Bacilus Anthracis Anthrax Sheref Hassan Rachel Handwerger Maria

Bacilus Anthracis Anthrax Sheref Hassan Rachel Handwerger Maria Garrido

“The Agent” l CDC: Category “A” Biological Agent l Vegetative cell: large, gram-positive bacillus (1. 0 -1. 5 μm by 3. 0 -5. 0 μm) l Endospore: oval, central-to-sub terminal, does not usually swell (1. 0 x 1. 5 μm); CO 2 levels within the body inhibit sporulation l Forms long chains in vitro; forms single cells or short chains in clinical samples Aerobic or facultative anaerobic; non-motile; catalase-positive Nonhemolytic on sheep blood agar; susceptible to lysis by gamma phage Colonies: ground-glass appearance and consistency of beaten-egg whites and are 2 -5 mm in diameter after 16 -18 hours of incubation l l l

“The Agent” contd. . ● In environments rich in nutrients such as glucose, amino acids, and nucleosides, spores will germinate and form vegetative cells. Vegetative bacteria survive poorly outside of mammalian host. ● In a nutrient-deficient setting, vegetative cells will form spores. ● *Spores have been shown to survive in the environment for more than 40 years.

History of Anthrax: Early History l l l The first recorded report of anthrax was during the fifth Egyptian Plague in 1500 B. C. responsible for the death of a large amount of livestock. The sixth Egyptian Plague, also known as the “plague of boils” was probably the first recorded instance of cutaneous anthrax and was responsible for many deaths. The “Black Bane” in Europe during the 1600 s killed well over 60, 000 cattle and typified anthrax symptoms. 1863: French biologist Casimir-Joseph Davaine makes B. A. the first microorganism to be conculsively linked to a disease. 1876: B. A. isolated in pure culture for the first time by Robert Koch Was also called: wool sorter’s disease, Bradford disease, and rag picker’s disease.

History of Anthrax: Early History l l l 1897: The Bradford Rules are passed as law Anthrax Investigation Board. 1919: The Anthrax Prevention Act passed in England established a wool-disinfecting complex in Liverpool. Similar laws, gov’t regulations, better disinfectant methods and technology thereafter reduced Anthrax cases linked to occupational hazards, as well as making Anthrax viability and outbreaks rare in the modern world.

History of Anthrax: Natural Outbreaks Animal: o 1945: Iran outbreak causes 1 million sheep deaths

History of Anthrax: Natural Outbreaks - cont. . . Inhalation: o o o U. S. 1900 -1978: only 18 reported cases mostly among high risk groups such as goat mill or goatskin workers and wool or tannery workers (only 2 were laboratory associated) New Hampshire 1957: 5 cases reported in goat-hair processing plant No Case of inhalation anthrax was reported since 1978 in the U. S. prior to post-Sept. 11 th attacks.

History of Anthrax: Natural Outbreaks - cont. . . Cutaneous (most common): o o o U. S. 1944 -1994: 224 cases reported New Hampshire 1957: 4 cases reported in goat-hair processing plant Zimbabwe 1979 -1985: largest reported epidemic with over 10, 000 reported human cases and 182 deaths (nearly all cutaneous); explained by spread via insect vectors or contaminated meat; however, biological warfare tactics have been also postulated

History of Anthrax: Natural Outbreaks - cont. . . Gastrointestinal (uncommonly reported): 2 distinct syndromes: oral pharyngeal (unusual manifestation of infection and is of more interest) & abdominal disease o Since mid 1970’s: small outbreaks reported in Africa and Asia o Northern Thailand 1982: 24 cases of oral pharyngeal reported due to the consumption of contaminated buffalo meat Ø 1987: 14 cases involving both oral pharyngeal and abdominal disease Ø

Statistics of Anthrax Occurrences In the U. S. : • • • Approximately 130 cases occurred annually in the early 1900’s This number decreased with less than 10 cases reported each year since the early 1960’s and no occurrences from 1978 until after Sept. 11 th 2001 95% of naturally occurring cases are cutaneous and 5% are due to inhalation. (cases of gastrointestinal anthrax have not been recognized yet in the U. S. ) • • Only 18 cases of naturally occurring inhalation cases have been reported during the 20 th century with the most recent in 1976 Since 1990, only 2 cases (1992 and 2000) of naturally occurring infection have been reported (both cutaneous).

Statistics of Anthrax Occurrences Globally: • • • Approximately 2, 000 - 20, 000 cases of anthrax occur each year. Most cases are cutaneous with inhalation and gastrointestinal being less frequent. Human cases usually follow disease occurrences in ruminants and are most prevalent in Africa, the Middle East, and parts of Southeast Asia.

“Travelers Beware” Anthrax in animals is hyper-endemic or endemic in: (caution when eating meat in these areas) w w w w Most areas of the Middle East Most areas of equatorial Africa Mexico and Central America Chile, Argentina, Peru, Bolivia Certain Southeast Asian countries such as Myanam, Vietnam, Cambodia, & Thailand Papua New Guinea China Some Mediterranean Countries (for more information see WHOCC)

Bacillus anthracis l Review: Positive l Rod shaped l Aerobic l Spore Forming l Non-motile A l Gram A 1 A 2 A 3 A 4 B 1 B 2 B

Bacillus anthracis exists in two forms: 1. Inert spore form – – – 1 μm Rod shaped Aerobic (free O 2 present) Highly Resistant to extreme conditions (e. g. temperature, weather, radiation, etc. ) Spores have been known to last up to 40 yrs

Bacillus anthracis 2. Vegetative form – – 1 – 1. 5 μm by 5 – 8 μm Square-ended rod Anaerobic environment of host Only multiply within host in the presence of amino acids, nucleosides, and glucose

Bacillus anthracis l Bacillus anthracis is composed of: l. A capsule l A three part protein l Protective Antigen (PA) l Edema Factor (EF) l Lethal Factor (LF) l In the bacteria there is one chromosome and 2 plasmids (p. XO 1 and p. XO 2)

Bacillus anthracis Chromosome not involved in virulence l p. XO 1 codes for PA, EF, and LF l p. XO 2 codes for the capsule l

p. XO 2/Capsule p. XO 2 codes for the homopolymeric linked poly-D-glutamic acid capsule l Contains three genes: cap. BCA l 96, 231 bp, ~85 ORFs (16 sm. ones) l Antiphagocytic function l Is non-toxic by itself l Is important in the onset of the infection l Poorly understood l

p. XO 1 181, 654 nucleotides, ~143 ORFs l Codes for the three toxin genes which are transcribed simultaneously: l l pag. A (for PA) l cya (for EF) l lef (for LF) l Codes for regulatory proteins, from the atx. A gene, which causes ~10 x increase in transcription of all three toxins

PA l l Is non-toxic without EF and LF Coded from pag gene l l l A/T rich (69%) Cysteine-free 83 k. Da (735 -amino acid) Long, flat protein Function – to aid in the insertion of EF and LF toxins in the cytosol

PA Structure l Domain 1 (Green and Blue) ( residues 1 – 249, β-sandwich fold, l 2 Ca 2+, forms cleavage site during proteolytic activation l l Domain 2 (Magenta) ( Residues 250 – 487, β-barrel core l Forms membrane inserted channel /pore formation l l Domain 3 (Yellow) ( Residues 488 – 594, 4 -stranded mixed β-sheet, 4 small helices l Helps to oligomerize(? ) l l Domain 4 (Purple) l l Residues 595 – 735, β-sandwich Contains receptor binding domain Koehler, T. M. Anthrax. Springer. 2002.

PA Mechanism l. Mechanism: 1. PA 83 binds to cell receptor Furin or a furin-like molecule cleaves PA 83 between aa 164 -167 (Arg-Lys-Arg) to two fragments: PA 20 (released) and PA 63 (remains attached to the receptor) 2. http: //www. biotechjournal. com/Pathways/anthrax. htm

PA Mechanism (cont. ) 3. 4. PA 63/receptor complexes join to form a heptamer at reduced p. H The heptamer complex can then bind to one LF or EF molecule at each subunit http: //www. biotechjournal. com/Pathways/anthrax. htm

PA Mechanism (cont. ) 5. 6. 7. The heptamer is internalized via RME Acidification of the vesicle causes the heptamer to form a pore in the membrane EF and LF are released through the pore into the cytosol Koehler, T. M. Anthrax. Springer. 2002.

General Mechanism Koehler, T. M. Anthrax. Springer. 2002.

EF l Coded from cya gene l A/T rich (71%) l Cysteine-free 89 k. Da (767 -amino acid) l Mainly β strands l N-terminal ~250 aa bind to PA 63 l Remaining molecule possesses catalytic activity in substrate and calmodulinbinding subdomains l A-B toxin l

EF Function l l EF receptors are present on most cells EF is a calmodulin dependent adenylate cyclase l Therefore only functions in eukaryotic cells Catalyzes breakdown of ATP to c. AMP l ~200 fold increase Why Calmodulin? l 1% total cellular protein l Responsible for mediating Ca 2+ signaling in cells

EF Mechanism l Mechanism 1. 2. EF enters the cell by binding to PA (called Ed. Tx) Inside, EF binds to calmodulin at the Nterminal low Ca 2+ affinity site in a very unique way: – – – One half grips lower lobe while the other grasps the upper lobe and twists The method of binding forms a contact region of 6, 000 Å2 – an extremely large binding region! The conformational change experienced by both molecules inhibits Ca 2+ from binding and inhibits the reverse reaction of c. AMP to ATP

EF Mechanism (cont. ) 2. 3. The conformational change in EF allows it to act as an adenylate cyclase and bind to ATP c. AMP is formed due to the binding of EF to ATP Liddington, R. C. , A Molecular Full Nelson. Nature. 415: 373 -374, 2002.

l What EF happens as a result of this activity? l Causes edema by altering H 2 O and ion movement l Inhibits immune response against Bacillus anthracis http: //www. biocarta. com/pathfiles/h_anthrax. Pathway. asp

EF l Inhibits activity of TF NF-ĸB in macrophages l Strains with only EF and PA (LF-) are not likely to be lethal
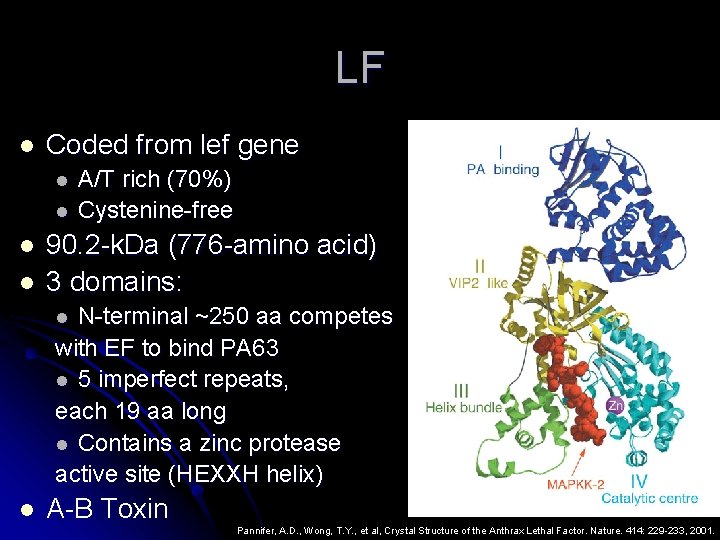
LF l Coded from lef gene l l A/T rich (70%) Cystenine-free 90. 2 -k. Da (776 -amino acid) 3 domains: N-terminal ~250 aa competes with EF to bind PA 63 l 5 imperfect repeats, each 19 aa long l Contains a zinc protease active site (HEXXH helix) l l A-B Toxin Pannifer, A. D. , Wong, T. Y. , et al, Crystal Structure of the Anthrax Lethal Factor. Nature. 414: 229 -233, 2001.

LF Function LF only binds to macrophages l LF is responsible for most of the toxicity l l Can l induce lysis in about 90 -120 minutes LF is a zinc dependent metalloprotease which cleaves MAPKK

LF Mechanism and Function LF is internalized via PA (called Le. Tx) l Mechanism and Result of MAPKK cleavage: l l LF cleaves between 7 -10 residues on the N -terminus of the MAPKKs at a pair of proline residues (separtated by 0 – 4 aa) l E. g. MAPKK 1, 2, 3, 4, 6 Duesbery, N. S. , Vande Woude, G. F. , Anthrax Toxins. Cellular and Molecular Life Sciences. 55: 1599 -1609, 1999.

LF Mechanism and Func. (cont) l This cleavage serves to inactivate the MAPKK l MAPKK cleavage leads to lysis of the macrophages, although the mechanism is unclear http: //www. biotechjournal. com/Pathways/anthrax. htm

LF Mechanism and Func. (cont) l Some MAPKK aid in the activation of TF NF- ĸB l This illustrates how EF and LF work together to promote macrophage lysis

LF Mechanism and Func. (cont) l At sublytic levels it stimulates production of IL-1β and TNFα l l l At lytic levels the mechanism is unclear, but it is possible that in the early stages they inhibit the immune response to allow the bacteria to proliferate, and at later stages it stimulates them to cause lysis of cells Causes leakage in cell membrane without ATP increased Ca 2+ conc. in cell Strains with only LF and PA (EF-) are likely to be lethal, although to a lesser degree

Review l EF l l Increased production of c. AMP Causes swelling Inhibits immune response l Allows for invasion! LF l l l Cleaves MAPKKs Inhibits immune response Destroys macrophages l Cells can’t fight off infection they Die!
Overall Mechanism Review Duesbery, N. S. , Vande Woude, G. F. , Anthrax Toxins. Cellular and Molecular Life Sciences. 55: 1599 -1609, 1999.

Clinical Features of B. Anthracis Anthrax can manifest itself in 3 forms: cutaneous, gastrointestinal, & inhalational (pulmonary). q The type contracted depends on the modes of transmission, which include: q § § Contact with infected tissues of dead animals (eg, butchering, preparing contaminated meat) cutaneous Consumption of contaminated undercooked meat gastrointestinal Contact with contaminated hair, wool, or hides (during processing) or contact with products made from them inhalational &/or gastrointestinal biological warfare mainly inhalational &/or gastrointestinal

Pathology Inhalational Anthrax: Ø Ø Ø Endospores are introduced into the body and alveoli via inhalation. (small size of spores permits this) Macrophages phagocytose and lyse endospores, and then travel to regional lymph nodes. Spores germinate to become vegetative cells within macrophages; they then leave macrophages and multiply within the lymphatic system. (1 -43 days) Bacteria enter the bloodstream and can lead to septic shock and toxemia with system wide edema and necrosis; hematogenous spread can lead to hemorrhagic meningitis. True pneumonia is rare w/ inhalational anthrax but focal, hemorrhagic, necrotizing pneumonic lesions may be observed. Major causes of death is compression of lungs and septic shock.

Pathology cont… Inhalational Anthrax: Ø Development of symptoms and pathogenesis occurs in 2 stages: 1) 2) Ø Ø Ø Exhibition of cold and flu like symptoms malaise, fever, prominent cough, nausea, vomiting, drenching sweats, dyspnea, chest pain, and headaches Develops rapidly (hours) and is characterized by acute dyspnea, subsequent cyanosis, pleural effusion, widened mediastinum, pulmonary edema, hemorrhagic symptoms of stage 1 become severe (>24 hrs and leads to death) ID 50 (infective dosage that will infect 50% of exposed) is 8, 000 -50, 000 spores. LD 50 (Lethal dosage that will kill 50% of exposed) is 2, 50055, 000 spores. Untreated 80% mortality rate

Pathology cont… Cutaneous Anthrax: Ø Ø Ø Endospores enter body through the skin usually via preexisting skin lesions, abrasions, and skin mucus membranes. Low-level germination and toxin production occurs at entrance site leading to localized necrosis with eschar formation and edema (massive in some cases) Eschar formation: • Day 2 papule becomes round ulcer • Day 3 plaque develops around edema site and central papule necrotizes, ulcerates, and forms a normally painless black eschar After 1 -2 weeks eschar falls off and leaves ulcer that can heal by week 3. • Ø Day 1 pruritic red papule appears at the initial site of infection
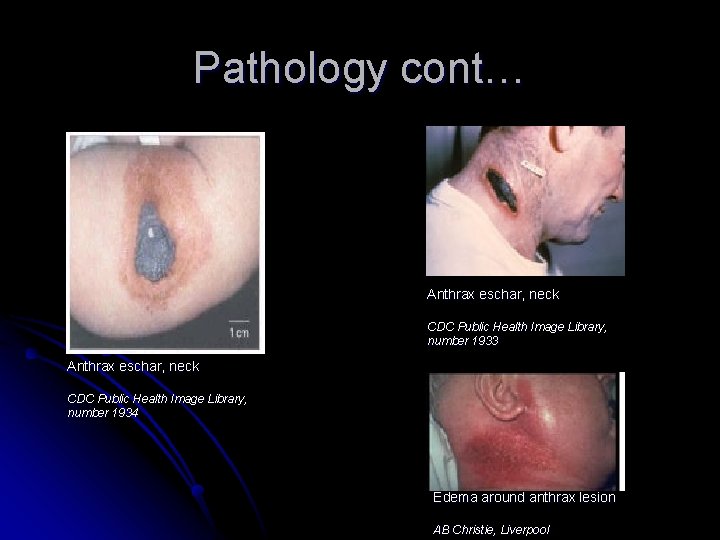
Pathology cont… Anthrax eschar, neck CDC Public Health Image Library, number 1933 Anthrax eschar, neck CDC Public Health Image Library, number 1934 Edema around anthrax lesion AB Christie, Liverpool

Pathology cont… Cutaneous Anthrax: Ø Endospores are often phagocytosed by macrophages and carried to local lymph nodes, which can result in painful lymphadenopathy and lymphangitis Ø Hematogenous spread with resultant toxemia can occur, with symptoms such as headache and fevers (up to 102 °F) although such spread is not common with appropriate antibiotic therapy Ø On very rare occasions system wide infection can occur and results in death within days. Ø ID 50 & LD 50 are unknown for cutaneous anthrax Ø Untreated mortality rate is 20% Ø With antibiotic treatment death is very rare.

Pathology cont… Gastrointestinal Anthrax: Ø Ø Ø Pathogenesis and statistics are unclear due to rarity of this form Vegetative cells from uncooked meat rather than spores (germination unnecessary) In oral pharyngeal anthrax, bacteria enter via the oral or pharyngeal mucosal ulcers can occur initially, followed by local/regional lymphadenopathy and localized edema. In abdominal anthrax, entrance occurs @ the ileum or cecum Intestinal lesions, regional lymphadenopathy, and edema of the bowel wall and ascites initially causes nausea, vomiting, and malaise progressing into bloody diarrhea, acute abdomen, or sepsis. Mortality may drop with antibiotic treatment (inconclusive)
Summary of Pathology

Diagnosis § § § Because of rarity of gastrointestinal cases diagnosis focus is limited. Onset of characteristic symptoms should signal patient to consult a physician immediately for antibiotic treatment. Cutaneous anthrax can be distinguished by 2 key features: painlessness of lesions and relatively large extent of associated edema (presence of eschars) Distinguishing features of inhalational anthrax: presence of widened mediastinum and pleural effusions on chest radiograph or CT (chest compound tomographic) scan with minimum evidence of pneumonia.

Diagnosis cont… Pulmonary anthrax chest X-ray showing widened mediastinum Inhalational Anthrax: § § Dept of Radiological Pathology Armed Forces Institute of Pathology Easier to diagnose when occurs in several suspicious cases than single case (or at high risk situations postal employee) chest x-ray showing a widened mediastinum, infiltrates, pleural effusion, etc. = likely anthrax infection CT scan showing hyperdense hilar and mediastinal nodes (white arrow, middle diagram) + extensive mediastinal edema = anthrax infection Molecular Biology tests only available at Laboratory Response Network (LRN) labs but highly reliable

Diagnosis cont… Distinguish Anthrax from Influenza-Like Illness (ILI): § § § Early symptoms (fever, chills, myalgias, fatigue, malaise, and nonproductive cough) are similar. ILI patients have rhinorrhea and this is uncommon in Anthrax patients have shortness of breath, an uncommon trait of ILI patients. Nausea and vomiting are more common in anthrax infection Abnormal chest radiographs and CT scans Anthrax infection

Post-exposure Prophylaxis: Treatment and Therapy Limited number of cases and data for treatment ¯ Must be administered before critical toxin levels are reached ¯ Early attempts (cutaneous) surgery, but fear of septicemia (early 20 th century) ¯ Pasteur, Jourbert, & Fortineau antibiotics (focus ¯ of modern treatment now) ¯ Early therapy utilized Penicillin G procaine but has been shifted to other antibiotics due to Penicillin-resistant strain appearance

Treatment and Therapy cont… Fluoroquinone class antibiotics are now used Ciprofloxacin in conjunction w/ Doxycycline as first line ¯ Usual dosages: ¯ Cutaneous: adults & pregnant women 500 mg Ciprofloxacin and 100 mg Doxycycline twice daily; children varies, based on age and weight (All for at least 60 days) Ø Inhalational & Gastrointestinal via IV: adults and pregnant women 400 mg Ciprofloxacin OR 100 mg Doxycycline every 12 hrs AND 2 additional antimicrobials (rifampin, vancomycin, penicilin, ampicillin, chloramphenicol, imipenem, clindamycin, & clarithromycin); children Ciprofloxacin OR Doxycycline varies, based on age & weight AND one or two of the previously mentioned antimicrobials. (Therapy switched to oral treatment when clinically appropriate and lasts for at least 60 days) Ø
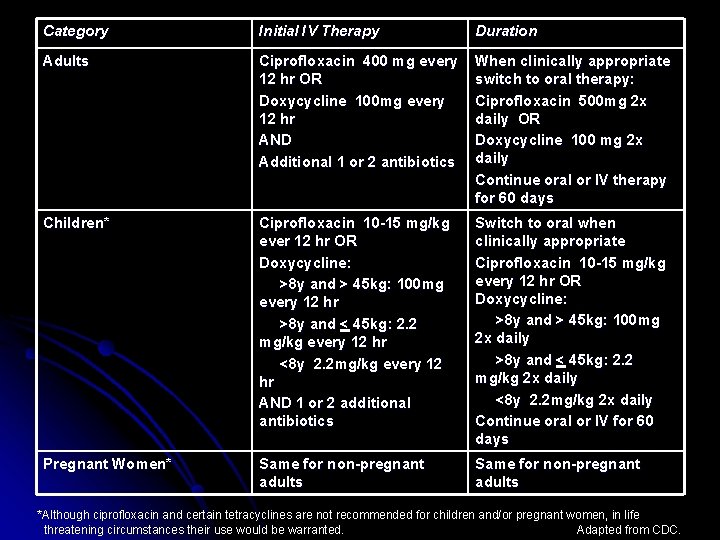
Category Initial IV Therapy Duration Adults Ciprofloxacin 400 mg every 12 hr OR Doxycycline 100 mg every 12 hr AND Additional 1 or 2 antibiotics When clinically appropriate switch to oral therapy: Ciprofloxacin 500 mg 2 x daily OR Doxycycline 100 mg 2 x daily Continue oral or IV therapy for 60 days Children* Ciprofloxacin 10 -15 mg/kg ever 12 hr OR Doxycycline: >8 y and > 45 kg: 100 mg every 12 hr >8 y and < 45 kg: 2. 2 mg/kg every 12 hr <8 y 2. 2 mg/kg every 12 hr AND 1 or 2 additional antibiotics Switch to oral when clinically appropriate Ciprofloxacin 10 -15 mg/kg every 12 hr OR Doxycycline: >8 y and > 45 kg: 100 mg 2 x daily >8 y and < 45 kg: 2. 2 mg/kg 2 x daily <8 y 2. 2 mg/kg 2 x daily Continue oral or IV for 60 days Pregnant Women* Same for non-pregnant adults *Although ciprofloxacin and certain tetracyclines are not recommended for children and/or pregnant women, in life threatening circumstances their use would be warranted. Adapted from CDC.

Pre-exposure Prophylaxis: Anthrax Vaccine ¯ 1881: first developed by Louis Pasteur ¯ 1937: vaccine modified to use attenuated non pathogenic strain of B. anthracis w/ dormant spores (other than former Soviet Union and associate countries, this vaccine is preferred over live attenuated vaccine) ¯ U. S. currently uses AVA (Anthrax Vaccine Adsorbed) aka Bio. Thrax™ licensed in 1970 and produced by Bioport Corp.

Anthrax Vaccine cont… ¯ ¯ ¯ ¯ AVA: made from cell-free infiltrates of microaerophilic cultures of an avirulent, nonencapsulated strain of B anthracis concentrated with PA (protective antigen) final no live or dead bacteria are in final product Final product contains: 1. 2 mg/m. L aluminum, added as aluminum hydroxide in 0. 85% sodium chloride (Al = adjuvant that increase immune response); & 25 mcg/m. L benzethonium chloride and 100 mcg/m. L formaldehyde, added as preservative How efficacious? : several animal studies & 1 controlled human trial induced immune response in 83% of adults vaccinated with one dose and 91% for 2 or more doses. ** Correlation between antibody titer and protection against infection not yet defined. Efficacy duration unknown, but animal tests suggest 1 -2 years after 2 doses. Vaccine not licensed for children due to lack of any pediatric testing, but likely to be safe based on results with other inactive vaccines. 1998: Department of Defense makes vaccination for anthrax mandatory for all troops (AVIP) over 400 quit or court marshaled due to fear of adverse side effects.

Anthrax Vaccine cont… ¯ Dosage: ª In U. S. : 6 doses of 0. 5 m. L at weeks 0, 2, & 4 AND months 6, 12, & 18, as well as annual boosters ª In U. K. : 6 doses of 0. 5 m. L at weeks 0, 3, 6, & 32 with annual boosters. ¯ Post-exposure vaccination: ª Vaccine can be used with antibiotics after inhalation exposure ª 3 -dose regime @ weeks 0, 2, & 4 in combination with at least 30 -days of antimicrobial therapy (w/out vaccine 60 -day antimicrobial therapy is needed) ª Vaccine not licensed for post-exposure therapy. Therefore, it is administered as an investigational new drug (IND) under FDA regulations.

Anthrax Vaccine cont… ¯ Adverse effects: ª ª USAMRIID data: inflammatory reaction greater than 5 cm in diameter around the site of injection found in about 2. 4 - 3. 9% of cases 1990 -200: Vaccine Adverse Event reporting System (VAERS) receives 1, 544 reports of adverse events after vaccination most frequent included: injection site hypersensitivity (334), injection site edema (283), injection site pain (247), headache (239), arthralgia (232), asthenia (215), and pruritus (212).

Anthrax Vaccine cont… ¯ Possible future Improvements on vaccine: ª Cloning the PA gene into organisms of low pathogenicity, such as B. subtilis, and creating recombinant vaccines ª Creating mutant strains of vaccines that utilize non-human aromatic compounds for virulence may establish better immunicity (minimizes self-cell recognition problems) w/out risk of deadly infection ª Experimenting with purified PA preparations with different combinations of adjuvant molecules (similar to AVA approach) 1) PA with monophosphoryl lipid A (MPL) no cold storage chain required and possibly more efficacious in conferring immunity 2) expression of the cereolysine AB gene in B. anthracis gives hemolytic properties to the bacteria; conferred immunity against both H-7 strain wildtype and H-7 AB recombinant when immunized by recombinant STI-1 strain containing the cereolysine AB gene 3) AVA may be more effective 100% immunity in 2 -dose immunized rhesus monkeys when compared to 100% mortality for non-immunized monkeys

“Safety First” Pre-Anthrax testing/handling Post-Anthrax handling: Decontamination

ANTHRAX AS A BIOLOGICAL WEAPON WHY USE ANTHRAX? l Relatively easy to acquire, in vitro growth (any lab media @ 37°C) and induction of sporulation is easy.

WHY USE ANTHRAX? l l Most likely agent for large-scale biological attack. Inhalational anthrax is the most potent of the three disease forms. ~80% mortality and with prompt treatment ~50% mortality. Anthrax spores can be easily spread in the environment. Ultimate reservoir is soil where they can survive for decades. Spores are stable to drying, heat, UV light, gamma radiation, and many disinfectants.

WHY USE ANTHRAX? – cont. l l l Spore size of 3 -6 μm suitable for aerosolization and ideal for human alveolar deposition. Early differentiation of inhalational anthrax from common infections is difficult. Anthrax spores decay at the rate of less than 0. 1 per cent a minute, which is very slow for an organism.

WHY USE ANTHRAX? – cont. l USDD reported that 3 defense employees with some technical skills but without expertise on bioweapons could manufacture a simulant of BA in less than a month and for $1 M. l Aerosol release would be difficult to establish, spores are odorless and invisible and have potential to travel many kms before dissipating.

Some data on offensive capacity of anthrax spores l l l (1960) Experiments w/ simulant B. globigii in NYC subway system: release of similar amount of B. anthracis during rush hour would result in 10, 000 deaths. In this test more than 1 M NY were exposed to the Bacillus. Aerial spray of BA along 100 km line under ideal meteorological cond could produce 50%lethality rates as far as 160 km downwind. US study near Johnston Atoll in SP reported that a plane “sprayed a 32 mile long line of agent that traveled for more than 60 miles before it lost its infectiousness”.

Some data on offensive capacity of anthrax spores –cont. l l l WHO estimated that 50 kg of BA released in a pop of 500, 000 would result in 95, 000 fatalities + 125, 000 incapacitated. 50 kg over urban pop of 5 M = 100, 000 deaths and 250, 000 sick. 100 kg = bw 130, 000 and 3 M deaths over an area size of W-DC. This lethality matches that of a hydrogen bomb.

Some data on offensive capacity of anthrax spores –cont. l l l CDC suggested a cost of $26. 2 B per 100, 000 persons exposed. Human LD 50 is ~2, 500 to 55, 000 inhaled BA spores but studies on primates suggest that as few as 1 -3 spores may be sufficient to cause infection. Little information of possible risks of direct contamination of food or water with anthrax spores.

So, who (possibly) has anthrax spores ready? l l l 1995 Iraq acknowledged producing and weaponizing BA to the UN special commission. Former Soviet Union also known to have a BA production program. Existence of offensive biological weapons programs in at least 13 -17 countries including Syria, Iran, Libya, China, SK, NK, Taiwan and Israel. (US Dep of Def)

Britain – Gruinard island 1942 l l l Britain conducted research during 1942 at Gruinard Island (Scotland). Sheep were taken to an open field, secured in wooden frames, and exposed to a bomb that scattered the spores. The sheep started dying three days later. An infected sheep's carcass washed ashore from the island led to the deaths of seven cattle, two horses, three cats and up to 50 sheep in a nearby village. They had probably been infected by a dog which had itself been infected by the carcass.

Britain – Gruinard island 1942 l l Island was so contaminated that the island was kept in quarantine for 48 years. In 1986 an English company was paid ₤ 500, 000 to decontaminate the 520 -acre island by soaking the ground in 280 tons of formaldehyde diluted in 2000 tons of seawater.

Sverdlovsk case – Former Soviet Union – April 2, 1979 l l l Odorless, colorless, unnoticeable aerosol was accidentally released from a secret Soviet biological weapons facility in Sverdlovsk. 96 people within a short distance fell sick; 66 died (68. 75%MR). All where inhalational anthrax, not single case was cutaneous. Animals found dead 50 km southeast of Sverdlovsk. New cases developed as late as 43 days after the presumed date of released. None during months or years afterward.

Sverdlovsk incident (1979) u 120 to >400 became ill, 75 died (at least, probably ~ 90) u. Soviet officials initially reported that outbreak was due to contaminated meat u. Autopsy findings of hemorrhagic mediastinitis u. Infection later linked to an aerosol leak from a nearby biologic weapons facility operated by the Soviet army Meselson et al, Science, 1994
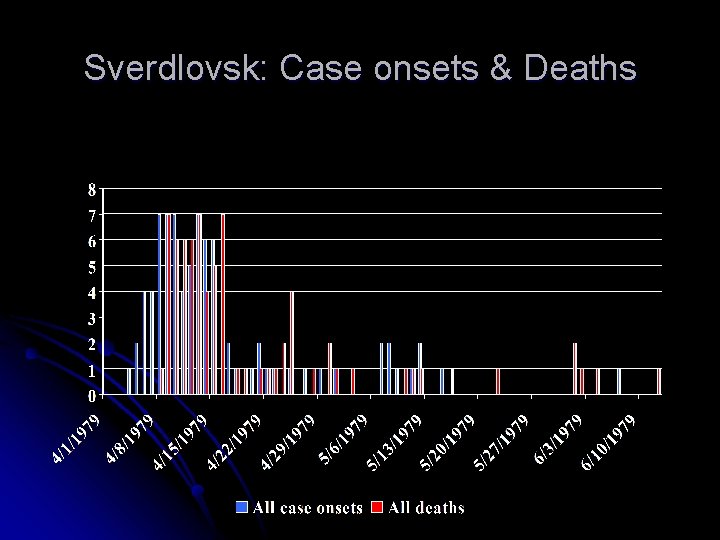
Sverdlovsk: Case onsets & Deaths

Gulf war - 1991 1990 Iraq had 50 R 400 bombs filled with anthrax. l 8, 500 L of anthrax. l Bombs were not used due to a supposed fear by Iraq that winds and meteorological conditions could make them also a target for their own bombs. l

Aum Shinrikyo failed attack 1995 l Terrorist group, Aum Shinrikyo, responsible for the release of Sarin in a Tokyo subway on 1995, dispersed aerosols of anthrax and botulism throughout Tokyo on at least 8 occasions. Attacks failed because strain released closely matched the Sterne 34 F 2 strain used for animal vaccination programs and is not a significant risk for humans.

US Anthrax attacks - 2001 l Robert Stevens, 63, photo editor at the supermarket tabloid The Sun, published by American Media Inc. , died on October 5, 2001 from contracting pulmonary anthrax from an envelope.

US Anthrax attacks – 2001 cont l l Oct, 2 – Nov, 20 22 cases of anthrax were confirmed: 11 inhalational anthrax and 11 (7 confirmed and 4 suspected) of cutaneous anthrax. 7 states along east coast of US: CT (1), FL (2), MD (3), NJ (5), NYC (8), PA (1), VA (2). Case-fatality ratio for inhalational anthrax: 45%. Mean duration bw exposure and onset of symptoms: 4. 5 days.

US Anthrax attacks – 2001 cont l l 4 BA + powdercontaining envelopes were recovered. 2 mailed around Trenton, NJ and postmarked Sep 18, 2001.

US Anthrax attacks – 2001 cont l l Envelopes processed at US PS Trenton Mail Processing Ctr and Morgan Central Postal Facility (NYC). Both facilities and at least 5 more affiliated with NJ facility had environmental samples positive for BA. No envelopes found in FL but BA was isolated from environmental sampling at the AMI building (were 2 cases from FL worked).

US Anthrax attacks – 2001 cont l l l Oct 9 envelopes sent from NJ to Washington DC (congress: Hart Building). Sen. Daschle’s letter was opened by office staff on Oct, 15. Prompt recognition of powder led to rapid initiation of post-exposure prophylaxis. Scientists estimate that the letter sent to Sen. Daschle originally contained about 2 grams of anthrax.

US Anthrax attacks – 2001 cont l l l Concentration - in the range of 1 trillion spores per gram meant that the letter could have contained 200 million times the average dose necessary to kill a person. 625 persons from Hart Building were tested (nasal swaps) and 28 found positive for BA. Environmental sampling of Building showed heavily contamination. Building was closed for decontamination on Oct 17, 2001.

US Anthrax attacks – 2001 cont l Letter to Sen. Leahy was in 1/280 barrels of unopened mail collected from Capitol Hill after Sen. Daschle’s letter was discovered.

US Anthrax attacks – 2001 cont l l l An estimated 32, 000 persons initiated antimicrobial prophylaxis but completion of the 60 day course was recommended for 10, 300 persons. Cross-contamination of mail occurred but risk was low considering the high amount of mail processed in NJ and NYC. 2 patients w/ inh. anthrax had no exposure to media or government facilities or possible sources of naturally occurring anthrax yet both were infected w/ BA isolates identical to outbreak strain.

US Anthrax attacks – 2001 cont l l All of the anthrax spores in the mail were found to be of an identical strain (Ames strain). This strain is one that the U. S. military used for study at USAMRIID and distributed to other government and university labs as well as to other governments including Great Britain. Decontamination of Hart Building took three months and cost was estimated at $23 M.

New Findings l l Secondary aerosolization of viable BA spores under common office activities was tested and found to be possible. Test was perform almost a month after the incident. Institute for Genomic Research (Rockville, MD) completed sequencing the anthrax bacillus in late November 2001, but has decided to delay its publication and access to research community for security reasons.

What to do in case of an attack? l l US military M 17 and M 40 gas masks provide excellent protection against 1 -5 μm particles ($324). Other articles include a portable mask ($79. 95) and anthrax test-kit ($24. 95) Use of pre-exposure and post-exposure antibiotics (US attack isolates were sensitive to fluoroquinolones, . Pre-exposure and post-exposure vaccination? ? Decontamination of any exposed spaces.

How to determine it is anthrax? Anthrax test-kit ($24. 95) l 1998 -Manz lab- new method for PCR: chemical amplification continuous-flow PCR on a chip. Problem: still slow and too much equipment for remote site use. l Burke lab: PCR product could be sent directly onto miniature gel: 30 min. l Phillip Belgrader: PCR in microfuge tubes: 7 min. l

References MICROBIOLOGY REFERENCES: 1. 2. 3. 4. 5. Drum, C. L. , Yan, S. Z. , et al. Structural Basis for the Activation of Anthrax Adenylyl Cyclase Exotoxin by Calmodium. Nature. 415: 396 -402, 2002. Duesbery, N. S. , Vande Woude, G. F. , Anthrax Toxins. Cellular and Molecular Life Sciences. 55: 1599 -1609, 1999. Koehler, T. M. Anthrax. Springer. 2002. Liddington, R. C. , A Molecular Full Nelson. Nature. 415: 373 -374, 2002. Mehta, A. , Zubay, G. , et al. Anthrax Chapter. 2003.

References (cont. ) 7. 8. 9. 10. 11. 12. Pannifer, A. D. , Wong, T. Y. , et al, Crystal Structure of the Anthrax Lethal Factor. Nature. 414: 229 -233, 2001. Patocka, J. , Splino, M. Anthrax Toxin Characterization. Acta Medica. 45: 3 -6, 2002. Turnbull, P. C. B. , Quinn, C. P. , et al. Bacillus anthracis and Other Bacillus Species. Academic Press. 2001. http: //www. biocarta. com/pathfiles/h_anthrax. Pathw ay. asp http: //www. biotechjournal. com/Pathways/anthrax. h tm http: //www. es. dis. titech. ac. jp/~park/anthrax. html

References (cont. ) History & Clinical features references: 1. 2. 3. 4. 5. Brook, Itzhak. The Prophylaxis and Treatment of Anthrax. International Journal of Antimicrobial Agents. 20: 320 -325, 2002. Dixon, T. C. , M. Meselson, J. Guillemin, et al. Anthrax. New England Journal of Medicine. 341: 815 -826, 1999. Franz, D. R. , P. B. Jahrling, A. M. Friedlander, et al. Clinical Recognition and Management of Patients Exposed to Biological Warfare Agents. JAMA. 278 (5): 399 -411. 1997. Greenfield, Ronald A. , et al. Bacterial Pathogens as Biological Weapons and Agents of Bioterrorism. The American Journal of The Medical Sciences. Volume 323, Number 6. June 2002. Inglesby, MD, et. al. Anthrax as a Biological Weapon: Medical and Public Health Management. JAMA. Vol 281, No. 18. May 12, 1999.

References (cont. ) 6. 7. 8. 9. 10. 11. IOM. The Anthrax Vaccine: Is it safe? Does it work? Washington, D. C. : National Academy Press. March 2002. Peters, C. J. , D. M. Hartley. Anthrax Inhalation and Lethal Human Infection. Lancet. 359 (9307): 710 -711. Turnbull, P. C. B. , Quinn, C. P. , et al. Bacillus anthracis and Other Bacillus Species. Academic Press. 2001. Center for Infectious Disease Research & Policy: http: //www. cidrap. umn. edu/cidrap/content/bt/anthrax/biofacts/an thraxfactsheet. html Nas, Meryl M. D. Anthrax Epizootic in Zimbabwe, 19781980: Due to Deliberate Spread? : http: //www. anthraxvaccine. org/zimbabwe. html US Army Center for Health Promotion and Preventative Medicine: http: //chppmwww. apgea. army. mil/Homeland. Security/anthraxvideo/default. as p
- Slides: 91