Azithromycin During Tobramycin Therapy in Cystic Fibrosis Exacerbations

Azithromycin During Tobramycin Therapy in Cystic Fibrosis Exacerbations: To Hold or Not to Hold Evan Steere, Xing Song, Ph. D, Maren Wennberg, MS, Joel Mermis, MD, Deepika Polineni, MD, Russ Waitman, Ph. D

Background - Epidemiology Cystic fibrosis (CF) affects approximately 80, 000 people worldwide and more than 30, 000 people in the US In the US, about 1, 000 individuals are diagnosed every year Incidence varies widely across different races ~1 in 3, 000 -4, 000 live births among white persons ~1 in 4, 000 -10, 000 among Latin Americans ~1 in 15, 000 -20, 000 among African Americans N Engl J Med. 2019; 381(19): 1809 -1819. Pediatr Clin North Am. 2016; 63(4): 567 -584. 2018 Patient Registry Annual Data Report. 2018.

Background - Pathophysiology CF is an autosomal recessive gene disorder that results in decreased expression of the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) protein CFTR is expressed in many epithelial and blood cells CFTR functions primarily as an ion transport channel but is involved in many other cellular processes CF is characterized primarily as a disease of the lung, although numerous other organ systems are affected Gastrointestinal tract Endocrine system Reproductive system Lancet. 2009; 373(9678): 1891 -1904.

Background - Exacerbations Bacteria commonly colonize the respiratory tract of patients with CF Pseudomonas aeruginosa (PSAE) Staphylococcus aureus Can periodically cause exacerbations leading to acute decreases in lung function Often require inpatient admission, intravenous (IV) antibiotics, and supportive care Lung function is monitored by measuring the forced expiratory volume of air in 1 second (FEV 1) Pediatr Clin North Am. 2016; 63(4): 567 -584. 2018 Patient Registry Annual Data Report. 2018.

Background - P. aeruginosa Treatment of acute infection involves IV antibiotics selected based on current and previous resistance data Tobramycin (aminoglycoside) has good coverage of PSAE strains Azithromycin (macrolide) is recommended for use in patients with chronic colonization and is recommended to be considered in patients without Improves lung function and reduces exacerbation frequency Am J Respir Crit Care Med. 2013; 187(7): 680 -689.

Background - Tobramycin-Azithromycin Antagonism In vitro data suggests that exposure of azithromycin to PSAE causes resistance to tobramycin Upregulation of mex. X and mex. Y efflux genes Clinical data suggests that lung function improvement could be reduced in patients using tobramycin and oral azithromycin AIR-CF 2 trial – INH tobramycin + PO azithromycin Klinglel et al. – IV tobramycin + PO azithromycin The objective of this study was to determine if holding inpatient azithromycin in CF patients treated with IV tobramycin for a pulmonary exacerbation resulted in improved lung function during their hospital stay J Cyst Fibros. 2017; 16(3): 358 -366. Ann Am Thorac Soc. 2019; 16(7): 861 -867.

Methods - Design & Patients Single-center, retrospective cohort study Utilized EHR and Cystic Fibrosis Foundation Patient Registry (CFFPR) data Inclusion criteria: Documented ICD-9 or ICD-10 code for CF Inpatient hospital stay with IV tobramycin order Outpatient medication order for azithromycin at least 30 days prior to hospitalization Culture positive PSAE lung infection Exclusion criteria: Did not have FEV 1 measurement at beginning and end of hospitalization

Methods - Variables & Outcomes Independent variable: Inpatient azithromycin exposure Determined from Medication Administration Record (MAR) data All variables collected were included in the model initially Only those correlated with statistical significance were kept in the final model Outcome: Absolute change in FEV 1 during inpatient stay Beginning and end of stay FEV 1 only available for patients enrolled in CFFPR

Methods - Data Processing & Statistical Analysis All data was pre-processed in Oracle SQL Developer All statistical analyses were done in R Studio Case characteristics were calculated across groups as means for continuous variables and proportions for categorical variables Compared using 95% Confidence Intervals Outcomes were analyzed via linear regression
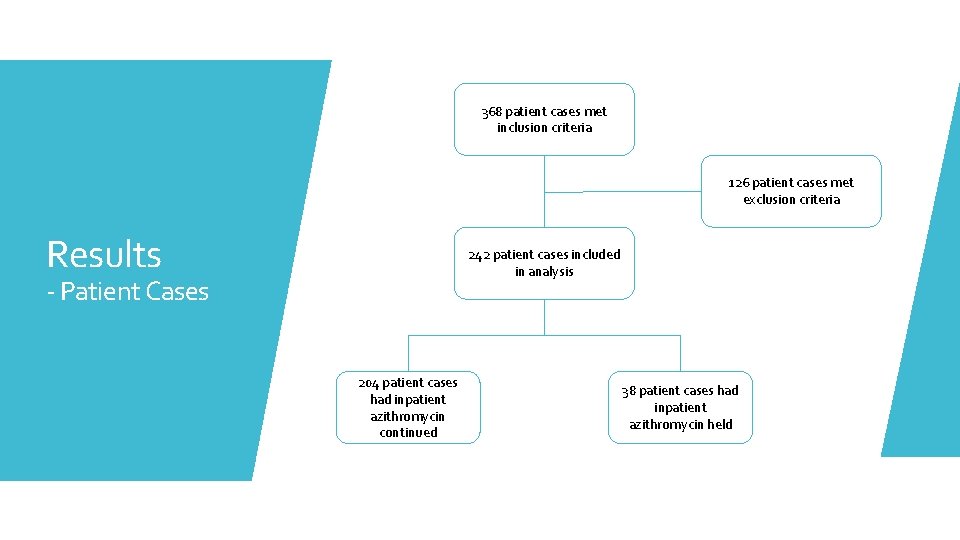
368 patient cases met inclusion criteria 126 patient cases met exclusion criteria Results 242 patient cases included in analysis - Patient Cases 204 patient cases had inpatient azithromycin continued 38 patient cases had inpatient azithromycin held
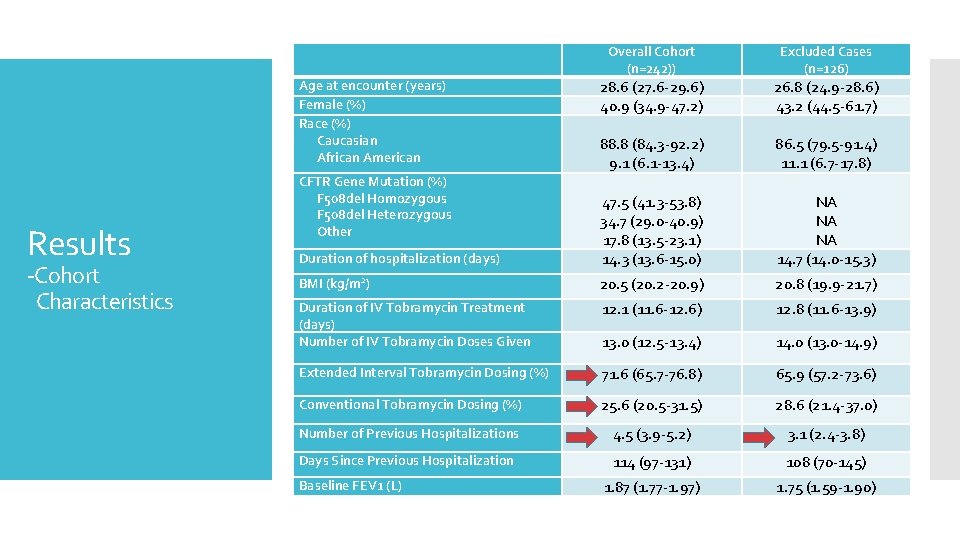
-Cohort Characteristics Excluded Cases (n=126) Duration of hospitalization (days) 28. 6 (27. 6 -29. 6) 40. 9 (34. 9 -47. 2) 88. 8 (84. 3 -92. 2) 9. 1 (6. 1 -13. 4) 47. 5 (41. 3 -53. 8) 34. 7 (29. 0 -40. 9) 17. 8 (13. 5 -23. 1) 14. 3 (13. 6 -15. 0) 26. 8 (24. 9 -28. 6) 43. 2 (44. 5 -61. 7) 86. 5 (79. 5 -91. 4) 11. 1 (6. 7 -17. 8) NA NA NA 14. 7 (14. 0 -15. 3) BMI (kg/m 2) 20. 5 (20. 2 -20. 9) 20. 8 (19. 9 -21. 7) Duration of IV Tobramycin Treatment (days) Number of IV Tobramycin Doses Given 12. 1 (11. 6 -12. 6) 12. 8 (11. 6 -13. 9) 13. 0 (12. 5 -13. 4) 14. 0 (13. 0 -14. 9) Extended Interval Tobramycin Dosing (%) 71. 6 (65. 7 -76. 8) 65. 9 (57. 2 -73. 6) Conventional Tobramycin Dosing (%) 25. 6 (20. 5 -31. 5) 28. 6 (21. 4 -37. 0) Number of Previous Hospitalizations 4. 5 (3. 9 -5. 2) 3. 1 (2. 4 -3. 8) Days Since Previous Hospitalization 114 (97 -131) 108 (70 -145) 1. 87 (1. 77 -1. 97) 1. 75 (1. 59 -1. 90) Age at encounter (years) Female (%) Race (%) Caucasian African American Results Overall Cohort (n=242)) CFTR Gene Mutation (%) F 508 del Homozygous F 508 del Heterozygous Other Baseline FEV 1 (L)
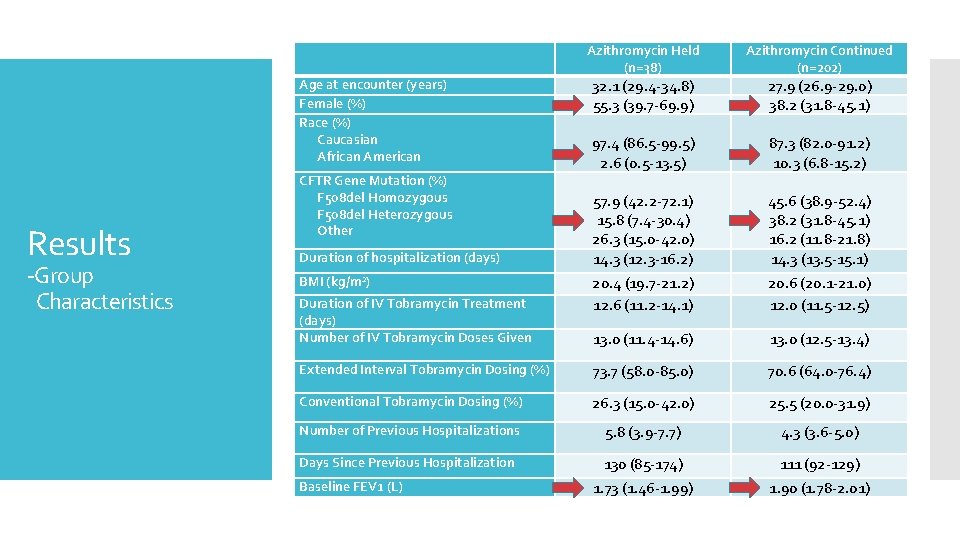
Azithromycin Held (n=38) Azithromycin Continued (n=202) 20. 4 (19. 7 -21. 2) 12. 6 (11. 2 -14. 1) 20. 6 (20. 1 -21. 0) 12. 0 (11. 5 -12. 5) 13. 0 (11. 4 -14. 6) 13. 0 (12. 5 -13. 4) Extended Interval Tobramycin Dosing (%) 73. 7 (58. 0 -85. 0) 70. 6 (64. 0 -76. 4) Conventional Tobramycin Dosing (%) 26. 3 (15. 0 -42. 0) 25. 5 (20. 0 -31. 9) Number of Previous Hospitalizations 5. 8 (3. 9 -7. 7) 4. 3 (3. 6 -5. 0) Days Since Previous Hospitalization 130 (85 -174) 1. 73 (1. 46 -1. 99) 111 (92 -129) 1. 90 (1. 78 -2. 01) Age at encounter (years) Female (%) Race (%) Caucasian African American Results -Group Characteristics CFTR Gene Mutation (%) F 508 del Homozygous F 508 del Heterozygous Other Duration of hospitalization (days) BMI (kg/m 2) Duration of IV Tobramycin Treatment (days) Number of IV Tobramycin Doses Given Baseline FEV 1 (L) 32. 1 (29. 4 -34. 8) 55. 3 (39. 7 -69. 9) 97. 4 (86. 5 -99. 5) 2. 6 (0. 5 -13. 5) 57. 9 (42. 2 -72. 1) 15. 8 (7. 4 -30. 4) 26. 3 (15. 0 -42. 0) 14. 3 (12. 3 -16. 2) 27. 9 (26. 9 -29. 0) 38. 2 (31. 8 -45. 1) 87. 3 (82. 0 -91. 2) 10. 3 (6. 8 -15. 2) 45. 6 (38. 9 -52. 4) 38. 2 (31. 8 -45. 1) 16. 2 (11. 8 -21. 8) 14. 3 (13. 5 -15. 1)
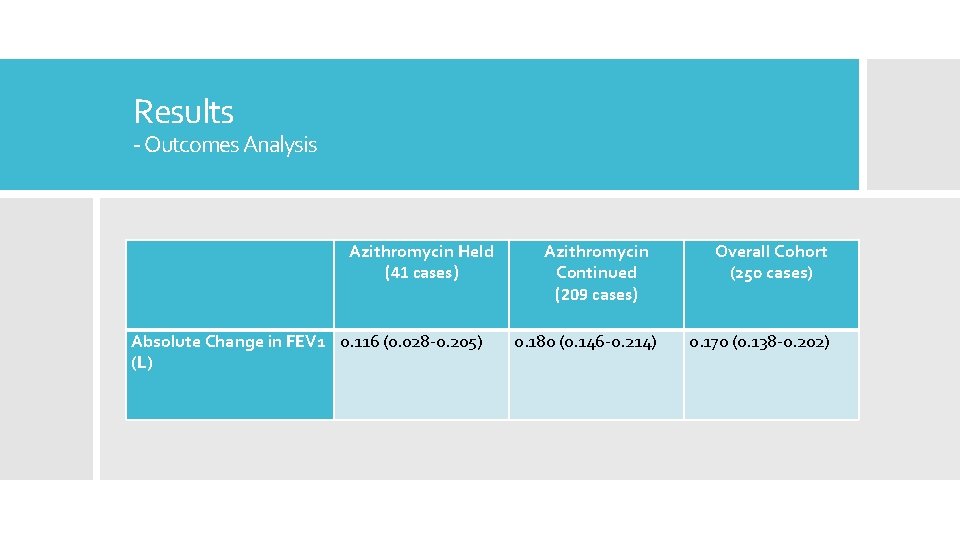
Results - Outcomes Analysis Azithromycin Held (41 cases) Absolute Change in FEV 1 0. 116 (0. 028 -0. 205) (L) Azithromycin Continued (209 cases) Overall Cohort (250 cases) 0. 180 (0. 146 -0. 214) 0. 170 (0. 138 -0. 202)

Coefficients P value Inpatient Azithromycin Exposure 0. 023 (-0. 059 to 0. 104) 0. 751 Age at Encounter -0. 005 (-0. 009 to -0. 001) 0. 020 0. 231 (0. 061 to 0. 401) 0. 008 - Outcomes Analysis Conventional Tobramycin Dosing R-squared = 0. 82 Extended Interval Tobramycin Dosing 0. 277 (0. 113 to -0. 440) 0. 001 Number of Previous Hospitalizations -0. 013 (-0. 019 to -0. 007) <0. 001 Baseline FEV 1 0. 071 (0. 031 to 0. 111) <0. 001 Results

Conclusions Holding outpatient azithromycin during inpatient exacerbation treatment with tobramycin therapy did not result in improved FEV 1 recovery Azithromycin has long t 1/2 & high lung concentrations Tobramycin also can induce mex. X and mex. Y efflux pump expression ¼ of the overall cohort was treated with tobramycin conventional dosing CFF guidelines state no difference in outcomes with extended interval dosing Age at encounter and number of previous hospitalizations were harmful covariates in regression model Baseline FEV 1, conventional tobramycin dosing, and extended interval tobramycin dosing were protective covariates in regression model Cases with mixed dosing regimens?

Strengths & Limitations Strengths: Adjusted for multiple potential confounders in regression model First analysis studying an intervention Detailed data from both EHR and CFF Patient Registry Medication orders, MAR, CF gene mutations, FEV 1 measurements at beginning and end of care episode Limitations: Encounter level data could be impacted by patients with more admissions Uneven case distribution across groups (41 vs 209) Did not adjust for additional antibiotic exposure or polymicrobial infection Short follow up time
- Slides: 16