B Anthracene glycosides They are a group of

B- Anthracene glycosides - They are a group of glycosides in which the aglycones are anthracene derivatives. - They are widely distributed in many families as Rubiaceae, Leguminosae and Euphorbiaceae in addition to some fungi. - They are derivatives of anthraquinone, anthranol and oxanthrone. - They are coloured substances and many of them are used technically as dyes. - The basic structure of these compounds is 9, 10 -anthraquinone, and their difference lie in the arrangement of the attached susbstituents.
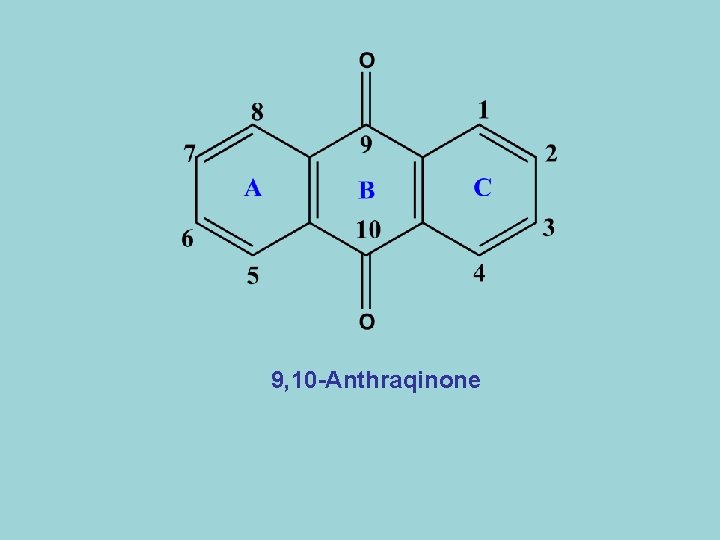
9, 10 -Anthraqinone
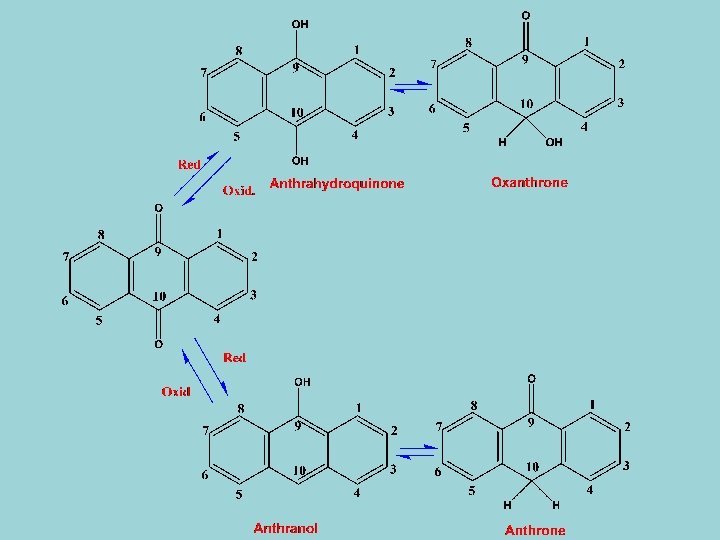


- The proportions of the various oxidation stages vary. For example, in fresh leaves of Cassia senna or C. Angustifolia anthrones dominate, but on drying during preparation of crude drug dianthrones are formed. - The glycosides may be present in -O- or -C- form. - Anthraquinones turn intense red colour in alkali, a reaction that can be used for their quantitative determination (Bornträger reaction). Anthrones and anthranols do not show this reaction. - Presence of anthranols in crude drugs can be easily detected since it gives a strong blue flourecence with alkali.

Medicinal uses of anthraquinone drugs - Anthraquinone drugs are used as purgative or laxative. - In practice, pure compounds are rarely prescribed, but mostly simple extracts of the crude drugs. - The sugar moiety in the glycosides present increases solubility and facilitates transport to the site of action. - The aglycone is the active part of the molecule, but it is mainly the anthrone form that is effective, - Laxation increased as the number of OH group increases.

- In the colon, bacteria hydrolyse the glycosides and reduce the liberated aglycones to anthrones which act directly on the large intestine to stimulate peristalsis. - Thus, crude drugs containing little or no anthrones nevertheless have laxative activity. - However, drugs rich in anthrones as frangula bark, have too strong an effect and must therfore be stored for a period of time to allow oxidation to the corresponding anthraquinones to takes place. . - A frequent or long-term use has been associated with increased risk of intestinal tumours, so the use of anthraquinones drugs should be restricted to short –term treatment.

Test for identity 1 - Bornträger´s test A- Free anthraquinone - Boil the powder with water, then filter and cool the filtrate. - To the filtrate add an equal volume of ether and shake. - To the ethereal layer add an equal volume of dilute ammonia solution. - The aqueous layer becomes pale rose-red, pink or violet. B- Test for combined anthraquinone - Boil the powder with alcoholic KOH then filter and cool the filtrate. - The filtrate is rendered acidic with HCl and extracted with ether. - To the ethereal layer add an equal volume of dilute ammonia solution. - The aqueous layer becomes rose-red colour.
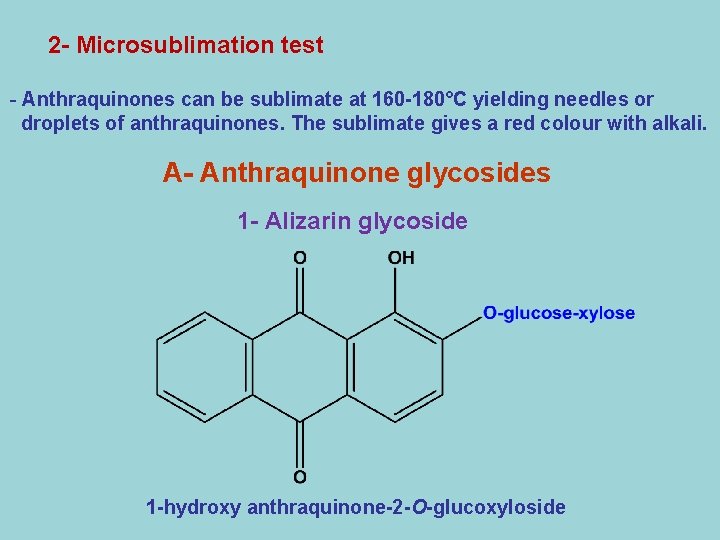
2 - Microsublimation test - Anthraquinones can be sublimate at 160 -180°C yielding needles or droplets of anthraquinones. The sublimate gives a red colour with alkali. A- Anthraquinone glycosides 1 - Alizarin glycoside 1 -hydroxy anthraquinone-2 -O-glucoxyloside
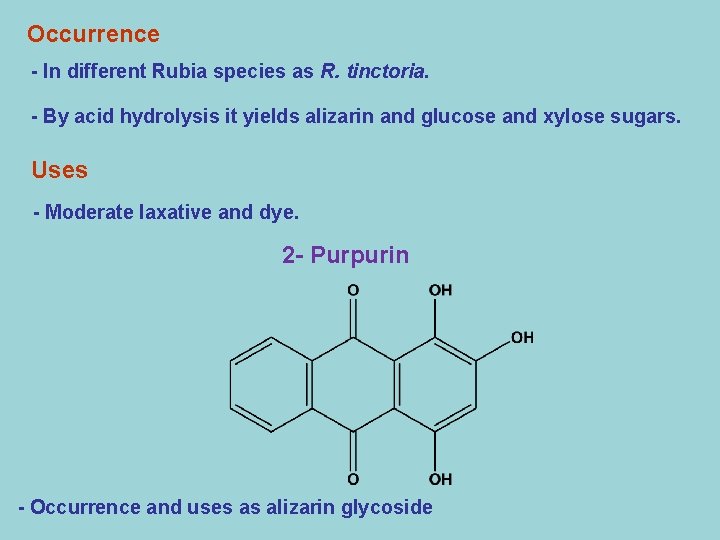
Occurrence - In different Rubia species as R. tinctoria. - By acid hydrolysis it yields alizarin and glucose and xylose sugars. Uses - Moderate laxative and dye. 2 - Purpurin - Occurrence and uses as alizarin glycoside
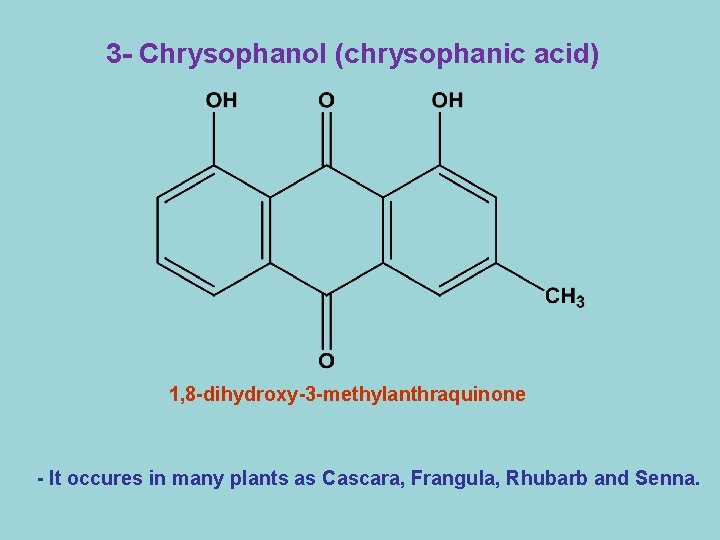
3 - Chrysophanol (chrysophanic acid) 1, 8 -dihydroxy-3 -methylanthraquinone - It occures in many plants as Cascara, Frangula, Rhubarb and Senna.
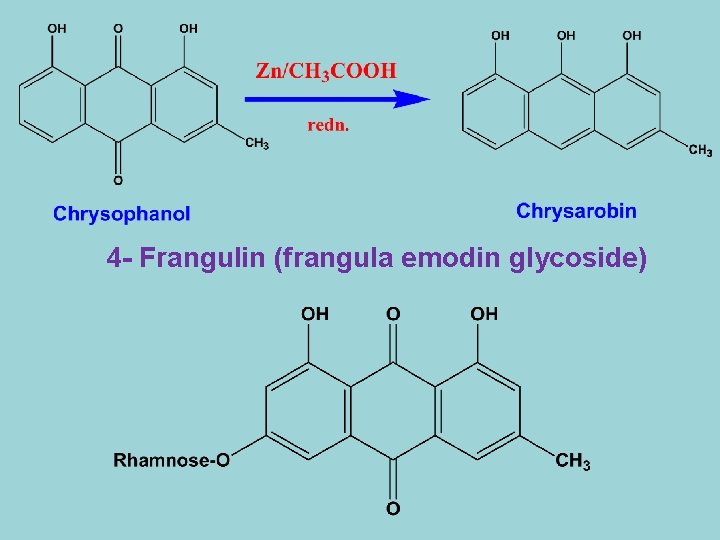
4 - Frangulin (frangula emodin glycoside)
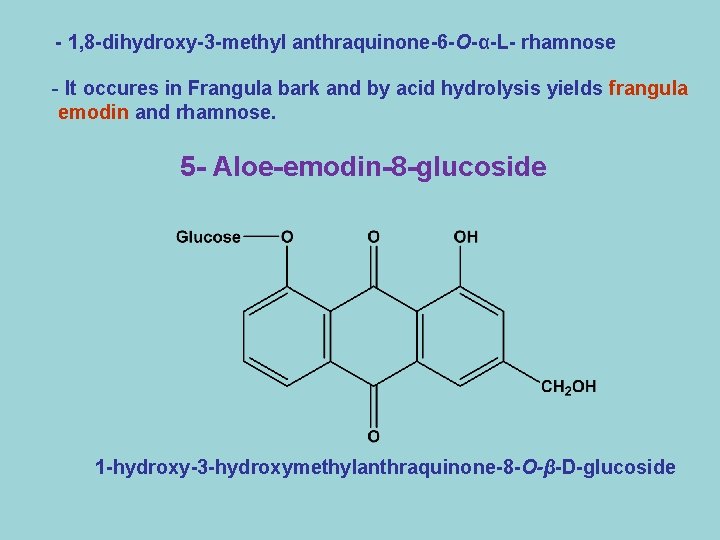
- 1, 8 -dihydroxy-3 -methyl anthraquinone-6 -O-α-L- rhamnose - It occures in Frangula bark and by acid hydrolysis yields frangula emodin and rhamnose. 5 - Aloe-emodin-8 -glucoside 1 -hydroxy-3 -hydroxymethylanthraquinone-8 -O-β-D-glucoside
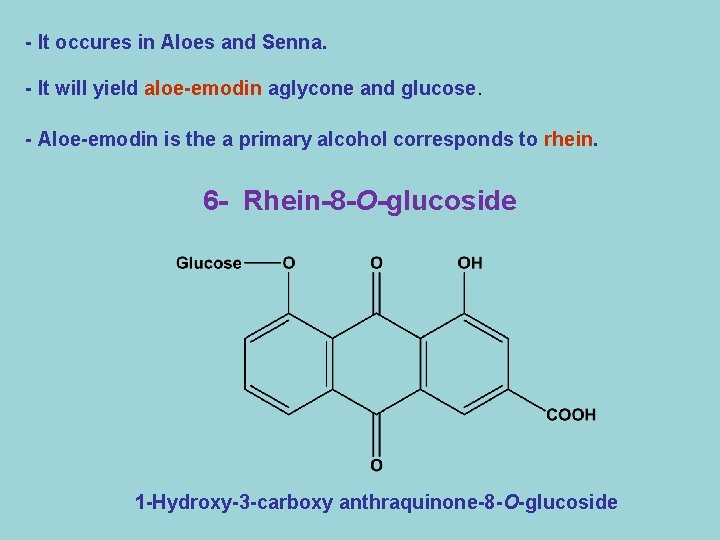
- It occures in Aloes and Senna. - It will yield aloe-emodin aglycone and glucose. - Aloe-emodin is the a primary alcohol corresponds to rhein. 6 - Rhein-8 -O-glucoside 1 -Hydroxy-3 -carboxy anthraquinone-8 -O-glucoside

- It occures in Rhubarb and Senna. - Hydrolysis by acid hydrolysis yields glucose and rhein. B- Anthranols and Anthrones 1 - Cascaroside A-D - These compounds are the main constituents of cascara bark. - These compounds have a glucose residue linked directly to one of the carbon atom, in other word they are C-glycoside. - There are four cascarosides, A-D forming two stereoisomeric pairs which differ in their aglycones.
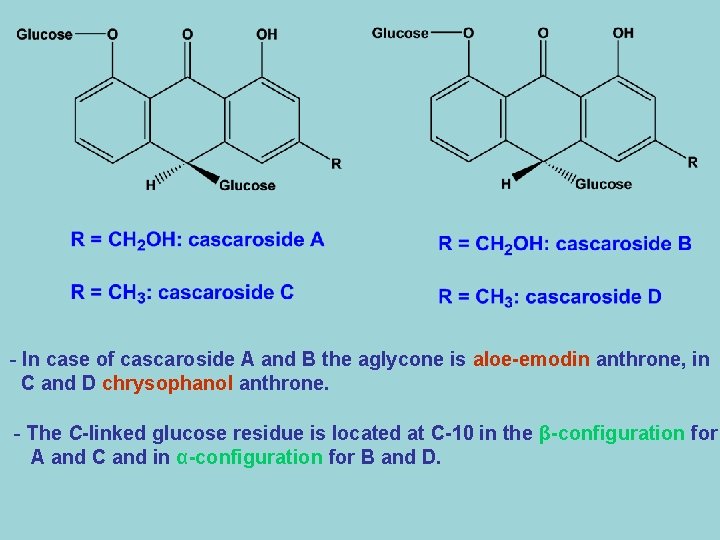
- In case of cascaroside A and B the aglycone is aloe-emodin anthrone, in C and D chrysophanol anthrone. - The C-linked glucose residue is located at C-10 in the β-configuration for A and C and in α-configuration for B and D.
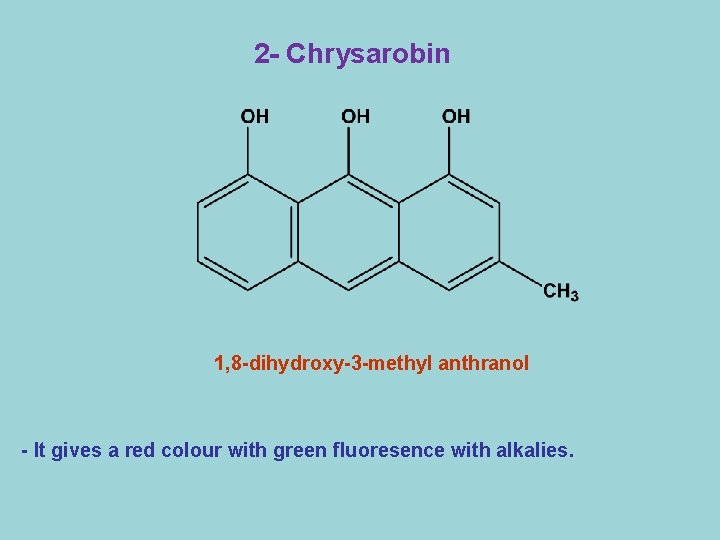
2 - Chrysarobin 1, 8 -dihydroxy-3 -methyl anthranol - It gives a red colour with green fluoresence with alkalies.
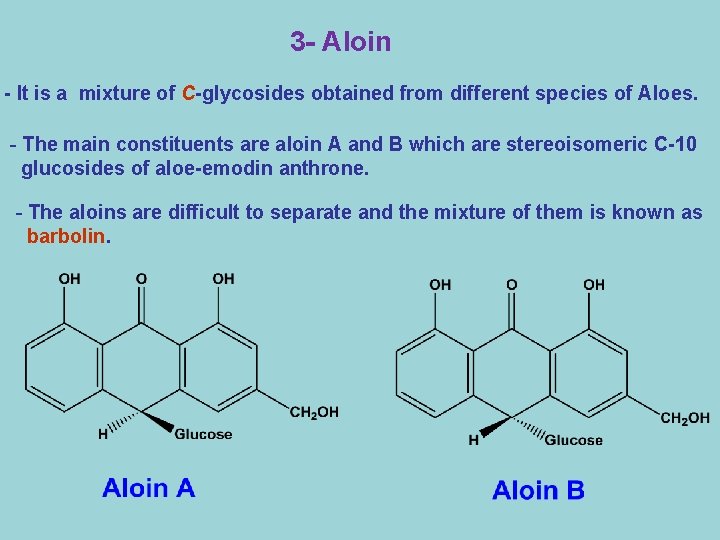
3 - Aloin - It is a mixture of C-glycosides obtained from different species of Aloes. - The main constituents are aloin A and B which are stereoisomeric C-10 glucosides of aloe-emodin anthrone. - The aloins are difficult to separate and the mixture of them is known as barbolin.

- For the preparation of aloes, the leaves are cut near the base and placed on a watertight bed, on which the juice draining from the leaves is collected. - After about 6 hours all the juice has emerged and it is then evaporated on an open air. - Aloe gel is used in sun burns, against radiation, also it is used in many hair and skin preparations.

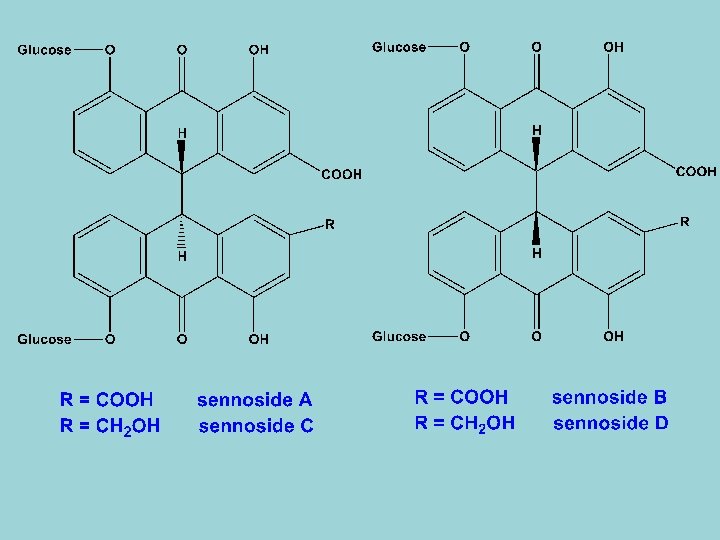
C- Dianthrone glycoside 1 - Sennoside A-D - Senna leaf contains a complex mixture of anthraquinone glycosides, the aglycones being monomeric anthraquinones as well as dianthrones. - The main dianthrones glycosides are sennoside A and B, the aglycones of which are stereoisomeric rhein dianthrones. - Smaller quantities of sennosides C and D are also found in senna leaf. They are glycosides of a stereoisomeric pair of heterodianthrones based on rhein anthrone and aloe-emodin anthrone. - Sennidins A, B, C and D are the aglycones of sennosides A, B, C and D respectively.
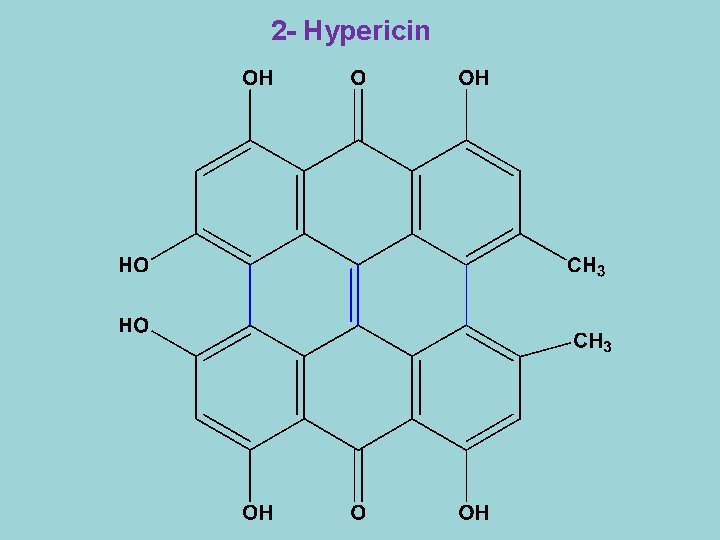
2 - Hypericin

- Hypericin is a red coloured, dimeric anthraquinone derivative which is present in the leaves and flowers of hypericum perforatum. - Hypericin is a photosensitizing agent and causes the so-called light sickness in animals feeding on Hypericum. - Only animals with white or light-coloured coats are subjected to this disease. - It has anti-viral activity against retroviruses such as influenza and Herpes simplex virus. The compound seems to act directly on the virus. - Hypericum is also known for the antidepressant activities of the ethanol extracts of the crude drug. The active constituents are not known, hypericin might be involved but also falvonoids have been suggested to be responsible.
- Slides: 23