Baby with Hypoglycaemia Dr Sanjeev Deshpande Consultant Neonatologist

Baby with Hypoglycaemia Dr Sanjeev Deshpande Consultant Neonatologist Shrewsbury & Telford Hospital NHS Trust

What would you get from this session? • When should we investigate for metabolic/hormonal disorders in neonatal hypoglycaemia? • Which investigations should we perform? • How do we interpret the results of these investigations?

Neonatal hypoglycaemia: learning from claims (Hawdon JM, et al. Arch Dis Child Fetal Neonatal Ed 2017) • 25 claims for neonatal hypoglycaemia • Total value of claims – £ 162 166 677 • Median (range) of settlement – £ 6. 3 m (2. 46 – 12. 64 m)

Metabolic/hormonal disorders causing hypoglycaemia are more likely to present during neonatal period …. . yet, most instances of neonatal hypoglycaemia are NOT due to metabolic/hormonal disorders

Disturbances of glycaemic status in the neonate are most often due to failure of metabolic adaptation

Metabolic Challenge to the neonate • Change in nutrition – continuous to intermittent – intravenous to enteral – glucose to fat • Immature enzymes & hormonal regulation • Concurrent cardio-respiratory adapation

How does normal metabolic adaptation take place?
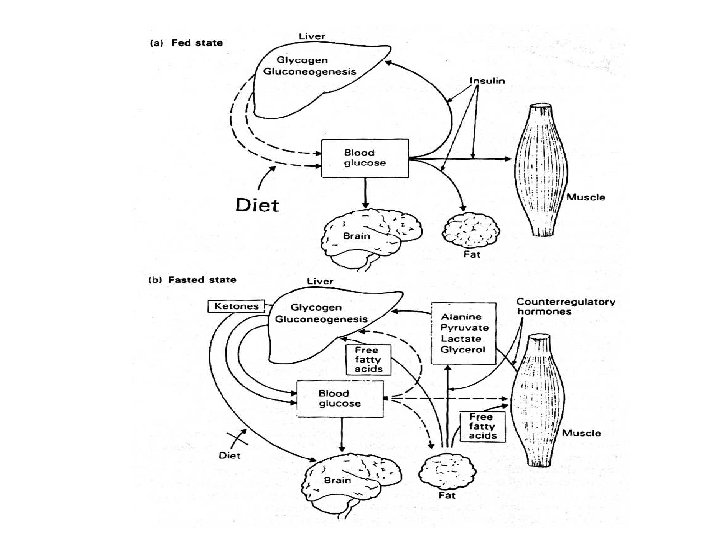

Metabolic adaptation at birth glycogenolysis gluconeogenesis lipolysis ketogenesis
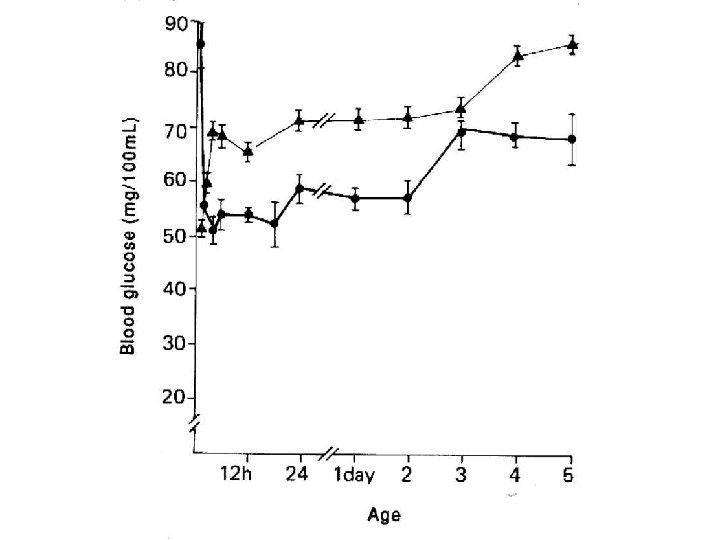
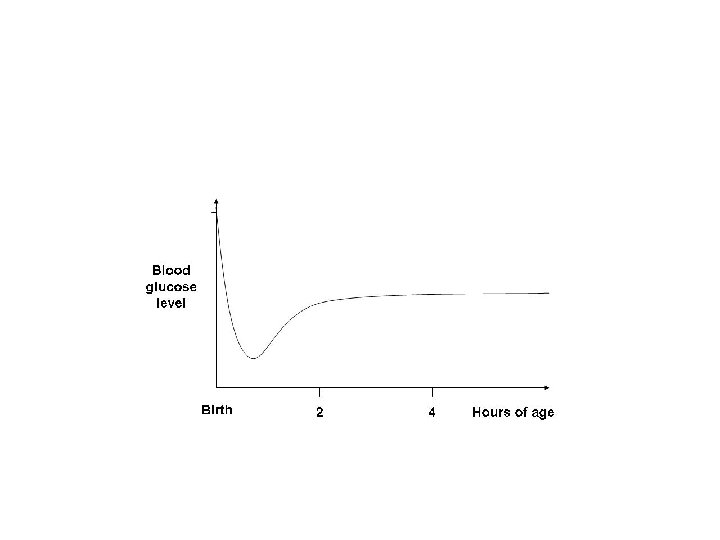

Blood glucose concentrations in fetal and early neonatal life • Fetal blood glucose • Birth • Plasma glucose falls to ~3 mmol/l by 1 -2 h • Even without feeding, the levels rise to ~3. 6 mmol/l by 3 -4 h of age • Remain ~3. 1 -3. 6 mmol/l until day 3 -4 Transitional Neonatal Hypoglycaemia

Glucose Free Fatty acids Ketone bodies Term AGA pre- Children feed after 24 h fast 3. 1 (0. 3) 5. 01 (0. 12) 0. 40 (0. 01) 0. 77 (0. 01) 0. 04 (0. 01) 0. 32 (0. 01) Transitional Hypoglycaemia • Hypoketotic • Incompletely suppressed insulin Glucose set-point for suppression of insulin is reduced Hawdon JM, et al Arch Dis Child 1992

Should we routinely screen for transitional neonatal hypoglycaemia? Transitional neonatal hypoglycaemia affects outcome Interventions for Transitional Neonatal Hypoglycaemia alter that outcome.

Does Transitional Neonatal Hypoglycaemia affect neurodevelopmental outcome?

Does intervention for Transitional Neonatal Hypoglycaemia alter neurodevelopmental outcome? • Sugar Baby Study (Harris DL. J Pediatr 2016) • Children with Hypoglycaemia and their Later Development (CHYLD) Studies

Healthy, asymptomatic, well grown term infants
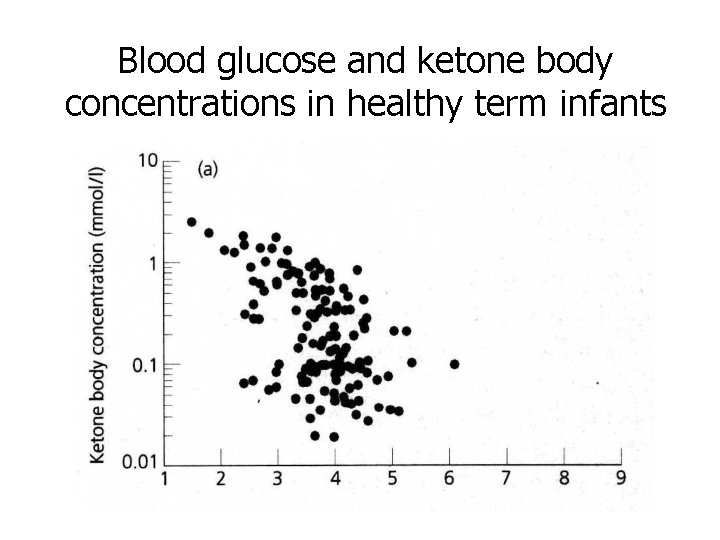
Blood glucose and ketone body concentrations in healthy term infants

Neonatal glucose economy • Demand for glucose – 6 -12 g/kg/d • Dietary supply of glucose – 200 ml colostrum/day = 3 g/kg/d • endogenous supply of glucose – glycogenolysis – gluconeogenesis - 2 mg/kg/min (1/3 rd) - 4 mg/kg/min (2/3 rd) • glucose recycling 33% • alanine 5 -10% • glycerol 5 -7%
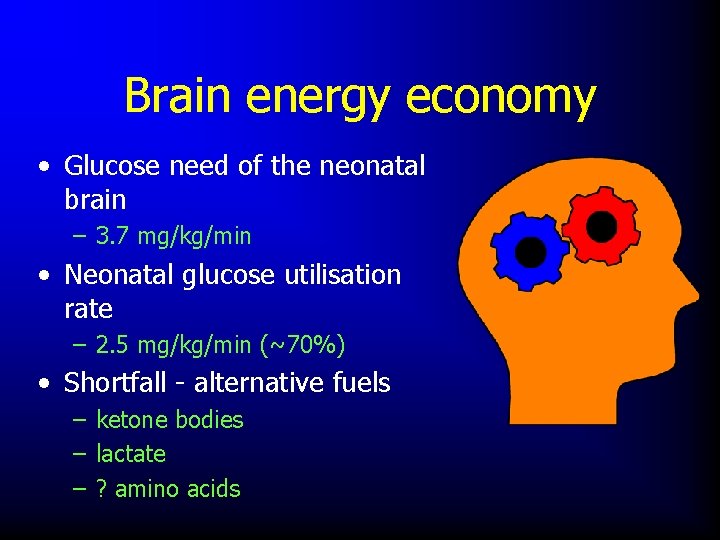
Brain energy economy • Glucose need of the neonatal brain – 3. 7 mg/kg/min • Neonatal glucose utilisation rate – 2. 5 mg/kg/min (~70%) • Shortfall - alternative fuels – ketone bodies – lactate – ? amino acids

Healthy, asymptomatic term infants • Do not check blood glucose during first 3 hours • Low blood glucose concentrations common – always accompanied by vigorous ketogenic response • Breast feeding promotes ketogenesis • No need for monitoring of blood glucose concentrations unless – symptomatic – ‘at risk’

At-risk term infants • Maternal – Diabetes – Preeclampsia, PIH – drug treatment • terbutaline • labetalol • ritodrine – intrapartum glucose administration • Neonatal – – Perinatal asphyxia Infection Polycythaemia Rh haemolytic disease – Hypothermia

Preterm infants
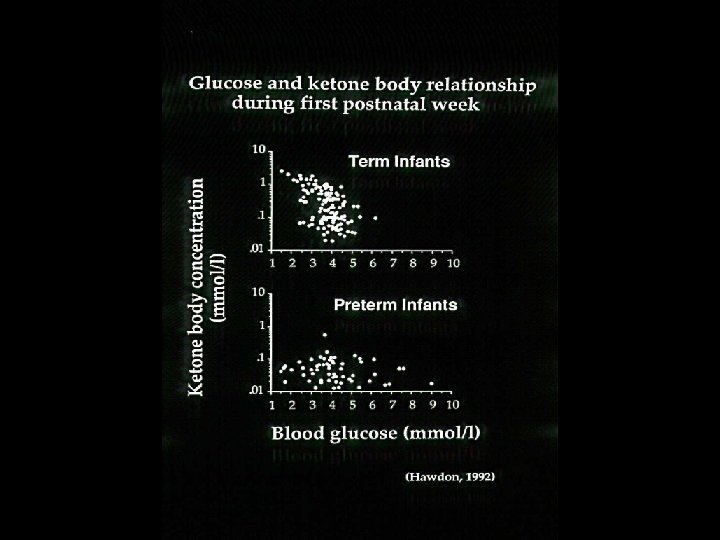

Evaluation of neonatal glycaemic status requires accurate and precise monitoring of blood glucose concentration
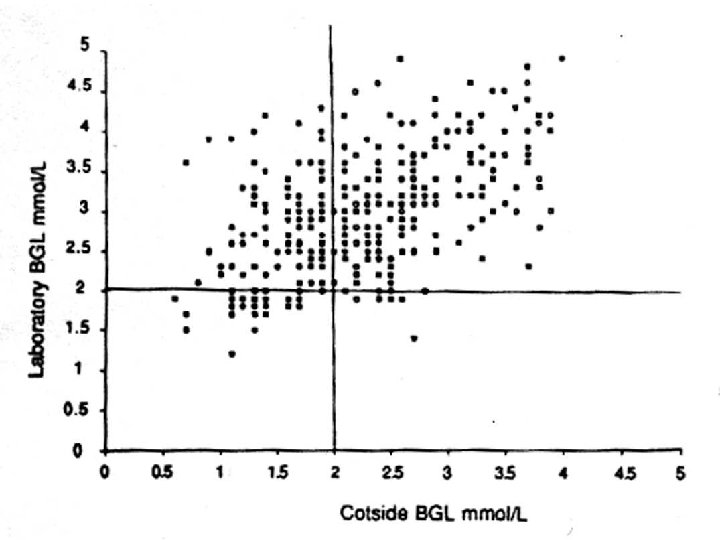

Despite glucose reagent strips being contraindicated for use in neonates, they were used in 35% of cases CEMACH - pregnancy in women with type 1 and type 2 diabetes (2002 -03)

Measurement of blood glucose • Large units – Glucose oxidase on whole blood • eg YSI 2300 stat plus – Glucose oxidase on plasma • eg Kodak Ektachem • Smaller Units – Multianalyte blood gas analysers • eg AVL/Roche Omni, Radiometer • Laboratory – eg Hexokinase – Isotope dilution GC-MS anaysis

Knowledge of glycaemic status requires. . . • • • blood glucose concentration availability of alternative fuels cerebral blood flow changes regional differences in brain underlying pathology

Significant hypoglycaemia is not, and can never be defined by a single number that is applicable universally at all times

Operational thresholds A concentration of blood glucose at which clinicians should consider intervention, based on currently available best evidence – pragmatic intervention thresholds – NOT thresholds for adverse outcomes

Operational thresholds Blood Glucose level Babies with abnormal clinical signs ≥ 2. 6 mmol/l At-risk infant with no clinical signs >2. 0 mmol/l Hyperinsulinaemic infant >3. 3 mmol/l

When should we investigate neonatal hypoglycaemia? • Well grown term infant – Severe hypoglycaemia – Seizures, coma • At-risk infants – – – Persistent or recurrent Resistant (>10 mg/kg/min glucose) Associated with neurological symptoms Associated with acidaemia Family history of SIDS, developmental delay

Approach to a baby with hypoglycaemia • History – – Gestation, birth weight Maternal diabetes, Pre-ecclampsia Maternal drug treatment (e. g. b-blockers) Perinatal stress (IUGR, perinatal asphyxia, infection) – Family history – Relationship to food, illness

Approach to a baby with hypoglycaemia Examination • Midline defects (cleft lip/palate, micropenis) o Hypopituitarism • Hepatomegaly o Glycogen storage diseases • Macroglossia, ear crease, omphalocele o Beckwith-Wiedemann syndrome • Weight loss, pigmentation o Congenital adrenal hyperplasia

Biochemical measurements in hypoglycaemia – Critical sample • Intermediary metabolites – Glucose – Lactate – 3 b-hydroxybutyrate – Free fatty acids • Acylcarnitine profile • Galactosemia screen • Hormones – Insulin – Cortisol – Growth hormone • Urine – Organic acids – Reducing substances – Ketones

Appropriate response to hypoglycaemia • Suppressed insulin • Increased cortisol, catecholamines, glucagon, and growth hormone • Increased free fatty acids • Increased ketone bodies • Normal/low blood lactate
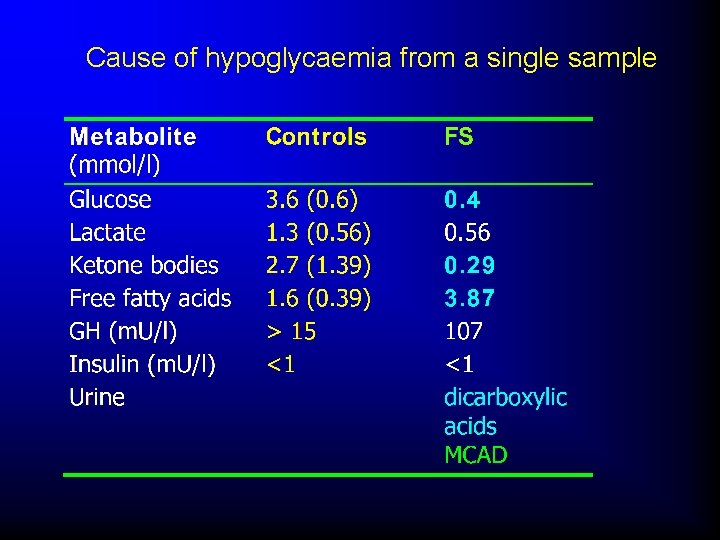
Cause of hypoglycaemia from a single sample

Management of fatty acid oxidation disorders • Regular feeding • Avoidance of fasting – <6 hrs 0 -3 months – 8 -10 hrs 3 -18 months – child-specific thereafter • Management during stress – illness, surgery, trauma • Use of uncooked corn-starch • ? L-carnitine
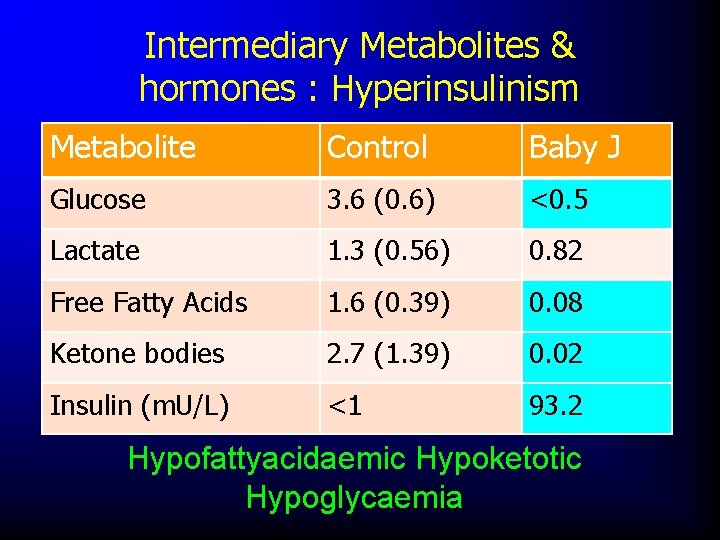
Intermediary Metabolites & hormones : Hyperinsulinism Metabolite Control Baby J Glucose 3. 6 (0. 6) <0. 5 Lactate 1. 3 (0. 56) 0. 82 Free Fatty Acids 1. 6 (0. 39) 0. 08 Ketone bodies 2. 7 (1. 39) 0. 02 Insulin (m. U/L) <1 93. 2 Hypofattyacidaemic Hypoketotic Hypoglycaemia

The diagnostic criteria for hyperinsulinism • Glucose requirement > 10 mg/kg/min to maintain blood glucose >2. 6 -3 mmol/l • Laboratory blood glucose <2. 6 mmol/l • Detectable insulin at point of hypoglycaemia with raised C-peptide • Inappropriately low blood fatty acids and ketone body concentrations at hypoglycaemia • Glycaemic response to glucagon when hypoglycaemic

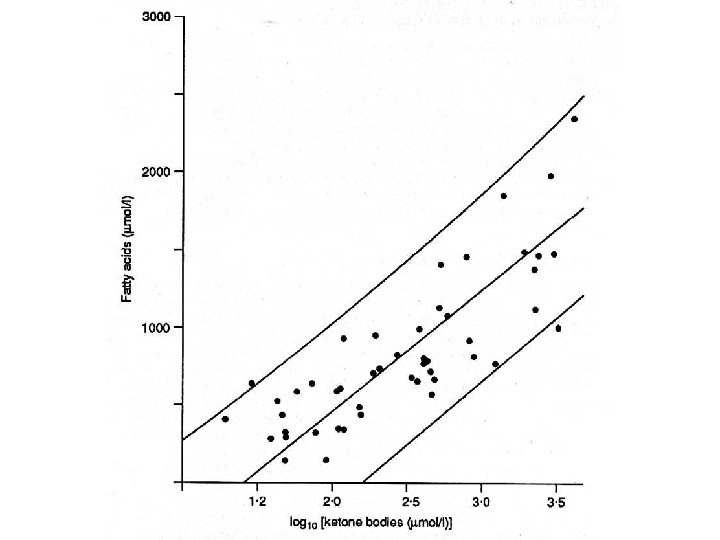
Cause of hypoglycaemia from a single sample

IW • Healthy parents • Second pregnancy – Low risk; booked for midwifery-led care – Unremarkable anomaly scan @20 weeks • Spotaneous labour @41 weeks – Meconium stained liqour – Normal vaginal delivery – Regular respirations @ 2 min
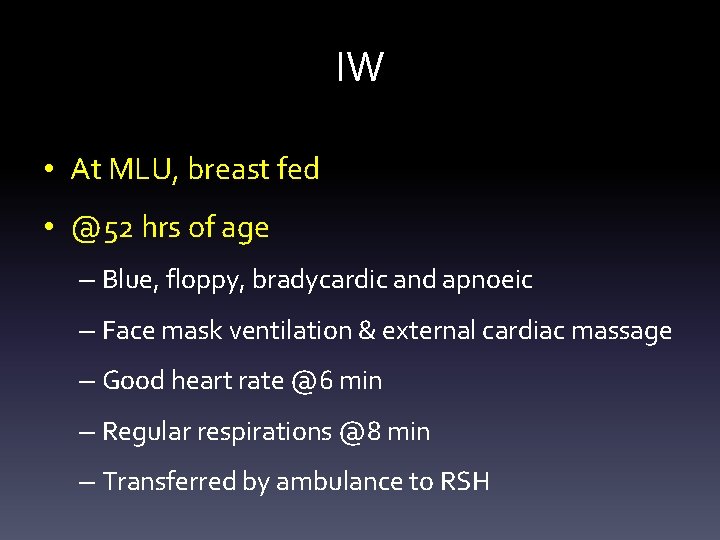
IW • At MLU, breast fed • @52 hrs of age – Blue, floppy, bradycardic and apnoeic – Face mask ventilation & external cardiac massage – Good heart rate @6 min – Regular respirations @8 min – Transferred by ambulance to RSH
IW • BM @15 min - 1 mmol/l – Given a feed • On arrival at RSH – Cardiorespiratory stable – Quite and hypotonic – Blood glucose <1. 1 mmol/l • Stable thereafter on iv glucose infusion and later oral feeds
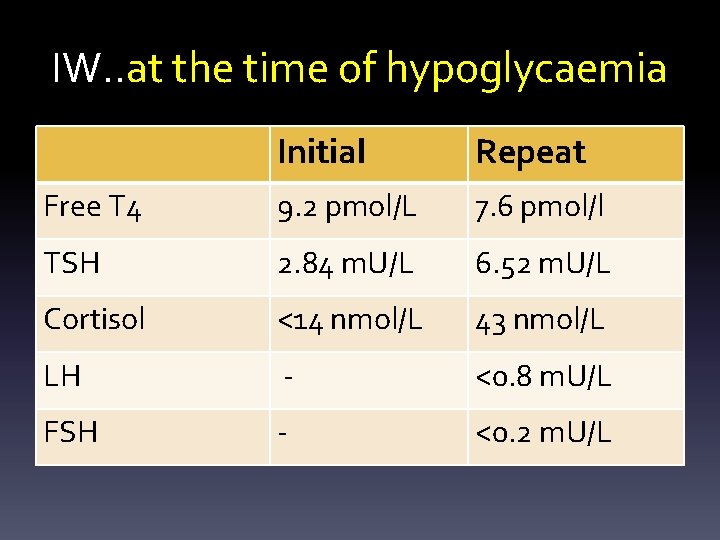
IW. . at the time of hypoglycaemia Initial Repeat Free T 4 9. 2 pmol/L 7. 6 pmol/l TSH 2. 84 m. U/L 6. 52 m. U/L Cortisol <14 nmol/L 43 nmol/L LH - <0. 8 m. U/L FSH - <0. 2 m. U/L
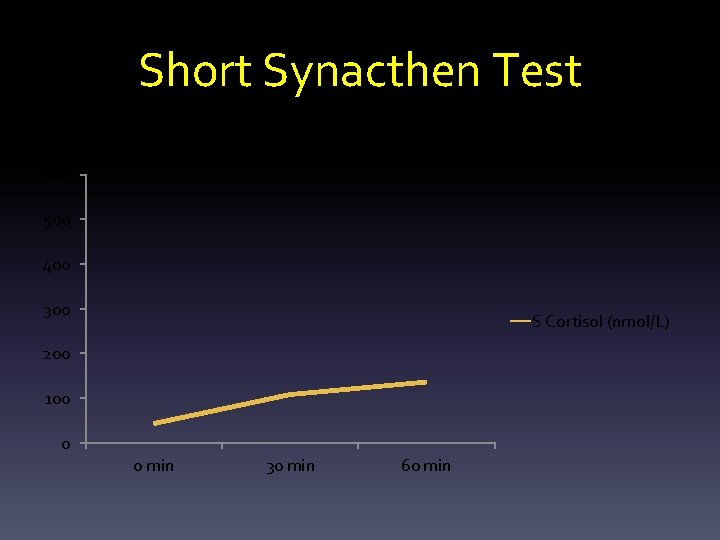
Short Synacthen Test S Cortisol (nmol/L) 600 500 400 300 S Cortisol (nmol/L) 200 100 0 0 min 30 min 60 min
IW - MRI • Hypoplastic Pituitary gland • Ectopic posterior pituitary – Pituitary stalk inturruption syndrome • Normal optic nerves, chiasm • Intact corpus callosum and septum pellucidum

IW - Management • Hydrocortisone - 10 microg/m 2/d • Levothyroxine – 25 microg/day

Congenital hypopituitarism • Isolated GH deficiency • ± other pituitary hormone deficiencies • Combined Pituitary Hormone deficiency – 1: 8000 • Septo-optic dysplasia (SOD) – 1: 50, 000 • Optic Nerve Hypoplasia • Midline brain abnormalities – Absence of Septum pellucidum and/or Corpus callosum • Hypopituitarism

Why should congenital hypothyroidism be detected in neonatal period? Neurological injury Collapse/death • Hypoglycaemia • Hypoadrenalism • Hypothyroidism • Hypoglycaemia • Hyperammonaemia
- Slides: 53