Back to silicate structures nesosilicates sorosilicates cyclosilictaes phyllosilicates
Back to silicate structures: nesosilicates sorosilicates cyclosilictaes phyllosilicates inosilicates tectosilicates
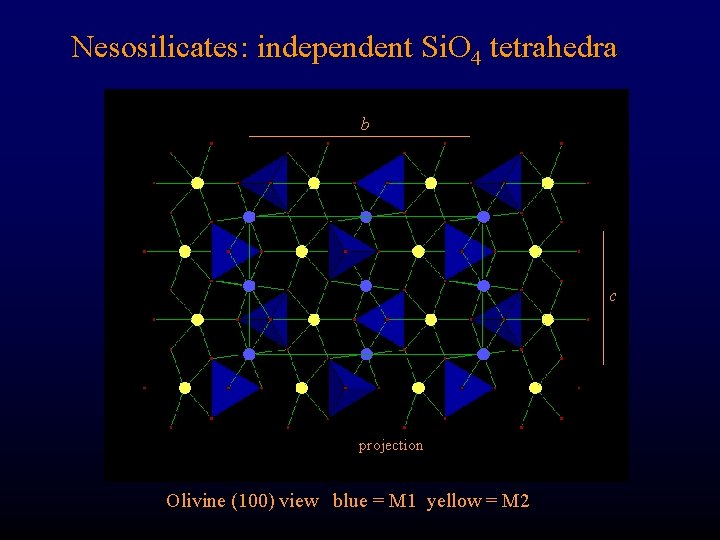
Nesosilicates: independent Si. O 4 tetrahedra b c projection Olivine (100) view blue = M 1 yellow = M 2
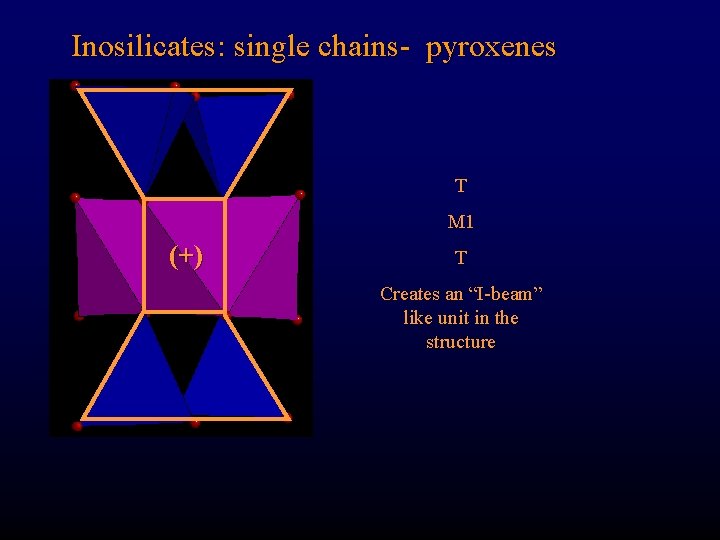
Inosilicates: single chains- pyroxenes T M 1 (+) T Creates an “I-beam” like unit in the structure
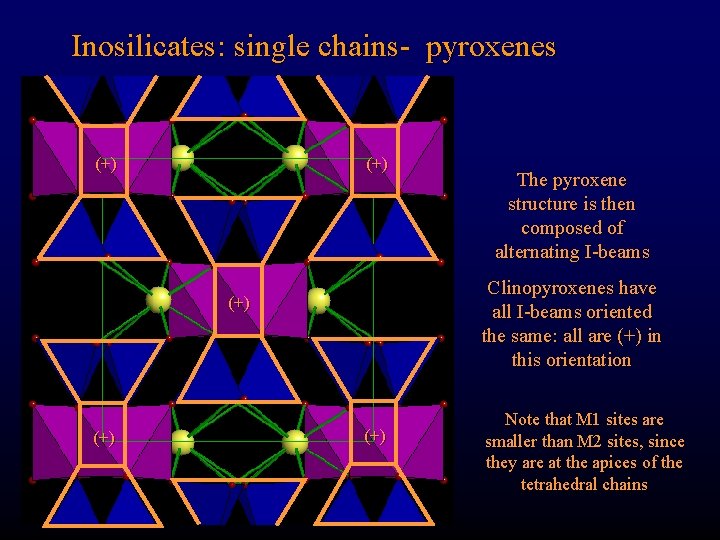
Inosilicates: single chains- pyroxenes (+) Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation (+) The pyroxene structure is then composed of alternating I-beams (+) Note that M 1 sites are smaller than M 2 sites, since they are at the apices of the tetrahedral chains
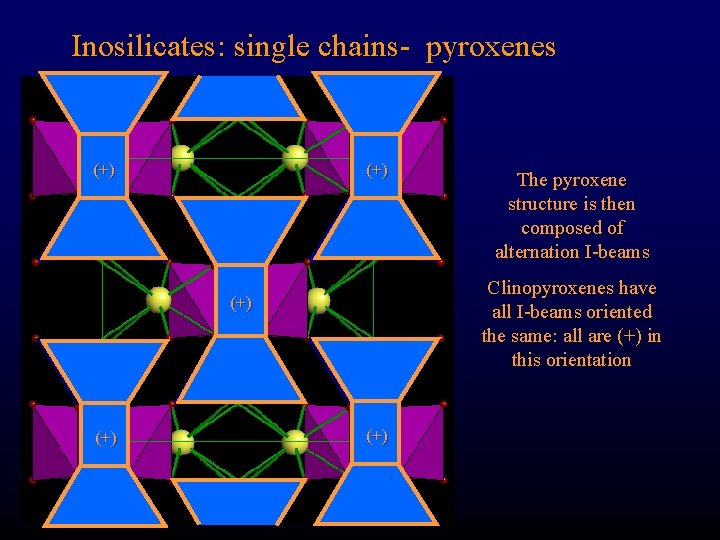
Inosilicates: single chains- pyroxenes (+) Clinopyroxenes have all I-beams oriented the same: all are (+) in this orientation (+) The pyroxene structure is then composed of alternation I-beams (+)
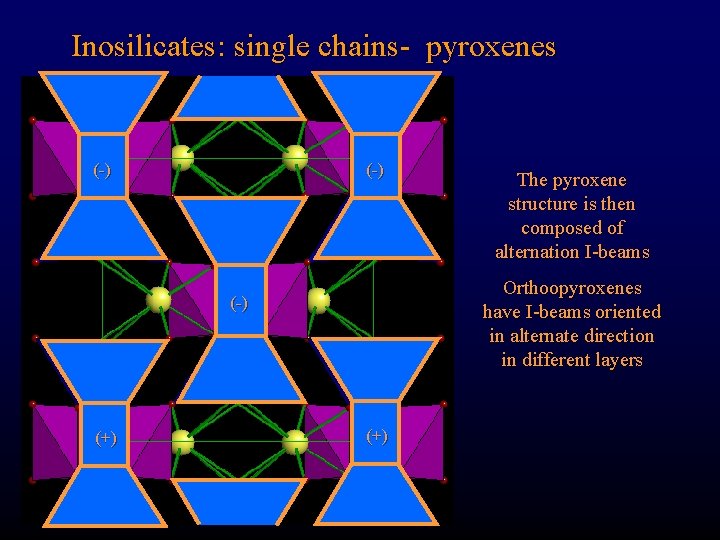
Inosilicates: single chains- pyroxenes (-) Orthoopyroxenes have I-beams oriented in alternate direction in different layers (-) (+) The pyroxene structure is then composed of alternation I-beams (+)
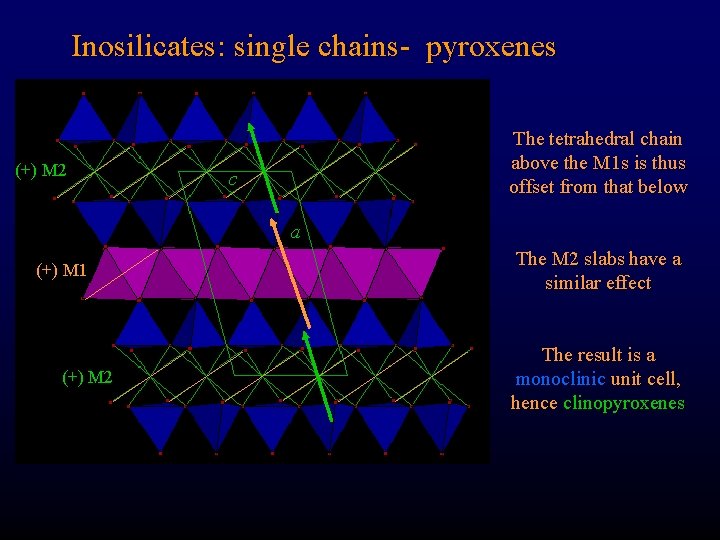
Inosilicates: single chains- pyroxenes (+) M 2 The tetrahedral chain above the M 1 s is thus offset from that below c a (+) M 1 (+) M 2 The M 2 slabs have a similar effect The result is a monoclinic unit cell, hence clinopyroxenes
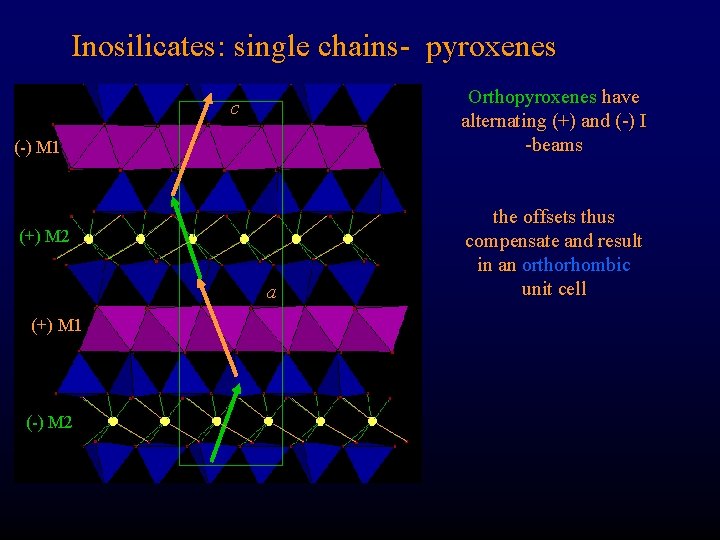
Inosilicates: single chains- pyroxenes Orthopyroxenes have alternating (+) and (-) I -beams c (-) M 1 (+) M 2 a (+) M 1 (-) M 2 the offsets thus compensate and result in an orthorhombic unit cell

Pyroxene Chemistry The general pyroxene formula: W 1 -P (X, Y)1+P Z 2 O 6 Where F W = Ca Na F X = Mg Fe 2+ Mn Ni Li F Y = Al Fe 3+ Cr Ti F Z = Si Al Anhydrous so high-temperature or dry conditions favor pyroxenes over amphiboles
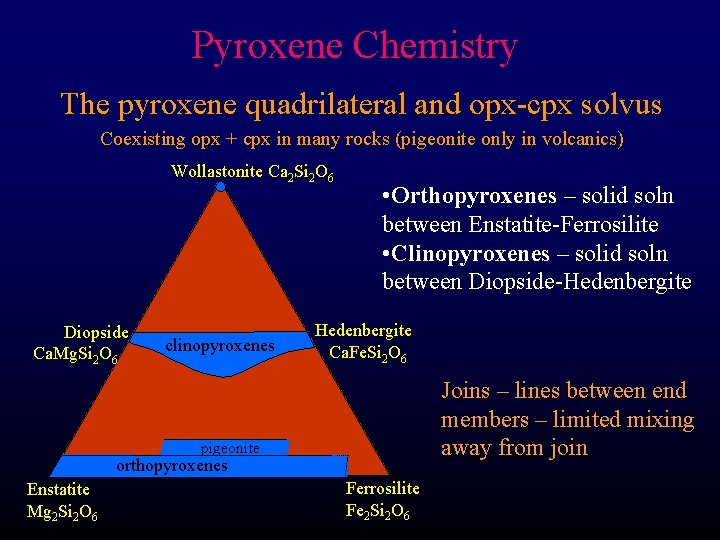
Pyroxene Chemistry The pyroxene quadrilateral and opx-cpx solvus Coexisting opx + cpx in many rocks (pigeonite only in volcanics) Wollastonite Ca 2 Si 2 O 6 Diopside Ca. Mg. Si 2 O 6 clinopyroxenes • Orthopyroxenes – solid soln between Enstatite-Ferrosilite • Clinopyroxenes – solid soln between Diopside-Hedenbergite Ca. Fe. Si 2 O 6 Joins – lines between end members – limited mixing away from join pigeonite orthopyroxenes Enstatite Mg 2 Si 2 O 6 Ferrosilite Fe 2 Si 2 O 6
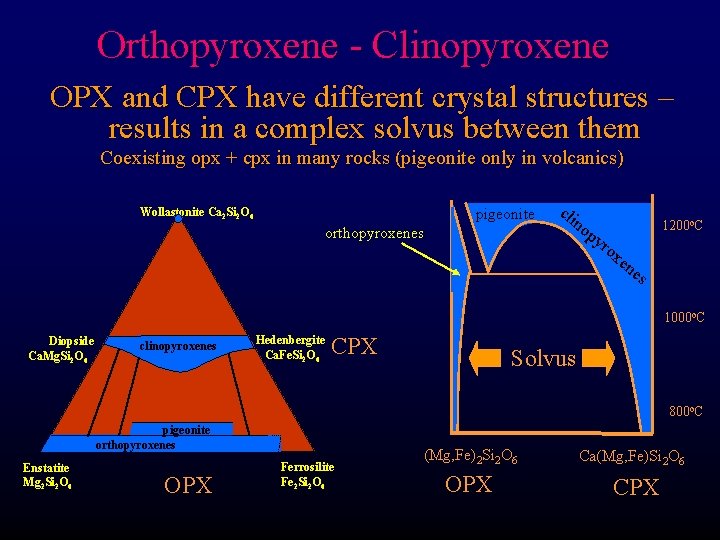
Orthopyroxene - Clinopyroxene OPX and CPX have different crystal structures – results in a complex solvus between them Coexisting opx + cpx in many rocks (pigeonite only in volcanics) pigeonite Wollastonite Ca 2 Si 2 O 6 orthopyroxenes cli no py ro 1200 o. C xe ne s 1000 o. C Diopside Ca. Mg. Si 2 O 6 clinopyroxenes Hedenbergite Ca. Fe. Si 2 O 6 CPX Solvus 800 o. C pigeonite orthopyroxenes Enstatite Mg 2 Si 2 O 6 OPX Ferrosilite Fe 2 Si 2 O 6 (Mg, Fe)2 Si 2 O 6 Ca(Mg, Fe)Si 2 O 6 OPX CPX
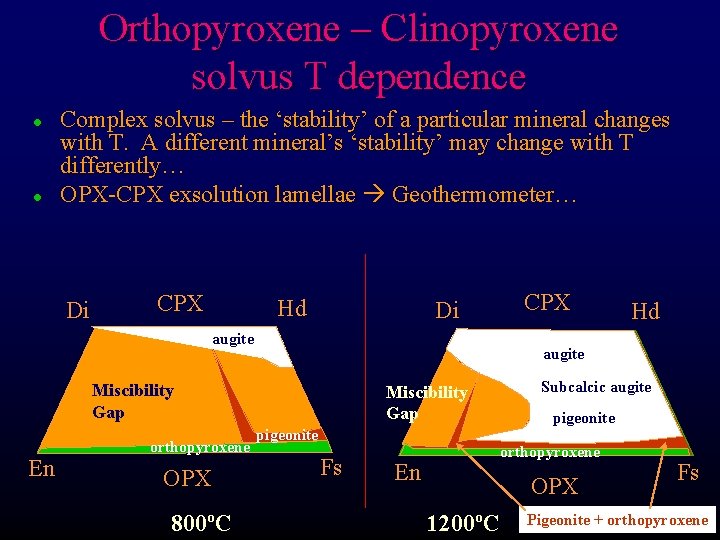
Orthopyroxene – Clinopyroxene solvus T dependence l l Complex solvus – the ‘stability’ of a particular mineral changes with T. A different mineral’s ‘stability’ may change with T differently… OPX-CPX exsolution lamellae Geothermometer… Di CPX Hd CPX Di augite Miscibility Gap En orthopyroxene OPX 800ºC Hd Subcalcic augite Miscibility Gap pigeonite Fs En pigeonite orthopyroxene OPX 1200ºC Fs Pigeonite + orthopyroxene
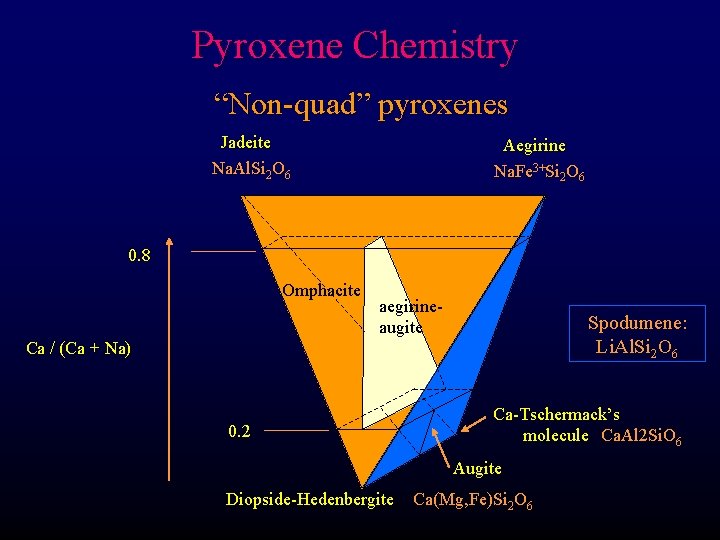
Pyroxene Chemistry “Non-quad” pyroxenes Jadeite Na. Al. Si 2 O 6 Aegirine Na. Fe 3+Si 2 O 6 0. 8 Omphacite aegirineaugite Spodumene: Li. Al. Si 2 O 6 Ca / (Ca + Na) 0. 2 Ca-Tschermack’s molecule Ca. Al 2 Si. O 6 Augite Diopside-Hedenbergite Ca(Mg, Fe)Si 2 O 6
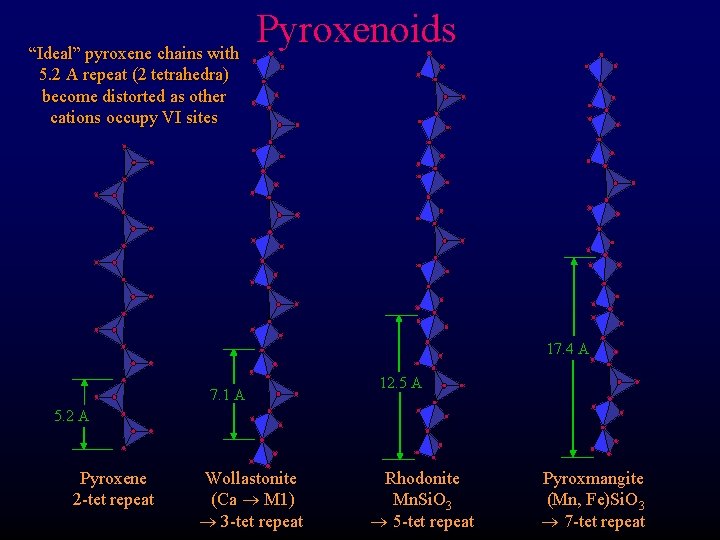
“Ideal” pyroxene chains with 5. 2 A repeat (2 tetrahedra) become distorted as other cations occupy VI sites Pyroxenoids 17. 4 A 7. 1 A 12. 5 A 5. 2 A Pyroxene 2 -tet repeat Wollastonite (Ca M 1) 3 -tet repeat Rhodonite Mn. Si. O 3 5 -tet repeat Pyroxmangite (Mn, Fe)Si. O 3 7 -tet repeat
Back to silicate structures: nesosilicates sorosilicates cyclosilictaes phyllosilicates inosilicates tectosilicates
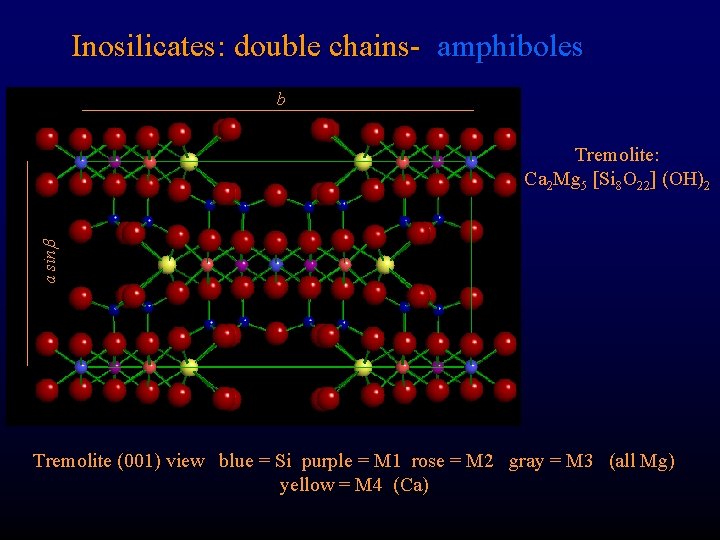
Inosilicates: double chains- amphiboles b a sin Tremolite: Ca 2 Mg 5 [Si 8 O 22] (OH)2 Tremolite (001) view blue = Si purple = M 1 rose = M 2 gray = M 3 (all Mg) yellow = M 4 (Ca)

Inosilicates: double chains- amphiboles b a sin Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Same I-beam architecture, but the I-beams are fatter (double chains) Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe)

Inosilicates: double chains- amphiboles b (+) a sin (+) (+) Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 Same I-beam architecture, but the I-beams are fatter (double chains) All are (+) on clinoamphiboles and alternate in orthoamphiboles Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 M 1 -M 3 are small sites M 4 is larger (Ca) A-site is really big Variety of sites great chemical range Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Inosilicates: double chains- amphiboles Hornblende: (Ca, Na)2 -3 (Mg, Fe, Al)5 [(Si, Al)8 O 22] (OH)2 (OH) is in center of tetrahedral ring where O is a part of M 1 and M 3 octahedra (OH) Hornblende (001) view dark blue = Si, Al purple = M 1 rose = M 2 light blue = M 3 (all Mg, Fe) yellow ball = M 4 (Ca) purple ball = A (Na) little turquoise ball = H

Amphibole Chemistry See handout for more information General formula: W 0 -1 X 2 Y 5 [Z 8 O 22] (OH, F, Cl)2 W = Na K X = Ca Na Mg Fe 2+ (Mn Li) Y = Mg Fe 2+ Mn Al Fe 3+ Ti Z = Si Al Again, the great variety of sites and sizes a great chemical range, and hence a broad stability range The hydrous nature implies an upper temperature stability limit

Amphibole Chemistry Ca-Mg-Fe Amphibole “quadrilateral” (good analogy with pyroxenes) Tremolite Ca 2 Mg 5 Si 8 O 22(OH)2 Anthophyllite Mg 7 Si 8 O 22(OH)2 Actinolite Cummingtonite-grunerite Orthoamphiboles Ferroactinolite Ca 2 Fe 5 Si 8 O 22(OH)2 Clinoamphiboles Fe 7 Si 8 O 22(OH)2 Al and Na tend to stabilize the orthorhombic form in low-Ca amphiboles, so anthophyllite gedrite orthorhombic series extends to Fe-rich gedrite in more Na-Al-rich compositions

Amphibole Chemistry Hornblende has Al in the tetrahedral site Geologists traditionally use the term “hornblende” as a catch-all term for practically any dark amphibole. Now the common use of the microprobe has petrologists casting “hornblende” into end-member compositions and naming amphiboles after a well-represented end-member. Sodic amphiboles Glaucophane: Na 2 Mg 3 Al 2 [Si 8 O 22] (OH)2 Riebeckite: Na 2 Fe 2+3 Fe 3+2 [Si 8 O 22] (OH)2 Sodic amphiboles are commonly blue, and often called “blue amphiboles”

Amphibole Occurrences Tremolite (Ca-Mg) occurs in meta-carbonates Actinolite occurs in low-grade metamorphosed basic igneous rocks Orthoamphiboles and cummingtonite-grunerite (all Ca-free, Mg-Fe-rich amphiboles) are metamorphic and occur in meta-ultrabasic rocks and some meta-sediments. The Fe-rich grunerite occurs in meta-ironstones The complex solid solution called hornblende occurs in a broad variety of both igneous and metamorphic rocks Sodic amphiboles are predominantly metamorphic where they are characteristic of high P/T subduction-zone metamorphism (commonly called “blueschist” in reference to the predominant blue sodic amphiboles Riebeckite occurs commonly in sodic granitoid rocks
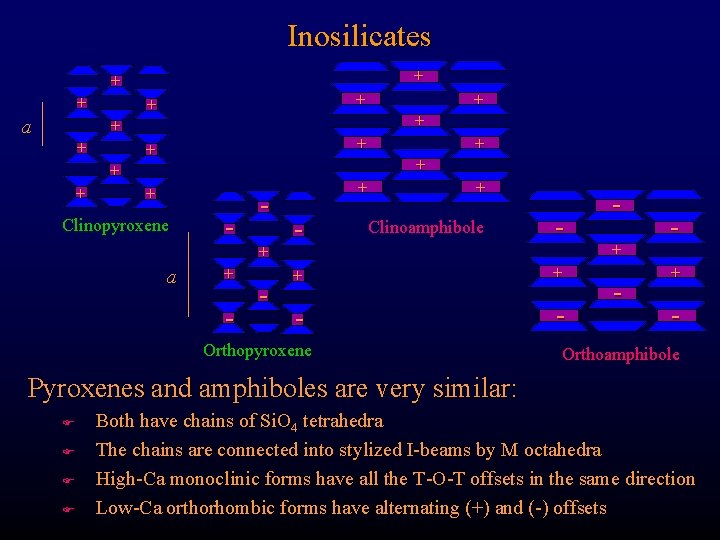
Inosilicates + + + a + + + + Clinopyroxene a + - - + Clinoamphibole - + + - - Orthopyroxene + - Orthoamphibole Pyroxenes and amphiboles are very similar: F F Both have chains of Si. O 4 tetrahedra The chains are connected into stylized I-beams by M octahedra High-Ca monoclinic forms have all the T-O-T offsets in the same direction Low-Ca orthorhombic forms have alternating (+) and (-) offsets
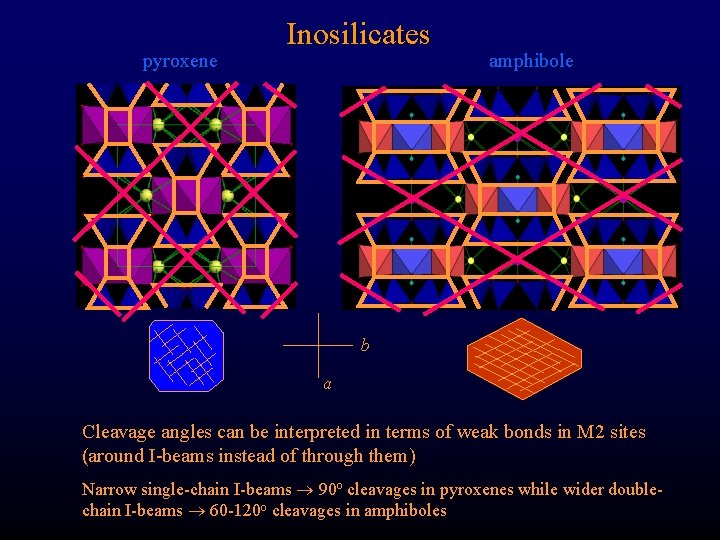
pyroxene Inosilicates amphibole b a Cleavage angles can be interpreted in terms of weak bonds in M 2 sites (around I-beams instead of through them) Narrow single-chain I-beams 90 o cleavages in pyroxenes while wider doublechain I-beams 60 -120 o cleavages in amphiboles
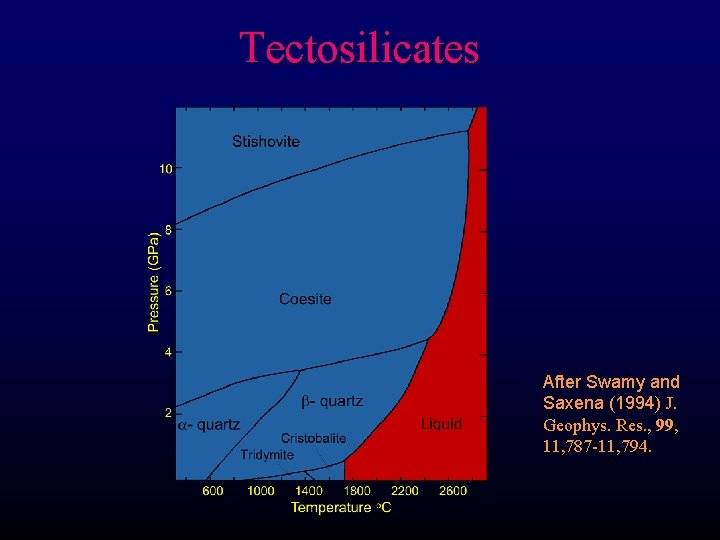
Tectosilicates After Swamy and Saxena (1994) J. Geophys. Res. , 99, 11, 787 -11, 794.

Tectosilicates Low Quartz 001 Projection Crystal Class 32

Tectosilicates High Quartz at 581 o. C 001 Projection Crystal Class 622

Tectosilicates Cristobalite 001 Projection Cubic Structure

Tectosilicates Stishovite High pressure Si. VI
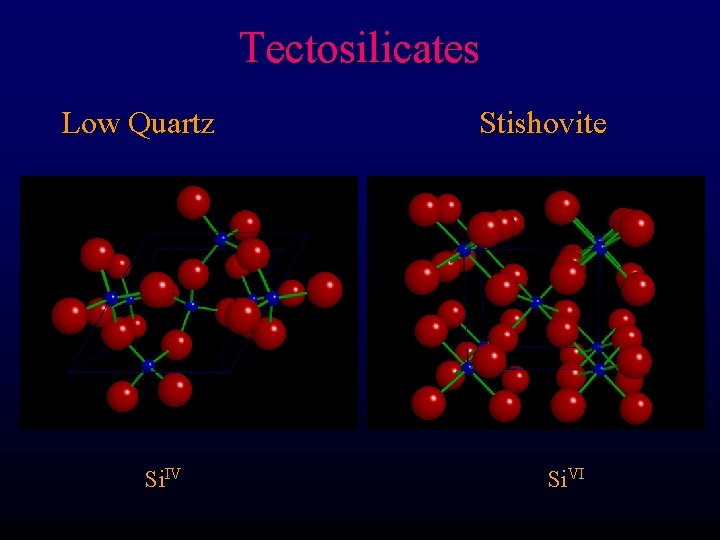
Tectosilicates Low Quartz Si. IV Stishovite Si. VI

Igneous Minerals l l Quartz, Feldspars (plagioclase and alkaline), Olivines, Pyroxenes, Amphiboles Accessory Minerals – mostly in small quantities or in ‘special’ rocks F F F F Magnetite (Fe 3 O 4) Ilmenite (Fe. Ti. O 3) Apatite (Ca 5(PO 4)3(OH, F, Cl) Zircon (Zr. Si. O 4) Titanite (Ca. Ti. Si. O 5) Pyrite (Fe. S 2) Fluorite (Ca. F 2)
- Slides: 34