B Grobty Mineralogy and Cristallography Mineralogy study of

B. Grobéty

Mineralogy and Cristallography Mineralogy: study of naturally occuring crystalline solids = minerals Cristallography: study of crystalline solids Both deal with: Gem quality grossular garnet - macroscopic geometry (morphology) of crystals microscopic build up of crystals chemical properties of crystal physical properties of crystals Polyhedral representation of the garnet structure Optical absorption spectrum of a grossular garnet

References The course will be presented mainly with help of powerpoint presentations, available under Moodle. Textbooks A. Putnis An Introduction to Mineral Sciences Cambridge University Press, 1992 ISBN: 0 -521 -42947 -1 C. Klein Mineral Science John Wiley and Sons Ltd, 2002 ISBN: 0 -471 -25177 -1 Moodle adress: http: //moodle. unifr. ch

Course organisation Teacher: Bernard Grobéty Office No. 3. 304 Tel. 8936 e-mail: bernard. grobety@unifr. ch Schedule and location: Lecture: Monday 1515 - 1700 Auditoire 2ème étage

Why is Mineralogy/Crystallography important? - Earth, interior planets: 60 - 70 Vol% solid, consisting of minerals - Processes affecting the Earth’s surface and interior depend on the properties of the solid constituents. - 70 - 80 elements used by mankind are extracted from minerals - The properties of minerals (and all other materials) depend, among others, on the microscopic build-up (= crystal structures) of the constituent phases. - Important technologies used by modern society is based on specific poperties of solids: - Information technology: optical properties of ruby, piezoelectric properties of quartz (laser telephone, electronic devices). - Computer: electrical properties of silicium and siliciumdioxide (= quartz) (processsors), magnetic properties of ferrites (memory) - Energy production: radioactive properties of uraniumoxide - Biology: structure and function of biological molecules (proteins, enzymes etc. ) are obtained from “solidified” versions of these molecules

Content of part I (WS) 1. Introduction 2. Symmetry - Symmetry in daily life and art - Symmetry operations - Point groups 3. 4. 5. Crystal morphology - Morphology, habitus - Crystal axes - Miller indices - Forms - Zones 6. 7. Stereographic Projection 8. 9. Point groups 10. - Monoclinic point groups 11. - Tetragonal point groups 7. - Cubic point groups 8. 9. 10. 6. Ionic bonds - Atom models - Ionization - Ionic bonding 10. Energetics of minerals - Enthalpy - Entropy - Gibbs Free energy 7. Paulings model - Coordination - Pauling rules - Halite, fluorite and perovskite structures 11. Point defects and diffusion - Point defects - Diffusion - Solid solutions 8. Periodic patterns - Translations, unit cell - Glide mirrors - Plane groups 9. notation andalusite Space groups - Glide mirrors - Screw axes - Space groups - Hermann-Maugin - Halite and structure

Content of part II (SS) 12. Analytical methods - Diffraction techniques - Spectroscopic methods - Electronmicroscopy 13. Color of minerals - Color - Crystal field splitting - Color of ruby - Electron transfer 14. Radioactivity - Alpha decay - Spontaneous fission - Metamictization - Fission tracks - Decay law 15. Line and planar defects - Edge and screw dislocation - Line defects and deformation - Twinning - Stacking faults - Antiphase boundaries 16. Polytypism and polysomatism - Phyllosilicate structures - Inosilicate structures - Mica polytypes - Biopyriboles 17. Mineral reactions - Displacive phase transitions - Reconstructive phase transitions - Order - disorder transitions - Exsolution - Heterogeneous reactions

marble thin section: marble is made up of calcite crystals metamorphic, monomineralic rock Minerals are the "elementary" components of rocks igneous, polymineralic rock granite thin section: granite is made of different minerals

Mineralogy/Crystallography: Related Fields Mineralogical/crystallographic information is needed in: Petrography Petrology Experimental Petrology Sedimentpetrography Geology Sedimentology Stratigraphy Paleontology Solid state chemistry, physics Spectroscopy Protein crystallography Material science: Metallurgy, Ceramics Special fields in Mineralogy: Ore Mineralogy Biomineralogy Mineralsystematics Mineralphysics: Optical Mineralogy Mineral Spectroscopy High pressure and temperature (mineral) physics Technical (Applied) Mineralogy Refractory Materials
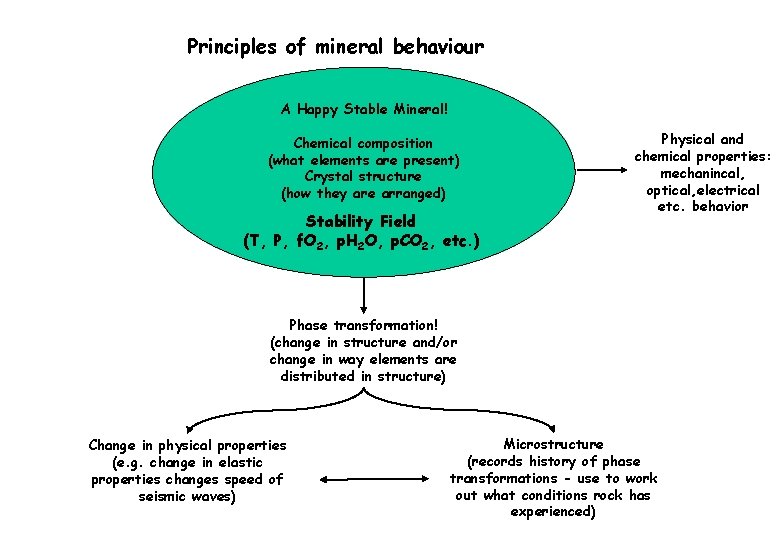
Principles of mineral behaviour A Happy Stable Mineral! Chemical composition (what elements are present) Crystal structure (how they are arranged) Stability Field (T, P, f. O 2, p. H 2 O, p. CO 2, etc. ) Physical and chemical properties: mechanincal, optical, electrical etc. behavior Phase transformation! (change in structure and/or change in way elements are distributed in structure) Change in physical properties (e. g. change in elastic properties changes speed of seismic waves) Microstructure (records history of phase transformations - use to work out what conditions rock has experienced)
- Slides: 10