Avian Influenza Treatment and prevention in man By

Avian Influenza Treatment and prevention in man By Dr. Ali Muhaidat Head of Vaccine and Sera Department MOH

The Disease n Avian Influenza is a contagious disease caused by a viral agent that infects birds mainly and some times pigs but is able to infect humans
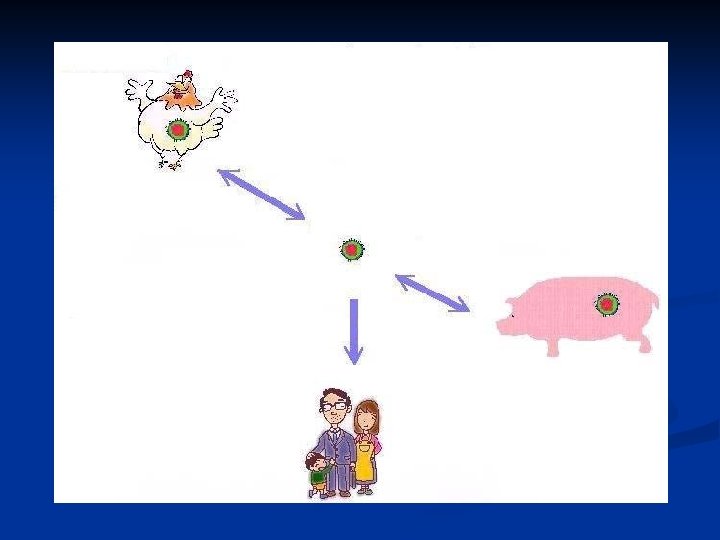

The Causative agent n The disease is caused by H 5 N 1 type A Influenza virus n A Member of Orthomyxo viruses group n An RNA enveloped virus

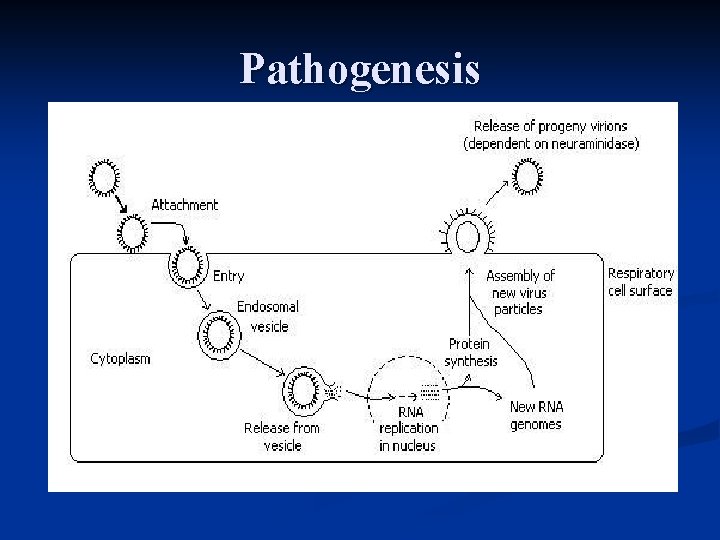
Pathogenesis

Treatment Available Drugs n Antiviral drugs may be used n There are two drugs in the neuraminidase inhibitors class • • n Oseltamivir (Tamiflu) Zanamivir (Relenza) The old types M 2 inhibitors amantadine and rimantadine may be used but resistance can develop rapidly

n Neuraminidase is responsible for the release of the virus from the infected cell by cleavage of receptors n The Neuraminidase inhibitors prevent the cleavage and so the release of the virus n By this mechanism Tamiflu and Relanza prevent the replication of the virus

Comparison between oseltamivir and zanamivir

Oseltamivir n n n Available as capsules and powder to be dissolved in water Can be taken orally Can be used for treatment of children > 1 year and in adults The dose in children is 2 mg/kg twice daily, in adults 75 mg twice daily for 5 days If used within 48 hrs of onset of symptoms reduced the length of illness by 1. 3 days

1. 2. 3. 4. Can be use in prevention of the disease in ages more than 13 years. The dose used for prevention is 75 mg daily for 6 weeks. The dose should be reduced in patient with renal impairment. Resistant isolates of the virus were noticed in 18% of treated children in a recent study. It reduced the incidence of influenza by 70% to 90%

Zanamivir n n n Available as powder for oral inhalation Needs a special device Can be used to treat children ( more than 7 years old) and adults A dose is 10 mg ( 2 inhalations ) twice daily for 5 days Reduces the length of illness by 1 to 2 days if used within 48 hrs of onset of symptoms Recent studies showed that it reduced the incidence of influenza by 70% to 80%

Zanamivir Powder in the sachet
Oral inhalation of Zanamivir powder

Vaccines n Vaccines against H 5 N 1 Influenza is not yet available n The candidate vaccine is under production and it is expected to be available next year

n The pandemic vaccine against HPAI, H 5 N 1 strain differs from seasonal influenza vaccine in many ways 1. Antigen content 2. Type of adjuvent 3. Dosage and route of administration 4. No efficacy trials can be done 5. Immunogenicity can be done

Are Vaccine producers ready for production? Chiron is in the process of producing the vaccine in Italy and in the UK n Sanofi Pasteur is also in the process of producing the vaccine n GSK will produce the second generation with a new adjuvant n Many countries are now in the process of purchasing the vaccine and they are now in the queue such as France and Finland, UK has already order their stock n

Vaccines Production Process n n n n Choice of virus reassortant strain Master seed strain production Working seed strain production Single harvest Viral inactivation Splitting Viral inactivation test

1. 2. 3. 4. 5. 6. Purification Identity testing and antigen content Sterility testing Formulation Filling Final product Q. C (Sterility, Identity, antigen content, PH, … )

Conclusion n Osltamivir and Zanamivir may be used for both purposes treatment and prophylaxis n Vaccines are the best way to prevent the disease production of antiviral drugs and high demand

Thank You
- Slides: 21