip. B Sip. C Sop. E Sop. B Rac GTP Plastin Cdc 42 GDP Spt. P Cdc 42 GTP Active WASP Actin polymerizing and bundling Arp 2/3 complex Inactive WASP

The Salmonella cycle of infection SPI 1 SPI 2 Initial contact Injection By type III secretion Actin polymerization and phagocytosis Phagosome stimulates new protein secretion Through a second type III machine A fresh actin wave of actin polymerization results in the vacuole being covered in actin. Lysosomes can’t fuse Nucleus Some time later an actin ADP-ribosylating enzyme disassembles the structure for unknown reasons (cell lysis & spread? ).
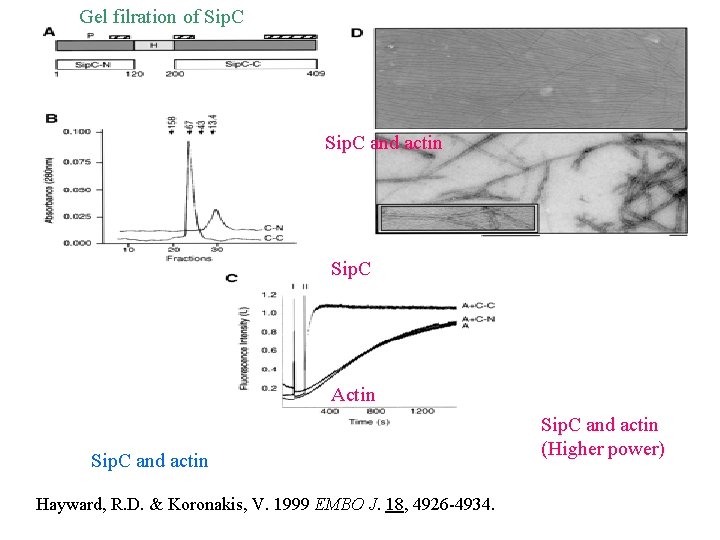
Gel filration of Sip. C and actin Sip. C Actin Sip. C and actin Hayward, R. D. & Koronakis, V. 1999 EMBO J. 18, 4926 -4934. Sip. C and actin (Higher power)
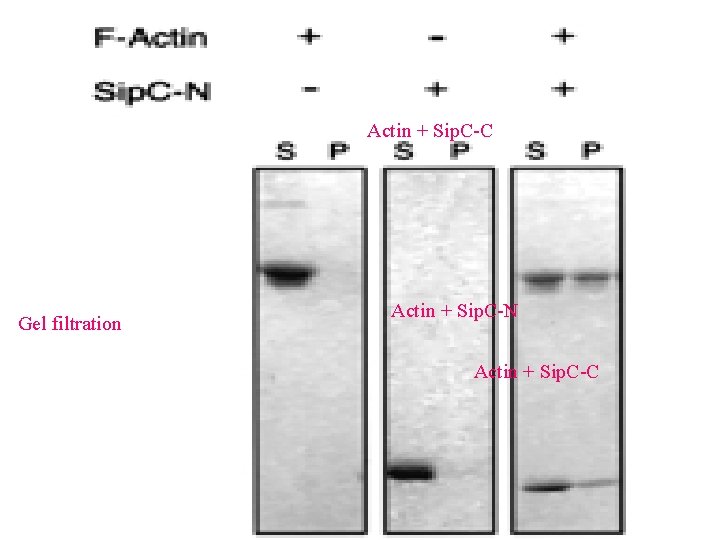
Actin + Sip. C-C Gel filtration Actin + Sip. C-N Actin + Sip. C-C
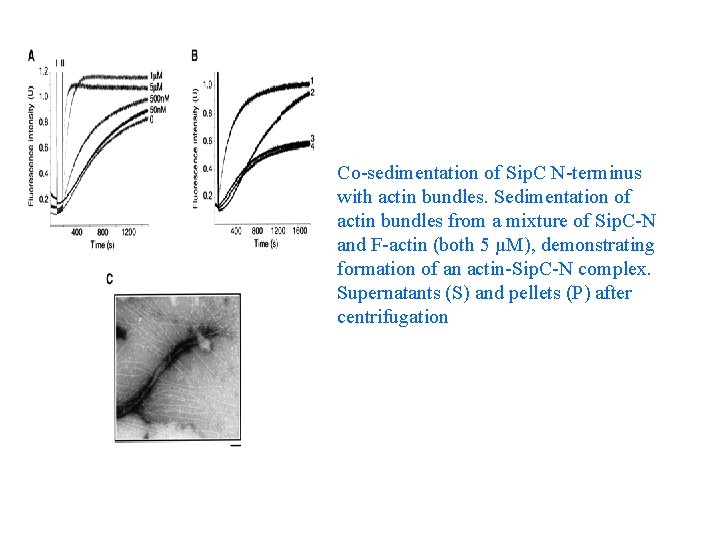
Co-sedimentation of Sip. C N-terminus with actin bundles. Sedimentation of actin bundles from a mixture of Sip. C-N and F-actin (both 5 µM), demonstrating formation of an actin-Sip. C-N complex. Supernatants (S) and pellets (P) after centrifugation
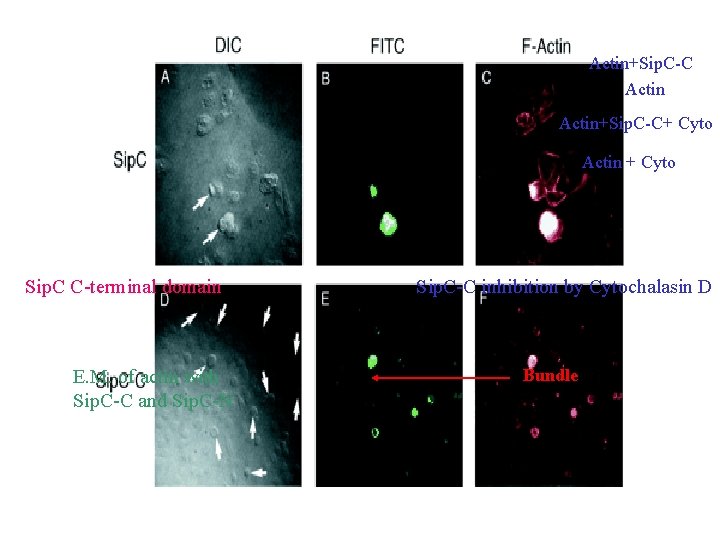
Actin+Sip. C-C+ Cyto Actin + Cyto Sip. C C-terminal domain E. M. of actin with Sip. C-C and Sip. C-N Sip. C-C inhibition by Cytochalasin D Bundle
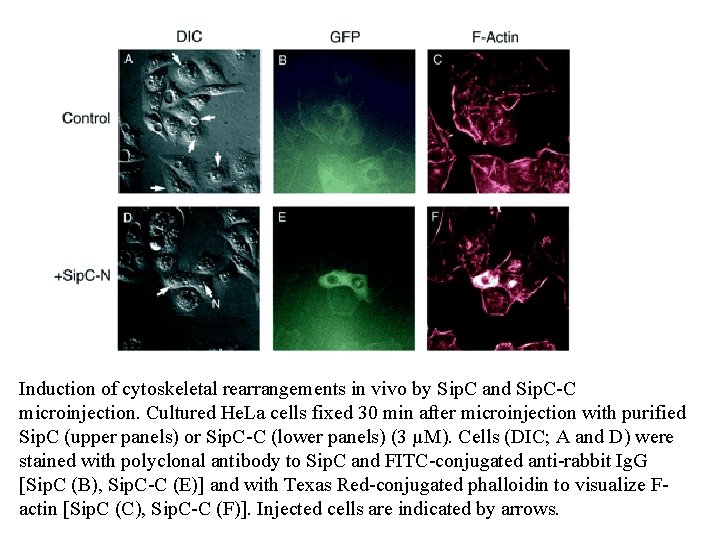
Induction of cytoskeletal rearrangements in vivo by Sip. C and Sip. C-C microinjection. Cultured He. La cells fixed 30 min after microinjection with purified Sip. C (upper panels) or Sip. C-C (lower panels) (3 µM). Cells (DIC; A and D) were stained with polyclonal antibody to Sip. C and FITC-conjugated anti-rabbit Ig. G [Sip. C (B), Sip. C-C (E)] and with Texas Red-conjugated phalloidin to visualize Factin [Sip. C (C), Sip. C-C (F)]. Injected cells are indicated by arrows.

Co-injection of Sip. C-N with GST-GFP. Cultured He. La cells (DIC; A and D) fixed 20 min after microinjection with GST-GFP alone (upper panels) or mixed with Sip. C-N (lower panels) (3 µM). GST-GFP was visualized directly [GSTGFP alone (B); + Sip. C-N (E)] and F-actin stained with Texas Red-conjugated phalloidin [GST-GFP alone (C); + Sip. C-N (F)]. Injected cells are indicated by arrows (N = nuclear injection).

- Slides: 43