Azeem Latib 1 MD Pierfrancesco Agostoni 2 Jo

Azeem Latib 1, MD, Pierfrancesco Agostoni 2, Jo Dens 3, Patrizio Lancellotti 4, Frankie C. C. Tam 5, Minglei Liu 6, Luca Testa 7 on behalf of the PREVAIL study investigators 1 Ospedale San Raffaele – Milano, Italy and Montefiore Medical Center, New York, NY, USA; 2 St. Antonius Ziekenhuis, Nieuwegein, Netherlands; 3 Ziekenhuis Oost Limburg – Campus Sint-Jan, Genk, Belgium; 4 CHU de Liège - Hôpital du Sart Tilman, Liège, Belgium; 5 Queen Mary Hospital, Hong Kong; 6 Medtronic PLC, Santa Rosa, USA; 7 IRCCS Policlinico San Donato, Italy © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 First clinical evaluation of the Prevail™ Paclitaxel-coated PTCA Balloon Catheter: Primary outcomes from the PREVAIL trial

☑ I have the following potential conflicts of interest to declare: Receipt of honoraria or consultation fees: Abbott, Cardiovalve, Medtronic, Nuvera, Philips, Supira, Venus Medtech, ICS, Cor. Flow, Transeptal Solutions, Bioventrix, Millipede, Mitralign, Acist Medical © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Speaker's name : Azeem Latib

Treatment of stenosed lesions during PCI with a drugcoated balloon (DCB) can be a useful technique in the setting of in-stent restenosis (ISR) or small vessels. A challenge with 1 st generation devices has been deliverability and drug absorption. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Why this study?

The PREVAIL trial is the first clinical study to evaluate the safety and efficacy of the Prevail™ DCB in the treatment of de novo and ISR lesions in patients with coronary artery disease. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 What did we study?
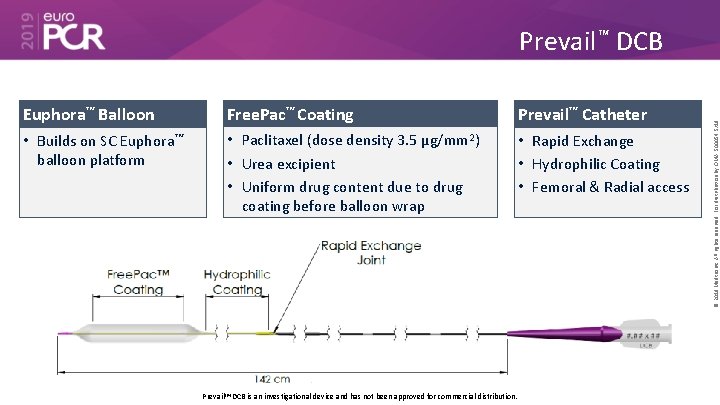
Euphora™ Balloon Free. Pac™ Coating Prevail™ Catheter • Builds on SC Euphora™ balloon platform • Paclitaxel (dose density 3. 5 μg/mm 2) • Urea excipient • Uniform drug content due to drug coating before balloon wrap • Rapid Exchange • Hydrophilic Coating • Femoral & Radial access Prevail™ DCB is an investigational device and has not been approved for commercial distribution. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Prevail™ DCB

PREVAIL Study Design CAD patients amenable to treatment with a Prevail™ Drug-Coated Balloon (diameter 2. 0 – 4. 0 mm) in coronary lesions (de novo, ISR) of ≤ 25 mm length 10 sites in Europe and Asia-Pacific Prevail™ DCB (N = 50) Follow-up 30 d 6 mo 12 mo Primary endpoint: In-stent (in-balloon) late lumen loss at 6 months (LLL, measured by QCA corelab) Secondary endpoints: Death, TVMI, revascularisation, stent thrombosis, TLF, TVF at 30 days, 6 and 12 months Acute success rates In-segment / in-stent (in-balloon) LLL, %DS and BAR at 6 months ASA indefinitely and P 2 Y 12 inhibitor for a minimum of 4 weeks Recommended DAPT: NCT 03260517 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Prospective, single arm, multi-centre study

PREVAIL Study Key Eligibility Criteria • Documented stable or unstable angina, and/or clinical evidence of ischemia • Successful predilatation of entire target lesion • Treatment eligible for DCB Lesion characteristics • Up to 3 target lesions in maximum 2 vessels (4 th lesion allowed with standard DES treatment) • RVD: 2. 0 – 4. 0 mm • Lesion length: ≤ 25 mm • TIMI flow: 2– 3 KEY EXCLUSION CRITERIA • Acute MI <72 h • Left main, bypass graft/SVG or bifurcation lesions • Previous PCI of target vessel: • ISR: < 3 months • de novo: < 9 months • Pretreatment using cutting balloons, atherectomy, laser and/or thrombectomy © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 KEY INCLUSION CRITERIA
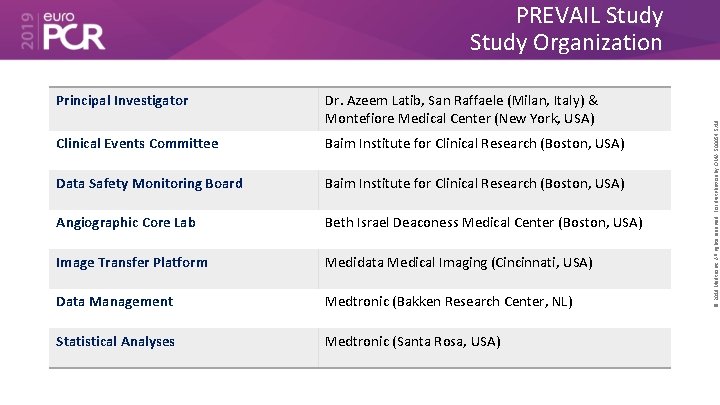
Principal Investigator Dr. Azeem Latib, San Raffaele (Milan, Italy) & Montefiore Medical Center (New York, USA) Clinical Events Committee Baim Institute for Clinical Research (Boston, USA) Data Safety Monitoring Board Baim Institute for Clinical Research (Boston, USA) Angiographic Core Lab Beth Israel Deaconess Medical Center (Boston, USA) Image Transfer Platform Medidata Medical Imaging (Cincinnati, USA) Data Management Medtronic (Bakken Research Center, NL) Statistical Analyses Medtronic (Santa Rosa, USA) © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 PREVAIL Study Organization
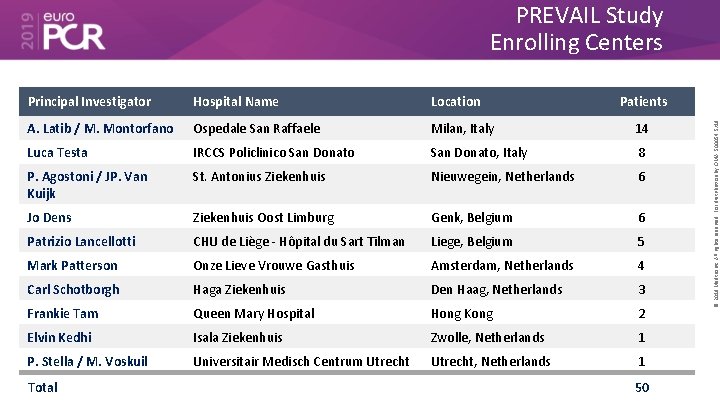
PREVAIL Study Enrolling Centers Hospital Name Location A. Latib / M. Montorfano Ospedale San Raffaele Milan, Italy 14 Luca Testa IRCCS Policlinico San Donato, Italy 8 P. Agostoni / JP. Van Kuijk St. Antonius Ziekenhuis Nieuwegein, Netherlands 6 Jo Dens Ziekenhuis Oost Limburg Genk, Belgium 6 Patrizio Lancellotti CHU de Liège - Hôpital du Sart Tilman Liege, Belgium 5 Mark Patterson Onze Lieve Vrouwe Gasthuis Amsterdam, Netherlands 4 Carl Schotborgh Haga Ziekenhuis Den Haag, Netherlands 3 Frankie Tam Queen Mary Hospital Hong Kong 2 Elvin Kedhi Isala Ziekenhuis Zwolle, Netherlands 1 P. Stella / M. Voskuil Universitair Medisch Centrum Utrecht, Netherlands 1 Total Patients 50 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Principal Investigator
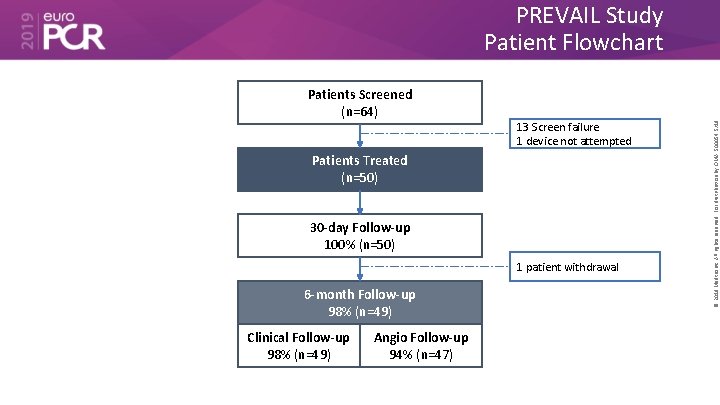
Patients Screened (n=64) 13 Screen failure 1 device not attempted Patients Treated (n=50) 30 -day Follow-up 100% (n=50) 1 patient withdrawal 6 -month Follow-up 98% (n=49) Clinical Follow-up 98% (n=49) Angio Follow-up 94% (n=47) © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 PREVAIL Study Patient Flowchart

© 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 RESULTS
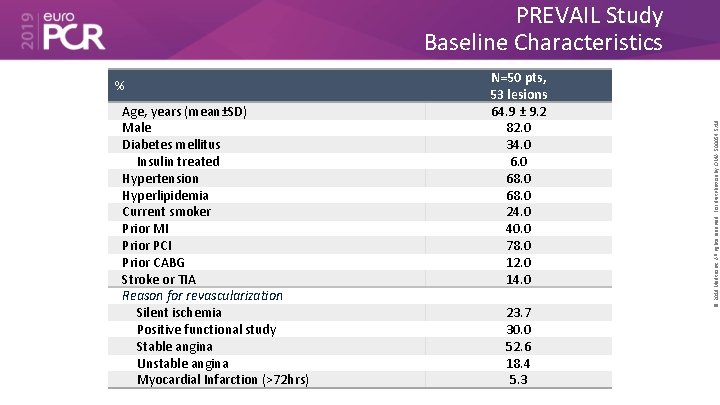
% Age, years (mean±SD) Male Diabetes mellitus Insulin treated Hypertension Hyperlipidemia Current smoker Prior MI Prior PCI Prior CABG Stroke or TIA Reason for revascularization Silent ischemia Positive functional study Stable angina Unstable angina Myocardial Infarction (>72 hrs) N=50 pts, 53 lesions 64. 9 ± 9. 2 82. 0 34. 0 68. 0 24. 0 40. 0 78. 0 12. 0 14. 0 23. 7 30. 0 52. 6 18. 4 5. 3 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 PREVAIL Study Baseline Characteristics
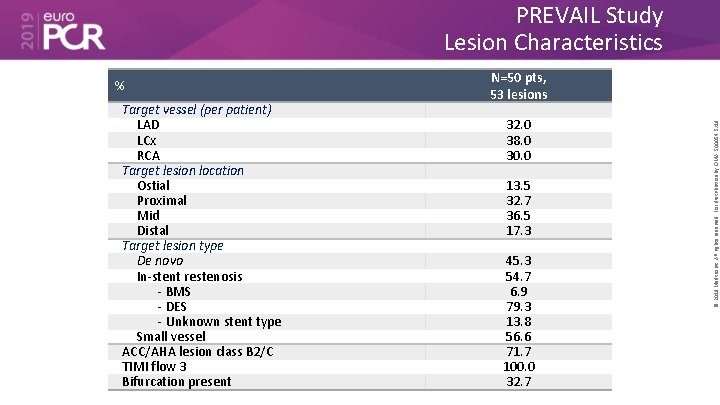
PREVAIL Study Lesion Characteristics Target vessel (per patient) LAD LCx RCA Target lesion location Ostial Proximal Mid Distal Target lesion type De novo In-stent restenosis - BMS - DES - Unknown stent type Small vessel ACC/AHA lesion class B 2/C TIMI flow 3 Bifurcation present N=50 pts, 53 lesions 32. 0 38. 0 30. 0 13. 5 32. 7 36. 5 17. 3 45. 3 54. 7 6. 9 79. 3 13. 8 56. 6 71. 7 100. 0 32. 7 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 %
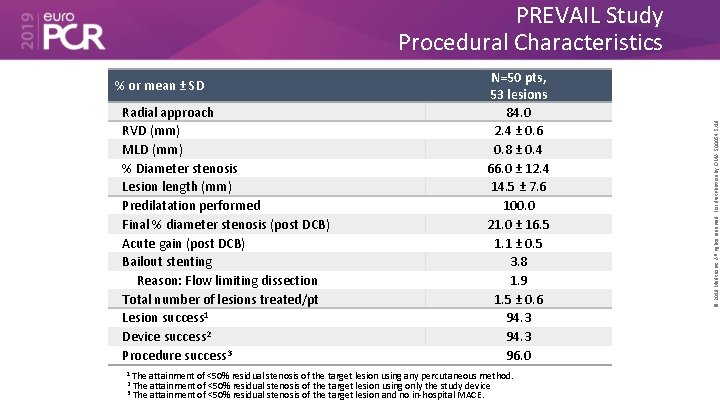
% or mean ± SD Radial approach RVD (mm) MLD (mm) % Diameter stenosis Lesion length (mm) Predilatation performed Final % diameter stenosis (post DCB) Acute gain (post DCB) Bailout stenting Reason: Flow limiting dissection Total number of lesions treated/pt Lesion success 1 Device success 2 Procedure success 3 N=50 pts, 53 lesions 84. 0 2. 4 ± 0. 6 0. 8 ± 0. 4 66. 0 ± 12. 4 14. 5 ± 7. 6 100. 0 21. 0 ± 16. 5 1. 1 ± 0. 5 3. 8 1. 9 1. 5 ± 0. 6 94. 3 96. 0 1 The attainment of <50% residual stenosis of the target lesion using any percutaneous method. 2 The attainment of <50% residual stenosis of the target lesion using only the study device 3 The attainment of <50% residual stenosis of the target lesion and no in-hospital MACE. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 PREVAIL Study Procedural Characteristics
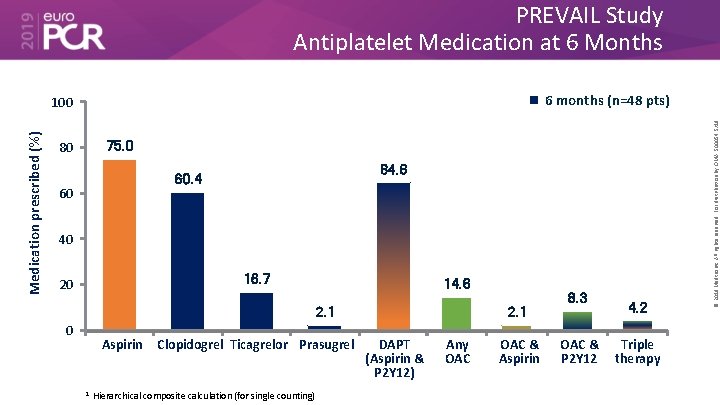
PREVAIL Study Antiplatelet Medication at 6 Months Medication prescribed (%) 75. 0 80 64. 6 60. 4 60 40 16. 7 20 14. 6 2. 1 0 Aspirin 1 Clopidogrel Ticagrelor Prasugrel Hierarchical composite calculation (for single counting) 2. 1 DAPT (Aspirin & P 2 Y 12) Any OAC & Aspirin 8. 3 OAC & P 2 Y 12 4. 2 Triple therapy © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 6 months (n=48 pts) 100
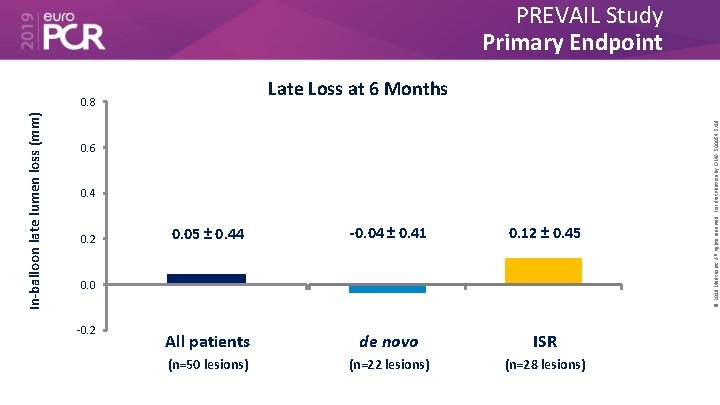
PREVAIL Study Primary Endpoint In-balloon late lumen loss (mm) 0. 6 0. 4 0. 2 0. 05 ± 0. 44 -0. 04 ± 0. 41 0. 12 ± 0. 45 All patients de novo ISR (n=50 lesions) (n=22 lesions) (n=28 lesions) 0. 0 -0. 2 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Late Loss at 6 Months 0. 8
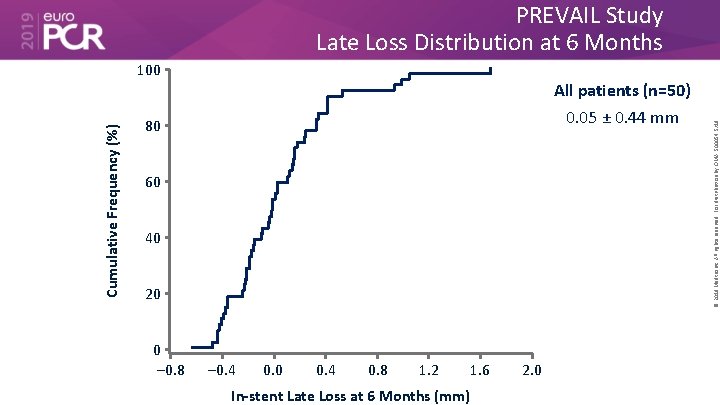
PREVAIL Study Late Loss Distribution at 6 Months All patients (n=50) 0. 05 ± 0. 44 mm 80 60 40 20 0 – 0. 8 – 0. 4 0. 0 0. 4 0. 8 1. 2 1. 6 In-stent Late Loss at 6 Months (mm) 2. 0 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Cumulative Frequency (%) 100
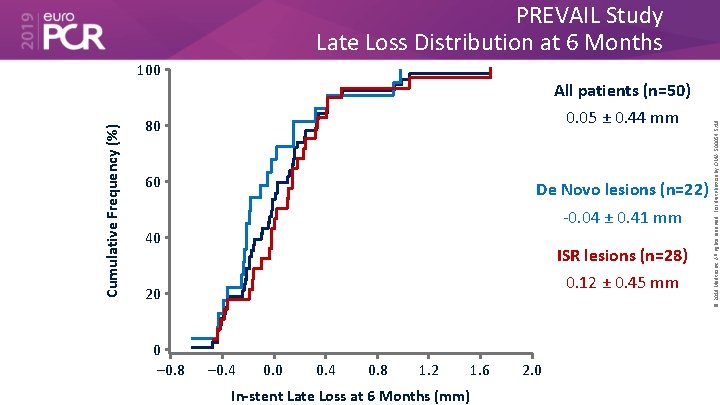
PREVAIL Study Late Loss Distribution at 6 Months All patients (n=50) 0. 05 ± 0. 44 mm 80 60 De Novo lesions (n=22) -0. 04 ± 0. 41 mm 40 ISR lesions (n=28) 0. 12 ± 0. 45 mm 20 0 – 0. 8 – 0. 4 0. 0 0. 4 0. 8 1. 2 1. 6 In-stent Late Loss at 6 Months (mm) 2. 0 © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Cumulative Frequency (%) 100
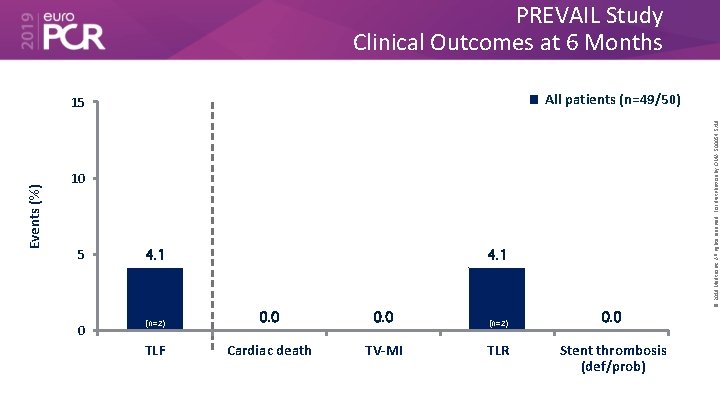
PREVAIL Study Clinical Outcomes at 6 Months Events (%) © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 All patients (n=49/50) 15 10 5 0 4. 1 (n=2) 0. 0 (n=2) TLF Cardiac death TV-MI TLR 0. 0 Stent thrombosis (def/prob)
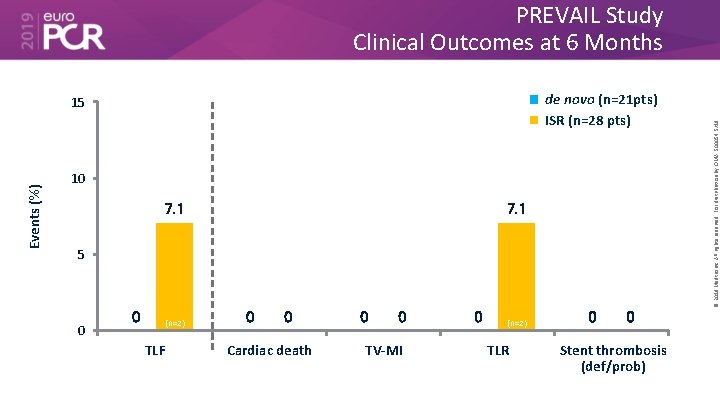
de novo (n=21 pts) ISR (n=28 pts) Events (%) 15 10 7. 1 5 0 0 (n=2) TLF 0 0 Cardiac death 0 0 TV-MI 0 (n=2) TLR 0 0 Stent thrombosis (def/prob) © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 PREVAIL Study Clinical Outcomes at 6 Months

• The Prevail™ DCB for the treatment coronary artery disease builds on the Euphora™ balloon platform and drug coating technology from the IN. PACT™ DCB family. • In the first 50 patients treated with the device, the 6 -month results showed a very favorable late loss of 0. 05 ± 0. 44 mm, with only 4. 1% TLR. • Importantly, there were no safety events observed out to 6 months (cardiac death, MI or ST) in this population, which included both de novo and ISR lesions. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 What are the essential results?

The Prevail™ DCB is designed for uniform drug distribution with improved deliverability; this device will be an incremental tool in the treatment of coronary artery disease. Prevail™ DCB is an investigational device and has not been approved for commercial distribution. © 2019 Medtronic. All rights reserved. For distribution by OMA. 500054 5/19 Why is this important?
- Slides: 22