Background cisplatin based chemotherapy has already been the

Background
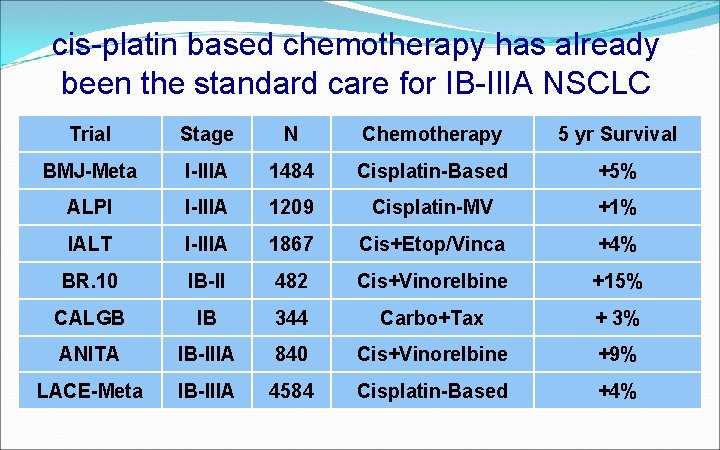
cis-platin based chemotherapy has already been the standard care for IB-IIIA NSCLC Trial Stage N Chemotherapy 5 yr Survival BMJ-Meta I-IIIA 1484 Cisplatin-Based +5% ALPI I-IIIA 1209 Cisplatin-MV +1% IALT I-IIIA 1867 Cis+Etop/Vinca +4% BR. 10 IB-II 482 Cis+Vinorelbine +15% CALGB IB 344 Carbo+Tax + 3% ANITA IB-IIIA 840 Cis+Vinorelbine +9% LACE-Meta IB-IIIA 4584 Cisplatin-Based +4%

NCCN: 2007 NSCLC treatment guideline (conclusion section) Prevent cigarette smoking – Prevent Lung Cancer Adjuvant cisplatin-based chemotherapy reduces the risk of death in completely resected patients Multimodality therapy is the standard of care for stage IIIA. Cure is possible Targeted agents hold great promise Personalized therapy, choosing treatments based on specific characteristics of tumors and patients has the potential to make NSCLC a “chronic disease. ”

Situation and issues concerning NSCLC postoperative adjunctive therapy in China Only 5 -year post-operative survival reports are available, while large-scale investigation results for post-operative adjuvant chemotherapy are absent China has the largest population of lung cancer cases, but lacks of corresponding phase III clinical study Clinical trials on lung cancer have already fallen behind neighbouring countries or areas, like Japan, Korea, Taiwan and Hongkong Results from Phase III clinical trials of adjuvante therapy following surgery for NSCLC is needed for chinese edition of guideline

Survival rate of NSCLC patients after surgical excision Surgery outcome for NSCLC in China Reporter (year) Number of surgery cases 5 -year survival rate Xiaomai Huang et. (1987) 765 33. 6% Meilin Liao et. (1988) 2636 40. 6% Jia’an Ding et. (1988) 2048 31. 8% Guangyan Pei et. (1991) 1336 33. 4% Shiye Lee et. (1992) 3568 34. 6% Nugang Zhang et. (1993) 2025 40. 8% Yilong Wu et. (1999) 1757 39. 8% Dekang Fang et. (1999) 1471 42. 4% Yan Sun et. Medical Oncology,People‘s Medical Publishing House 2001, P 658

The research of multi-disciplinary treatment for NSCLC has been the important part of China National Science & Technology Infrastructure Program of 11 th 5 -year Plan, which consists of series of clinical tries according to evidence based medicine principals and basic studies. The research will provide important theoretic support for the guideline for NSCLC, which is suitable for Chinese population.

Limitations of the traditional adjuvant chemotherapy LACE study indicates that post operative platin-containing chemotherapy only increased the 5 -year survival rate by 5% Searching for new treatment modality is ergent According to the treatment outcome for late stage tumor, chemotherapy combined with target treatment (antiangiogenesis therapy) Improves efficacy and is feasible. Such as E 4599, Endostar combined chemotherapy, Shenyi combined chemotherapy, etc.

Rationales for adjuvant chemotherapy combined with anti-angiogenesis agents for NSCLC Angiogenesis is a complicated process mediated by vascular endothelial growth factors (VEGF) Treatment on angiogenesis will put the tumorof tumor This therapy will reverse the resistance and their receptors cells, downstream ofexisting vesselstumor into necrosis effect on microvasculature system to atrophy
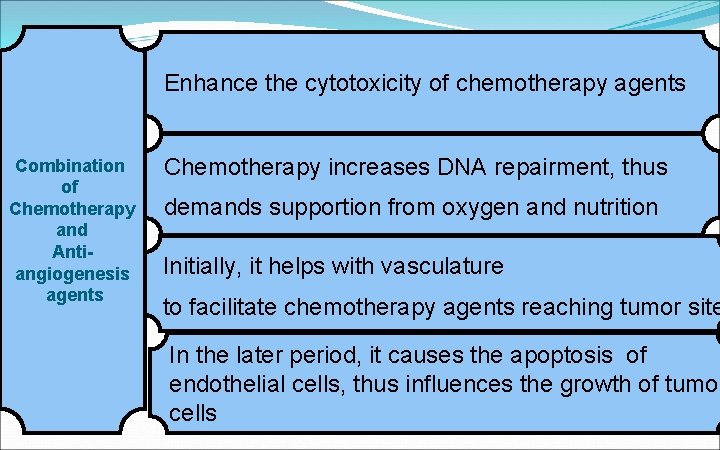
Enhance the cytotoxicity of chemotherapy agents Combination of Chemotherapy and Antiangiogenesis agents Chemotherapy increases DNA repairment, thus demands supportion from oxygen and nutrition Initially, it helps with vasculature to facilitate chemotherapy agents reaching tumor site In the later period, it causes the apoptosis of endothelial cells, thus influences the growth of tumor cells
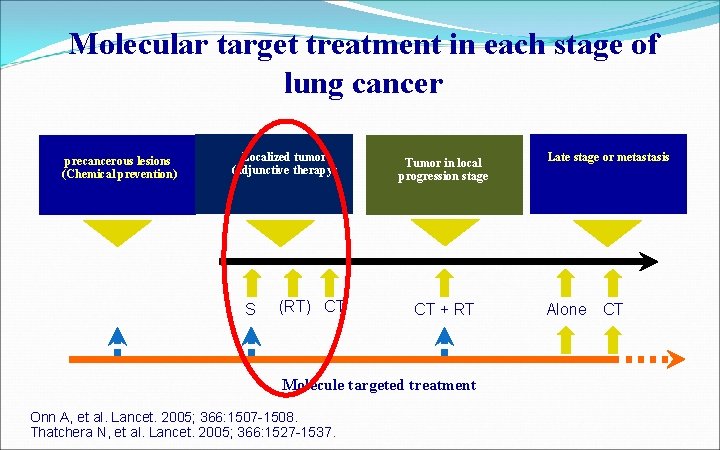
Molecular target treatment in each stage of lung cancer precancerous lesions (Chemical prevention) Localized tumor (adjunctive therapy) S (RT) CT Tumor in local progression stage CT + RT Molecule targeted treatment Onn A, et al. Lancet. 2005; 366: 1507 -1508. Thatchera N, et al. Lancet. 2005; 366: 1527 -1537. Late stage or metastasis Alone CT
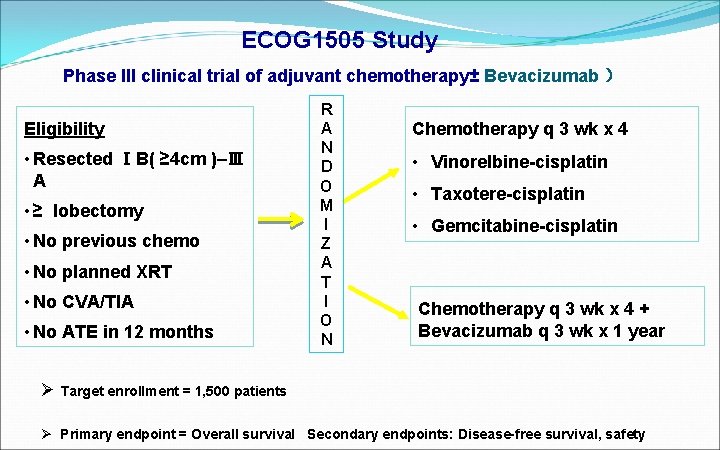
ECOG 1505 Study Phase III clinical trial of adjuvant chemotherapy± Bevacizumab ) Eligibility • Resected ⅠB( ≥ 4 cm )–Ⅲ A • ≥ lobectomy • No previous chemo • No planned XRT • No CVA/TIA • No ATE in 12 months R A N D O M I Z A T I O N Chemotherapy q 3 wk x 4 • Vinorelbine-cisplatin • Taxotere-cisplatin • Gemcitabine-cisplatin Chemotherapy q 3 wk x 4 + Bevacizumab q 3 wk x 1 year Ø Target enrollment = 1, 500 patients Ø Primary endpoint = Overall survival Secondary endpoints: Disease-free survival, safety

regimen selection: NVB/DDP The most widely used regimen in adjuvant chemotherapy for NSCLC : IALT,BR. 10, ANITA • The most stages are benefited: IIA-IIIA • Cost-effective: follow the rules in therapy economics

Endostar � Endostar is a recombinant human endostatin. � Preclinical data revealed that Endostar could inhibit tumor angiogenesis and growth. � In a phase III trial, patients with advanced NSCLC were treated with cisplatin/vinorelbine (NP) plus Eendostar or placebo. The addition of Endostar to NP regimen resulted in higher response rate, median time to progression, and clinical benefit rate compared with NP alone in advanced NSCLC patients. (Sun Y, Wang JW, Liu Y et al, Results of phase III trial of rhendostatin (YH-16) in advanced non-small lung cancer (NSCLC) patients. Proc ASCO 2005; 23: 7138 a) � Because of the promising results in advanced NSCLC, we investigated adjuvant NP regimen with or without Endostar in early-stage NSCLC, and the preliminary results of the first enrolled 545 patients were reported.

Objectives of the Study • Secondary objectives ØCompare both groups (with ØTo compare the OS of early- or without Endostar) : Relapse free survival (RFS) stage NSCLC patients who ØSafety receive either Navelbine+ ØAnalyze the quality of life ØEstablish prognosis factors of Cis-platin NSCLC through the analysis on (NP)+ Endostar or tissue and blood samples Navelbine+ Cis-platin (NP) ØInvestigate the relationship between smoking and prognosis after operation • Primary Objective
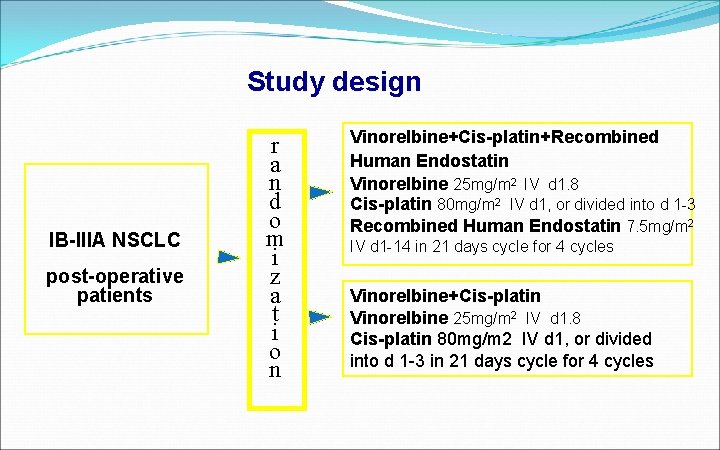
Study design IB-IIIA NSCLC post-operative patients r a n d o m i z a t i o n Vinorelbine+Cis-platin+Recombined Human Endostatin Vinorelbine 25 mg/m 2 IV d 1. 8 Cis-platin 80 mg/m 2 IV d 1, or divided into d 1 -3 Recombined Human Endostatin 7. 5 mg/m 2 IV d 1 -14 in 21 days cycle for 4 cycles Vinorelbine+Cis-platin Vinorelbine 25 mg/m 2 IV d 1. 8 Cis-platin 80 mg/m 2 IV d 1, or divided into d 1 -3 in 21 days cycle for 4 cycles

Sample size Main reference:ANITA study and ECOG 1505 study The median post-operative survival time in the NP group is defined as 55. 8 months (ANITA 65. 8 months) Recombined Human Endostatin combined therapy is expected to prolong median survival time by 27% (26. 5%, for ECOG 1505), that is from 55. 8 months to 70. 9 months The significance level will be 0. 05 (bilateral), statistical power 80%, enrollment time 2 years, sample size for each group will be 503, lost to follow-up 10%, total size 1107.

Inclusion criteria 1. NSCLC patients who are pathologically determined 2. Clinical will be classified as IB-IIIA phase with the to suffer fromstage adenocarcinoma, squamous cell carcinoma, tumors completely (left totaltreatment lung excision, lobi pulmonis 3. Surgery dateexcised to adjunctive ≤ 8 weeks large cell carcinoma, or mixed relapse type ofofabove. evidence of tumor from examination excision, resection); The scope clearance includes performed 4. 4. ;Nosleeve before adjunctive therapy 6. Subjects have no organ dysfunction; blood routine, homonymy hilus pulmonis andmajor mediastinum lymph score nodes 5. Aged between 18~70, physical status ECOG 0~ 1; 7. Patients have no prior chemotherapy radiotherapy; (including eminence); should be trimmed. allor hepatic, renal, incisal cardioedge (examination) turn out normal; 8. Patients arefunction compliant with the treatment laboratory test indicators must meet following requirements: and follow-ups, and understand the study well, Hematology: Leukocyte≥ 4. 0× 109/L; Neutrophils≥ 2. 0× 109/L count≥ 100× 109/L; Hemoglobin≥ 95 g/L。 and Platelet sign informed consent form. Renal function: Serum bilirubin is below 1. 5 X upper normal limit; ALT and AST are below 1. 5 X upper normal limit.

Exclusion criteria 1. Classified as a pathology category which does not 2. Total or partial resection of the right lung; 3. 4. Enrollment more than weeks from the surgery 2. meet inclusion criteria; Patients areis complicated with 8 any acute away or chronic diseases date; • Uncontrolled unstable angina, myocardial infarction or mental disorders or hypertension, laboratory abnormality; those complicated 6. Before enrollment, the patients suffer from 5. Patients who arewho pregnant or breast-feeding; history or Increase symptomatic congestive heart-failure ortaking uncontrolled diseases may the risk of participating this study or 7. 7. other progressive malignant than NSCLC Have allergic constitution orresults; knownitwill tojudged be allergic to inhave the past 12 months; this. Arrhythmia test drug, or an influence ontumor the study is 8. Patients who are poorly compliant to diagnosed the treatment; 8. ECG excluded; Except nonmelanoma skin cancer, shows there Isfor ischemia pathology orare clinically 8. be any test drug; by • investigators that under some circumstance patients not 9. cervical Patients who are judged by the investigators not 9. in situ carcinoma, and cured early-stage heart valve disease; suitable to participate this trial: suitable toorparticipate trial. active phase; cancer; fungi • Prostate In bacteria, virus causedthis disease
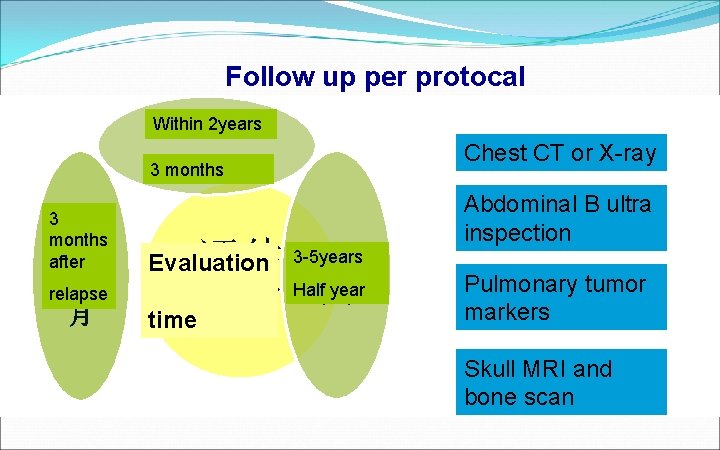
Follow up per protocal Within 2 years 2年内 3 months 3个月 3 months 肿瘤 after 复发 后3个 relapse 月 3 -5年 评估 3 -5 years Half year 半年 time时间 Evaluation Chest CT or X-ray 胸部CT或X片 Abdominal B ultra 腹部B超 inspection Pulmonary tumor 肺系统肿瘤标志物 markers 头颅MRI及骨扫描 Skull MRI and bone scan

Progress of the project

Total 43 centers 01 Cancer Hospital, Chinese Academy of Medical Sciences 02 03 Capital Medical University Beijing Chao. Yang Hospital 04 05 Shangdon Cancer Hospital & Institute 06 07 The Tumor Hospital of HARBIN Medical University 08 Beijing Cancer Hospital 09 Shandong University Qilu Hospital 10 Zhongshan Hospital Fudan University 11 Jinlin Cancer Hospital & Institute 12 The First Affiliated Hospital of Soochow University 13 Beijing Chest Hospital 14 The First Affiliated Hospital of Nanchang University 15 Shanghai Shuguang Hospital of Shanghai University of 16 T. C. M 17 Tumor Hospital of Guangzhou Medical college 18 The First Affiliated Hospital of Fujian Medical University Zhejiang Cancer Hospital 19 Shandong Chest Hospital 20 Sichuan Cancer Hospital 21 23 25 22 24 26 Jiangxi Cancer Hospital Liaoning Cancer Hospital & Institute The Affiliated Hospital of Xinjiang Medical University 28 The Affiliated Hospital of Medical College Gingdao University 30 Shanghai First People’s Hospital 32 Tianjin Medical University General Hospital 34 Huadong Hospital Affiliated to Fudan University 36 Shanghai Tenth People’s Hospital 37 Yunnan 1 st people's hospital Fujian Medical college attached Xiehe Hospital The First Attached Hospital of Wenzhou Medical college Tianjin Medical University Cancer Institute and Hospital Yunnan Cancer Hospital & Institute Sichuan University - West China Center of Medical Sciences Sir Run Shaw Hospital of Zhejiang University School of Medicine The 2 nd Affiliated Hospital of Harbin Medical University Changhai Hospital of Shanghai 38 Xinhua Hospital Affiliated to Shanghai Jiaotong University 39 The First Hospital of China Medical University 40 Tongji Medical College of Huazhong University of Science & Technology 41 The Second Affiliated Hospital of Dalian Medical University 42 The First Affiliated Hospital of Zhejiang University 27 29 31 33 35 Beijing Hospital Chinese Academy of Medical Sciences & Peking Union Medical College Shanghai Chest Hospital affiliated to Shanghai Jiaotong University

Approved by Ethical Commission, in April, 2007
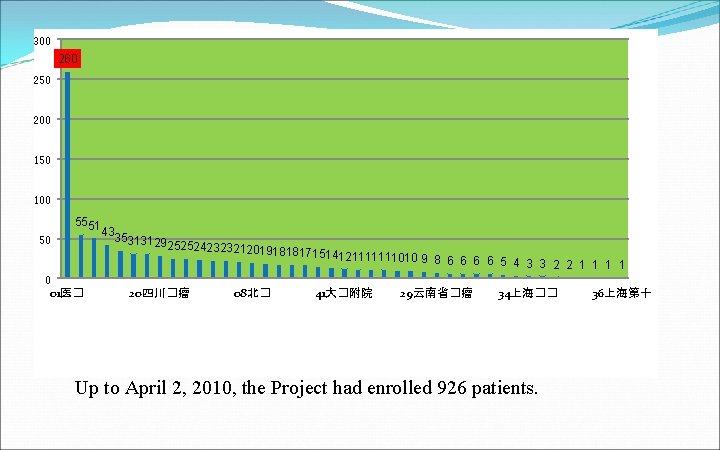
300 260 250 200 150 100 50 5551 43 35313129 25252423232120191818 171514121111111010 9 8 6 6 5 4 3 3 2 2 1 1 0 01医� 20四川�瘤 08北� 41大�附院 29云南省�瘤 34上海�� Up to April 2, 2010, the Project had enrolled 926 patients. 36上海第十

Comparison of enrollment speed with large, multinational, multi-center clinical trials Study name Study date JBR 10 ANITA CALGB 9633 CN 11(5) 1994. 72001. 4 1994. 112000. 11 1996. 92003. 11 2007. 62010. 3 Number 482 840 344 926 Enrollment time 6 years+9 months 81 months Patients enrolled per month 6 6 years 11. 7 72 months 101 centers 7 years+2 months 86 months 2 years+9 months 33 months 4 28
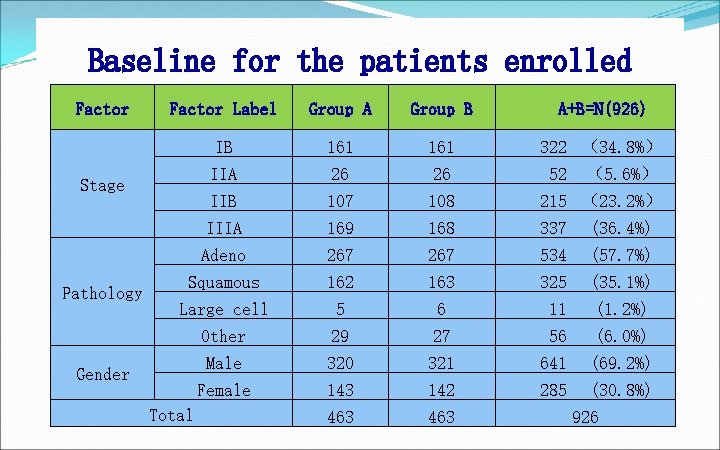
Baseline for the patients enrolled Factor Label IB IIA Stage IIB IIIA Adeno Squamous Pathology Large cell Other Male Gender Female Total Group A Group B 161 26 107 169 267 162 5 29 320 143 463 161 26 108 168 267 163 6 27 321 142 463 A+B=N(926) 322 (34. 8%) 52 (5. 6%) 215 (23. 2%) 337 (36. 4%) 534 (57. 7%) 325 (35. 1%) 11 (1. 2%) 56 (6. 0%) 641 (69. 2%) 285 (30. 8%) 926
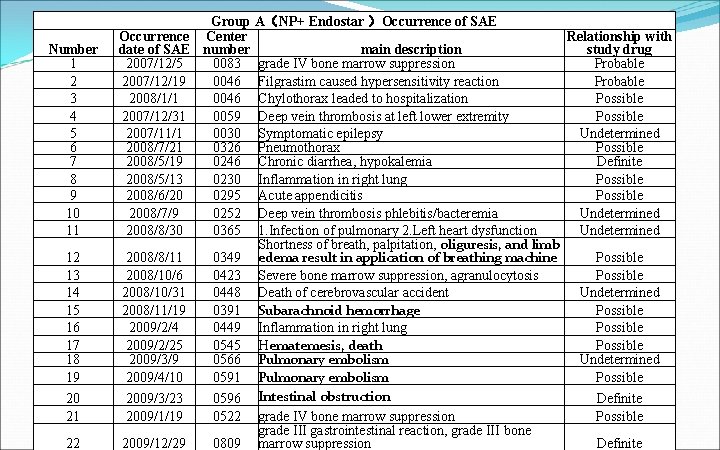
Number 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 Group A(NP+ Endostar )Occurrence of SAE Occurrence Center Relationship with date of SAE number main description study drug 2007/12/5 0083 grade IV bone marrow suppression Probable 2007/12/19 0046 Filgrastim caused hypersensitivity reaction Probable 2008/1/1 0046 Chylothorax leaded to hospitalization Possible 2007/12/31 0059 Deep vein thrombosis at left lower extremity Possible 2007/11/1 0030 Symptomatic epilepsy Undetermined 2008/7/21 0326 Pneumothorax Possible 2008/5/19 0246 Chronic diarrhea, hypokalemia Definite 2008/5/13 0230 Inflammation in right lung Possible 2008/6/20 0295 Acute appendicitis Possible 2008/7/9 0252 Deep vein thrombosis phlebitis/bacteremia Undetermined 2008/8/30 0365 1. Infection of pulmonary 2. Left heart dysfunction Undetermined Shortness of breath, palpitation, oliguresis, and limb 2008/8/11 0349 edema result in application of breathing machine Possible 2008/10/6 0423 Severe bone marrow suppression, agranulocytosis Possible 2008/10/31 0448 Death of cerebrovascular accident Undetermined 2008/11/19 0391 Subarachnoid hemorrhage Possible 2009/2/4 0449 Inflammation in right lung Possible 2009/2/25 0545 Hematemesis, death Possible 2009/3/9 0566 Pulmonary embolism Undetermined 2009/4/10 0591 Pulmonary embolism Possible 2009/3/23 0596 Intestinal obstruction Definite 2009/1/19 0522 grade IV bone marrow suppression Possible grade III gastrointestinal reaction, grade III bone 2009/12/29 0809 marrow suppression Definite
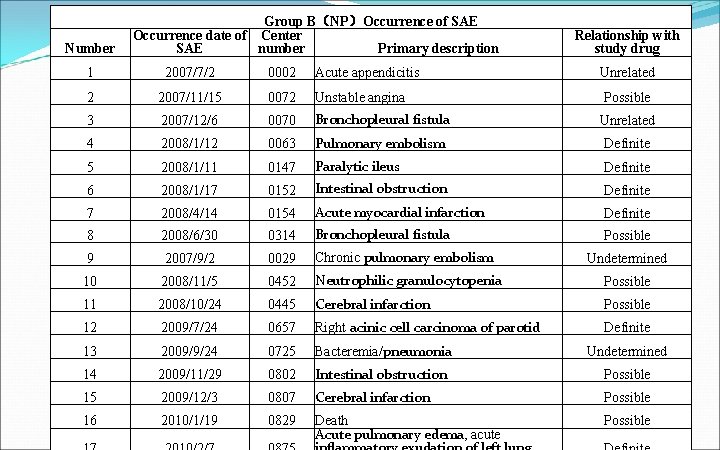
Number Group B(NP)Occurrence of SAE Occurrence date of Center SAE number Primary description Relationship with study drug 1 2007/7/2 0002 Acute appendicitis Unrelated 2 2007/11/15 0072 Unstable angina 3 2007/12/6 0070 Bronchopleural fistula Unrelated 4 2008/1/12 0063 Pulmonary embolism Definite 5 2008/1/11 0147 Paralytic ileus Definite 6 2008/1/17 0152 Intestinal obstruction Definite 7 2008/4/14 0154 Acute myocardial infarction Definite 8 2008/6/30 0314 Bronchopleural fistula Possible 9 2007/9/2 0029 Chronic pulmonary embolism Undetermined 10 2008/11/5 0452 Neutrophilic granulocytopenia Possible 11 2008/10/24 0445 Cerebral infarction Possible 12 2009/7/24 0657 Right acinic cell carcinoma of parotid Definite 13 2009/9/24 0725 Bacteremia/pneumonia Undetermined 14 2009/11/29 0802 Intestinal obstruction Possible 15 2009/12/3 0807 Cerebral infarction Possible 16 2010/1/19 0829 Death Acute pulmonary edema, acute Possible

Interim Summary of the first 588 patients

There are 43 patients (7. 3%) who did not undergo chemotherapy after randomization, thus the data from the rest 545 patients (92. 7%) was analyzed for survival and safty. 78. 63 % patients in group A and 76. 33% patients in group B completed 4 cycles of chemotherpy. Up to analysis, 26. 4% patients in group A and 23. 6% patients in group B had the relapsed disease.
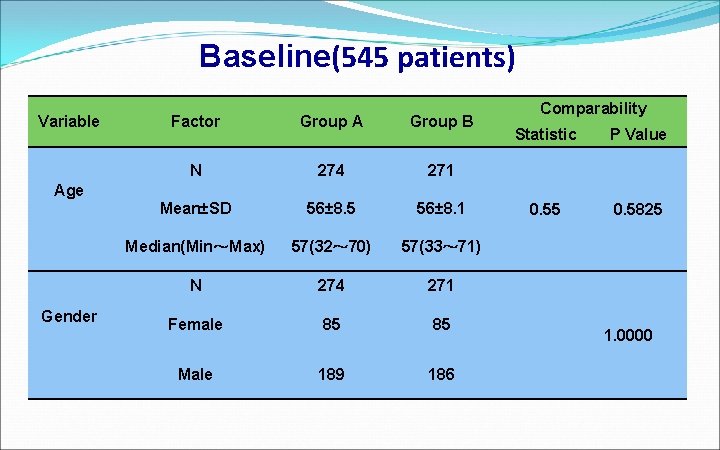
Baseline(545 patients) Variable Age Gender Factor Group A Group B N 274 271 Mean±SD 56± 8. 5 56± 8. 1 Median(Min~Max) 57(32~ 70) 57(33~ 71) N 274 271 Female 85 85 Male 189 186 Comparability Statistic P Value 0. 55 0. 5825 1. 0000
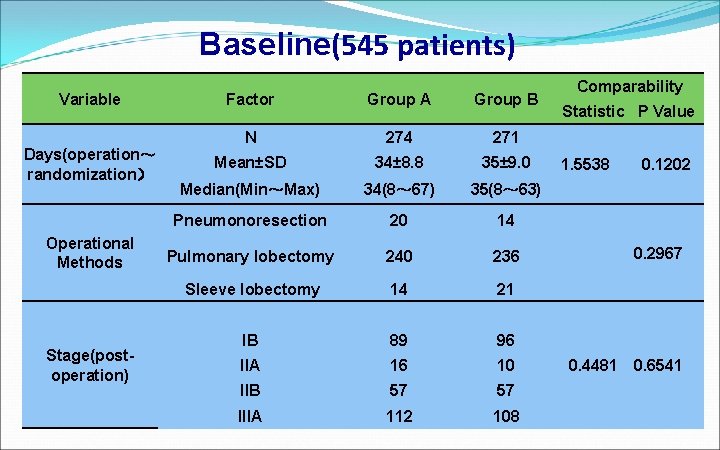
Baseline(545 patients) Variable Days(operation~ randomization) Operational Methods Stage(postoperation) Factor Group A Group B N 274 271 Mean±SD 34± 8. 8 35± 9. 0 Median(Min~Max) 34(8~ 67) 35(8~ 63) Pneumonoresection 20 14 Pulmonary lobectomy 240 236 Sleeve lobectomy 14 21 IB 89 96 IIA 16 10 IIB 57 57 IIIA 112 108 Comparability Statistic P Value 1. 5538 0. 1202 0. 2967 0. 4481 0. 6541
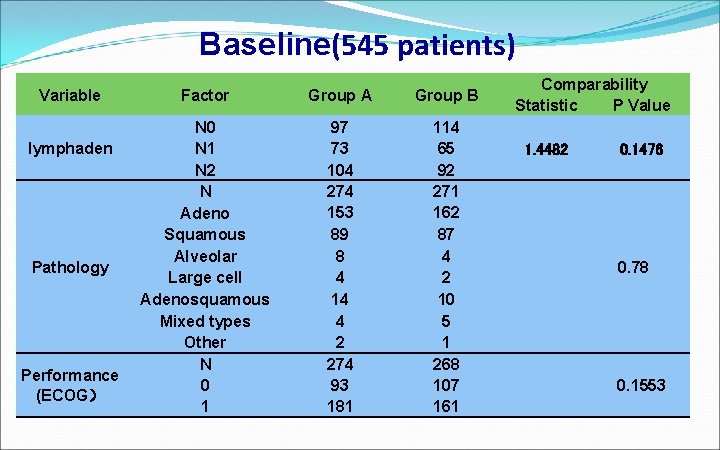
Baseline(545 patients) Variable lymphaden Pathology Performance (ECOG) Factor Group A Group B N 0 N 1 N 2 N Adeno Squamous Alveolar Large cell Adenosquamous Mixed types Other N 0 1 97 73 104 274 153 89 8 4 14 4 2 274 93 181 114 65 92 271 162 87 4 2 10 5 1 268 107 161 Comparability Statistic P Value 1. 4482 0. 1476 0. 78 0. 1553
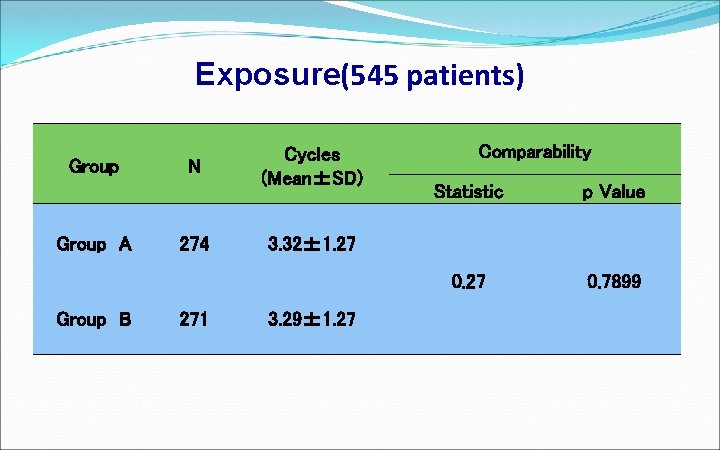
Exposure(545 patients) Group A Group B N 274 271 Cycles (Mean±SD) Comparability Statistic p Value 0. 27 0. 7899 3. 32± 1. 27 3. 29± 1. 27

Results (545 patients) Endpoints Group A Group B Median overall survival (m) - - Median RFS(m) 21. 9 18 (p=0. 3257) Survival time: because the follow-up for most patients is not up to 5 years, the majority of them survive. Therefore, the comparison on the median survival time of both groups is not available.
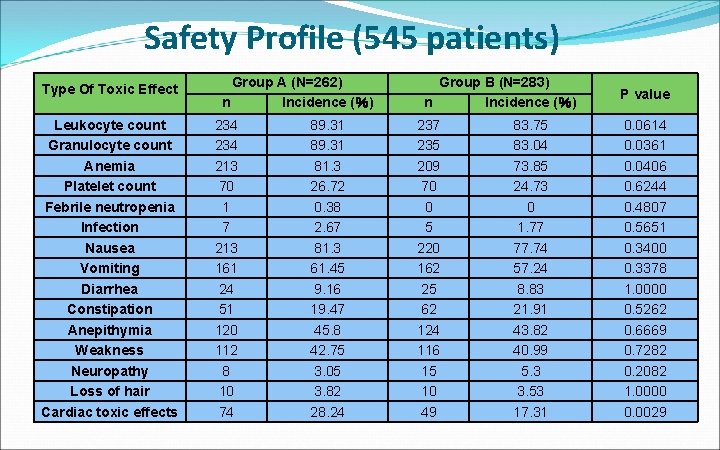
Safety Profile (545 patients) Type Of Toxic Effect Leukocyte count Granulocyte count Anemia Platelet count Febrile neutropenia Infection Nausea Vomiting Diarrhea Constipation Anepithymia Weakness Neuropathy Loss of hair Cardiac toxic effects Group A (N=262) n Incidence (%) 234 213 70 1 7 213 161 24 51 120 112 8 10 74 89. 31 81. 3 26. 72 0. 38 2. 67 81. 3 61. 45 9. 16 19. 47 45. 8 42. 75 3. 05 3. 82 28. 24 Group B (N=283) n Incidence (%) 237 235 209 70 0 5 220 162 25 62 124 116 15 10 49 83. 75 83. 04 73. 85 24. 73 0 1. 77 77. 74 57. 24 8. 83 21. 91 43. 82 40. 99 5. 3 3. 53 17. 31 P value 0. 0614 0. 0361 0. 0406 0. 6244 0. 4807 0. 5651 0. 3400 0. 3378 1. 0000 0. 5262 0. 6669 0. 7282 0. 2082 1. 0000 0. 0029
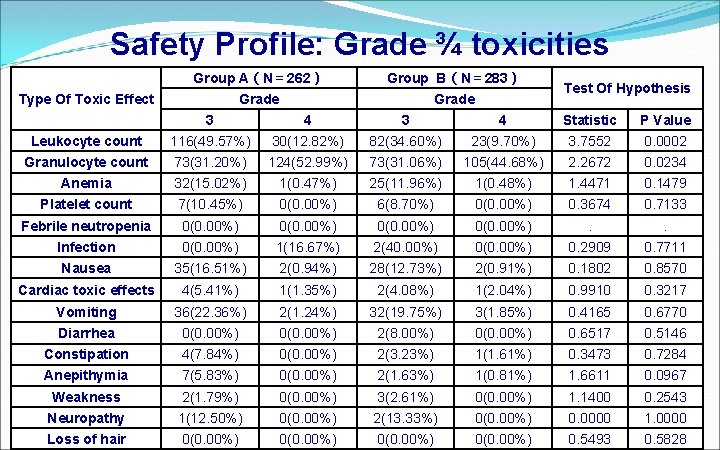
Safety Profile: Grade ¾ toxicities Group A(N= 262) Group B(N= 283) Grade Type Of Toxic Effect Test Of Hypothesis 3 4 Statistic P Value Leukocyte count 116(49. 57%) 30(12. 82%) 82(34. 60%) 23(9. 70%) 3. 7552 0. 0002 Granulocyte count 73(31. 20%) 124(52. 99%) 73(31. 06%) 105(44. 68%) 2. 2672 0. 0234 Anemia 32(15. 02%) 1(0. 47%) 25(11. 96%) 1(0. 48%) 1. 4471 0. 1479 Platelet count 7(10. 45%) 0(0. 00%) 6(8. 70%) 0(0. 00%) 0. 3674 0. 7133 Febrile neutropenia 0(0. 00%) . . Infection 0(0. 00%) 1(16. 67%) 2(40. 00%) 0(0. 00%) 0. 2909 0. 7711 Nausea 35(16. 51%) 2(0. 94%) 28(12. 73%) 2(0. 91%) 0. 1802 0. 8570 Cardiac toxic effects 4(5. 41%) 1(1. 35%) 2(4. 08%) 1(2. 04%) 0. 9910 0. 3217 Vomiting 36(22. 36%) 2(1. 24%) 32(19. 75%) 3(1. 85%) 0. 4165 0. 6770 Diarrhea 0(0. 00%) 2(8. 00%) 0(0. 00%) 0. 6517 0. 5146 Constipation 4(7. 84%) 0(0. 00%) 2(3. 23%) 1(1. 61%) 0. 3473 0. 7284 Anepithymia 7(5. 83%) 0(0. 00%) 2(1. 63%) 1(0. 81%) 1. 6611 0. 0967 Weakness 2(1. 79%) 0(0. 00%) 3(2. 61%) 0(0. 00%) 1. 1400 0. 2543 Neuropathy 1(12. 50%) 0(0. 00%) 2(13. 33%) 0(0. 00%) 0. 0000 1. 0000 Loss of hair 0(0. 00%) 0. 5493 0. 5828

Discuss Endostar plus NP regimen treated patients experienced a longer median relapse free survival time (21. 9 months versus 18 months) than vinorelbine plus cisplatin treated patients, although there was not statistically significantly improved at this time. Over two third of patients have finished four cycles of chemotherapy in each arm. The toxicity profiles for both treatment arms were generally mild and tolerable in this study. The patients’ enrolment and follow-up are still ongoing.

Thanks
- Slides: 38