Back to the Future Device Evaluation CMS View

Back to the Future: Device Evaluation: CMS View Marcel Salive, MD, MPH Coverage and Analysis Group

DISCLOSURES Marcel Salive, MD, MPH I have no real or apparent conflicts of interest to report.

CMS Mission & Vision Ø CMS’ Mission: l To ensure effective, up-to-date health care coverage and to promote quality care for beneficiaries. Ø CMS’ Vision: l To achieve a transformed and modernized health care system.

Informational Needs Should we pay for this service? Did we pay for the correct service correctly? Was the service we paid for performed optimally? Do patients and providers have sufficient information to make choices? = Coverage = Payment = Quality = Transparency

Steps to Coverage Determination and Payment Outside of CMS: Ø Congress determines benefit categories l This talk focuses on Medicare Part A/B Ø FDA approves drugs/devices for market Within CMS: Ø Coverage Ø Coding Ø Payment

Back to the Future: July 30, 1965

Social Security Act 1862(a)(1)(A) Reasonable & Necessary “…no payment may be made…for items or services - which, except for items and services described in a succeeding subparagraph, are not reasonable and necessary for the diagnosis or treatment of illness or injury or to improve the functioning of a malformed body member. ”

How Do We Apply R&N Today? Ø Sufficient level of confidence that evidence is adequate to conclude that the item or service: l l improves health outcomes generalizable to the Medicare population Ø Evidence assessed using standard principles of evidence-based medicine (EBM)

42 CFR 410. 32(a) “All diagnostic x-ray tests, diagnostic laboratory tests, and other diagnostic tests must be ordered by the physician who is treating the beneficiary, that is, the physician who furnishes a consultation or treats a beneficiary for a specific medical problem and who uses the results in the management of the beneficiary’s specific medical problem…”
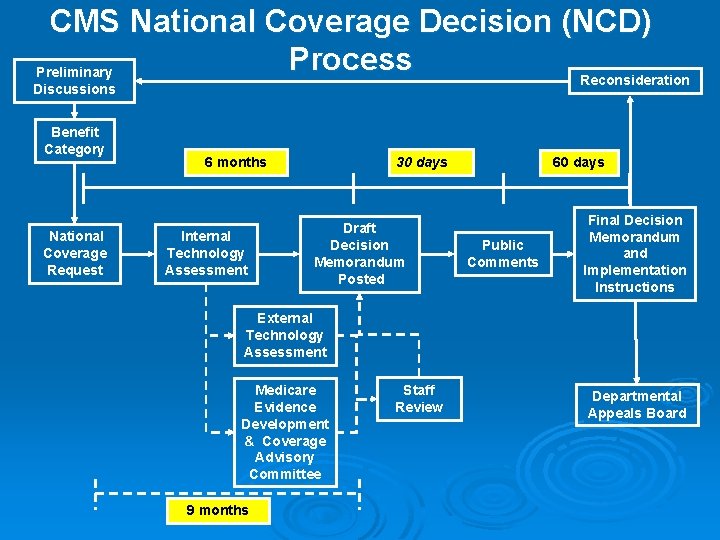
CMS National Coverage Decision (NCD) Process Preliminary Reconsideration Discussions Benefit Category National Coverage Request 6 months Internal Technology Assessment 30 days Draft Decision Memorandum Posted 60 days Public Comments Final Decision Memorandum and Implementation Instructions External Technology Assessment Medicare Evidence Development & Coverage Advisory Committee 9 months Staff Review Departmental Appeals Board

National Decisions Ø National Coverage Ø National Noncoverage Ø National Coverage with restrictions l l l Specific populations Specific providers/facilities Evidence development

Response to New Information Ø New safety information Ø Change in original evidence base Ø New competitor Ø New data on comparator

Contact Information Ø Websites: www. cms. hhs. gov/coverage 1 -800 -MEDICARE www. medicare. gov Ø Marcel Salive, MD, MPH l l 410/786 -0297 Marcel. Salive@cms. hhs. gov
- Slides: 13