B Heterocyclic compound with fused rings 1 2
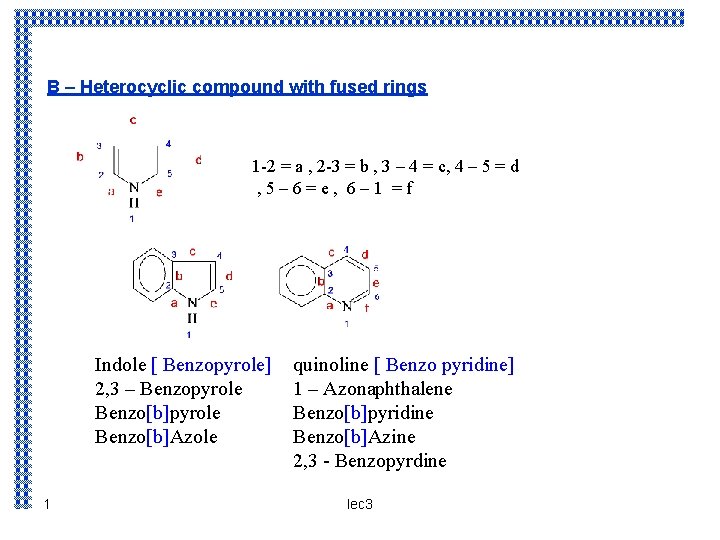
B – Heterocyclic compound with fused rings 1 -2 = a , 2 -3 = b , 3 – 4 = c, 4 – 5 = d , 5– 6=e, 6– 1 =f Indole [ Benzopyrole] 2, 3 – Benzopyrole Benzo[b]Azole 1 quinoline [ Benzo pyridine] 1 – Azonaphthalene Benzo[b]pyridine Benzo[b]Azine 2, 3 - Benzopyrdine lec 3
![Isoquinoline 2– Azo. Naphthalene 3 -4 -Benzopyrdine Benzo[c ] pyridine Benzo[c ] Azine 2 Isoquinoline 2– Azo. Naphthalene 3 -4 -Benzopyrdine Benzo[c ] pyridine Benzo[c ] Azine 2](http://slidetodoc.com/presentation_image/fab0f540501141a5eda9c1983b16d0e7/image-2.jpg)
Isoquinoline 2– Azo. Naphthalene 3 -4 -Benzopyrdine Benzo[c ] pyridine Benzo[c ] Azine 2 Benzo thiophene 2, 3 -Benzo thiophene Benzo[b ] thiol lec 3
![2, 3 - benzofuran Benzo[b ] oxole Benzo 2, 3 - oxole Thiazol[5, 4 2, 3 - benzofuran Benzo[b ] oxole Benzo 2, 3 - oxole Thiazol[5, 4](http://slidetodoc.com/presentation_image/fab0f540501141a5eda9c1983b16d0e7/image-3.jpg)
2, 3 - benzofuran Benzo[b ] oxole Benzo 2, 3 - oxole Thiazol[5, 4 -b]pyridine Thiazol[4, 5 -b]pyridine Pyrole [2, 3 -d]pyrimidine Azole[2, 3 -d]1, 3 -diazine 3 lec 3
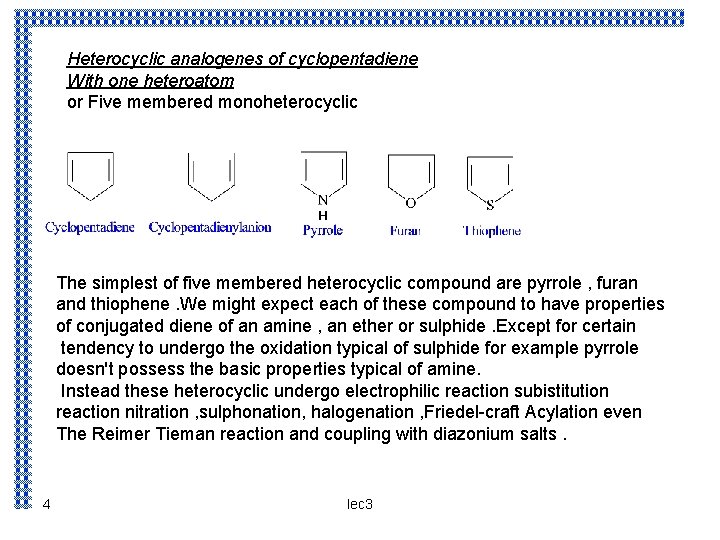
Heterocyclic analogenes of cyclopentadiene With one heteroatom or Five membered monoheterocyclic The simplest of five membered heterocyclic compound are pyrrole , furan and thiophene. We might expect each of these compound to have properties of conjugated diene of an amine , an ether or sulphide. Except for certain tendency to undergo the oxidation typical of sulphide for example pyrrole doesn't possess the basic properties typical of amine. Instead these heterocyclic undergo electrophilic reaction subistitution reaction nitration , sulphonation, halogenation , Friedel-craft Acylation even The Reimer Tieman reaction and coupling with diazonium salts. 4 lec 3

Heat of combustion indicate resonance stabilization to the extent 22 – 28 kcal/mol less than the resonance energy of benzene (36 kcal/mol)but much greater than of most conjugated diene (about 3 kcal/mol). On this basis pyrrole , furan and thiophene must be considered aromatic The orbital picture for example for pyrrole each atom of the ring carbon or nitrogen is held by σ bonds the atom uses three sp 2 orbitals each carbon has left one electrons occupy p orbitals overlap of the p orbitals give rise to π clouds one above and one below the plane of the ring. The π cloud contain a total of six electron the aromatic sextet extra pair on nitrogen which response for the basisty involve in the π cloud and its not available for sharing with acid in contrast to most amine , there for , pyrrole is a weak base it thus pyrrole is better represented by 5 lec 3
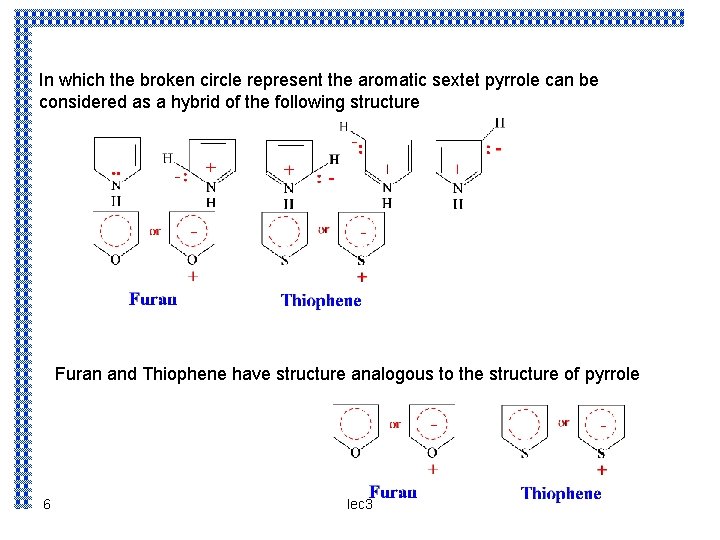
In which the broken circle represent the aromatic sextet pyrrole can be considered as a hybrid of the following structure Furan and Thiophene have structure analogous to the structure of pyrrole 6 lec 3
- Slides: 6