Avian Influenza Saad Gharaibeh DVM Ph D Dip

Avian Influenza Saad Gharaibeh DVM, Ph. D, Dip ACPV Dept. of Pathology and Animal Health Faculty of Veterinary Medicine Jordan University of Science and Technology Irbid 22110, Jordan saadgh@just. edu. jo 02/720 -1000 ext 22059

Avian Influenza History 1. 2. 3. 4. 5. 6. 7. 1878 Fowl plaque was described (Italy) 1901 Fowl plaque is caused by a virus 1955 It is type A influenza virus 1970 AGP test introduced 1972 Waterfowl is a reservoir 1979 Virulence and hemagglutinin cleavability was established 1997 Direct transmission of H 5 AIV from birds to humans

Avian Influenza Virus 1. Orthomyxoviridae 2. Pleomorphic RNA viruses, single stranded, negative sense genome. 3. Has glycoprotein projections HA, NA 4. Three antigenic types A, B, C (Avian influenzas are all type A) 5. 8 gene segments code for 10 proteins 6. Vary in pathogenicity
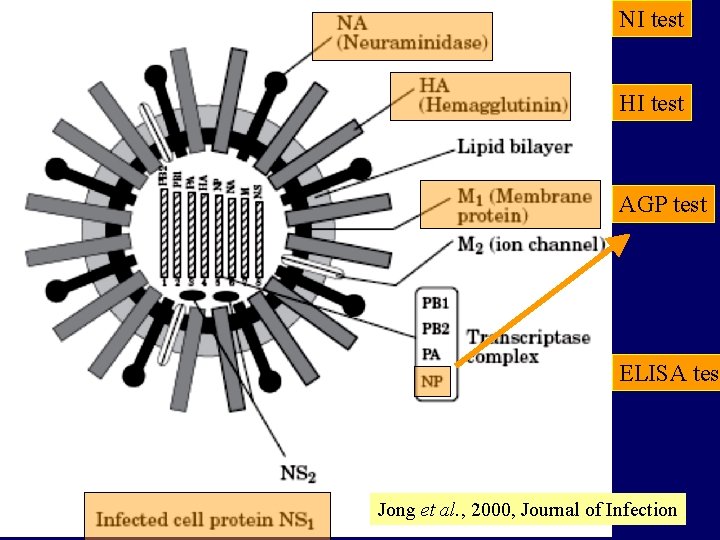
NI test HI test AGP test ELISA test Jong et al. , 2000, Journal of Infection
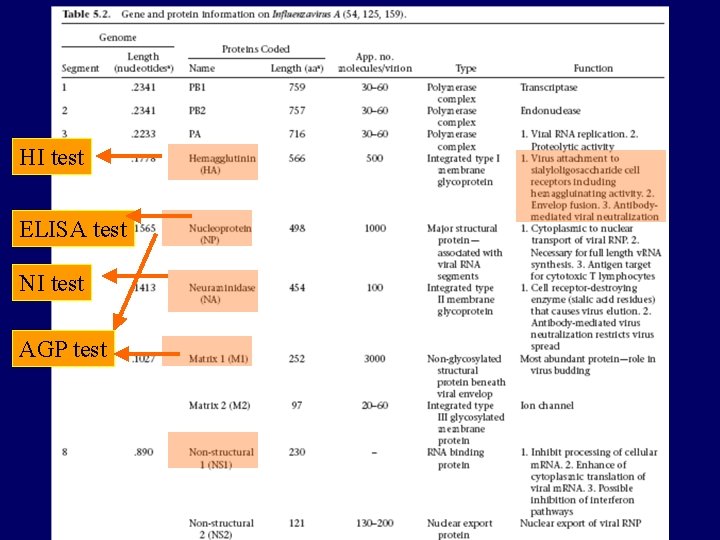
HI test ELISA test NI test AGP test

Nomenclature A/chicken/Hong Kong/220/97 (H 5 N 1) 1. 2. 3. 4. 5. 6. A: Type of virus A, B, C Chicken: Host of origin Hong Kong: Geographic origin 220: Strain Number (Case number) 97: Year of isolation (H 5 N 1): H & N subtype

Infectious Virus 1. Needs HA 0 cleaved into HA 1 & HA 2 2. Intracytoplasmic: a) Furin-like enzyme (ubiquitous proteases): HP b) Trypsine-like enzyme: All AIV
Cell Types for Replication 1. All AIV (trypsine-like enzymes): a) Respiratory epithelium b) GI epithelium 2. HP AIV (Furin-like enzymes): Variety of cells resulting in a systemic infection.

Approaches Used to Characterize AIV Pathogenicity 1. In vivo methods: a) b) 2. In vitro methods (evaluation of HA cleavability): a) b) c) 3. Laboratory Inoculation of chickens Chicken embryo lethality Plaques or CPE assays (CEF does not have trypsin) Direct detection of cleaved HA Nucleotide sequence of HA cleavage site Direct measure of pathogenicity potential: a) b) Identify pathogenicity increases during virus passage in chickens under controlled conditions Virulence in mice and ability to infect other mammals

Criteria for HP 1. AIV lethal for 6, 7, or 8 / 8 four-to-six-weekold susceptible chickens within 10 days following IV inoculation with 0. 2 ml of 1: 10 dilution of a bacteria free, infectious allantoic fluid. 2. H 5 or H 7 has amino acid sequence at the hemagglutinin cleavage site compatible with HPAIV 3. Non-H 5 or H 7 that kills 1 -5 chickens and grows in cell culture w/o added trypsin

Signalment & Clinical Signs (Low pathogenic AI disease) a) b) c) d) Respiratory signs Diarrhea Drop in egg production 7 -10 days 5 -30% Mild increase in Mortality (2 o bacterial infection will increase mortality)

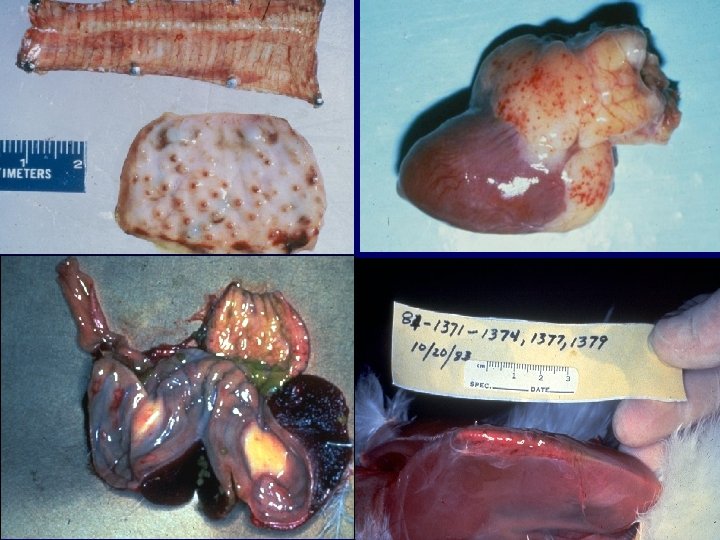
Gross Lesions (Low pathogenic AI disease) a) b) c) d) e) f) Catarrhal rhinitis / tracheitis Ocular discharge Airsaculitis Ovarian involution and hemorrhage Yolk peritonitis Swollen kidney and urates

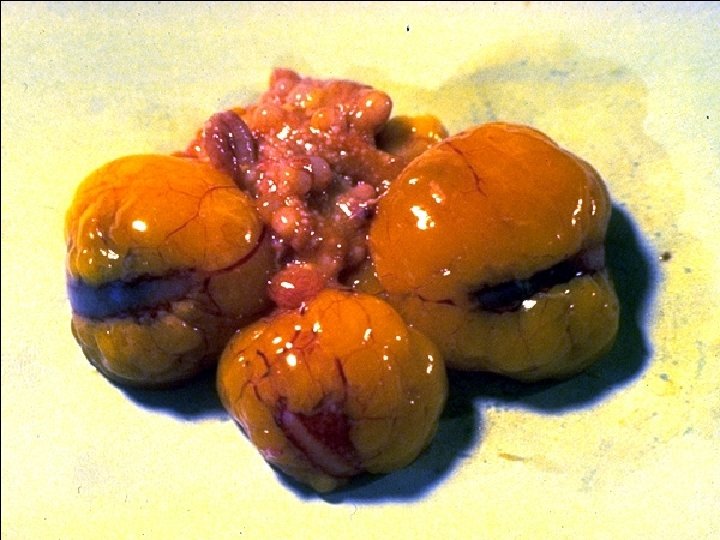

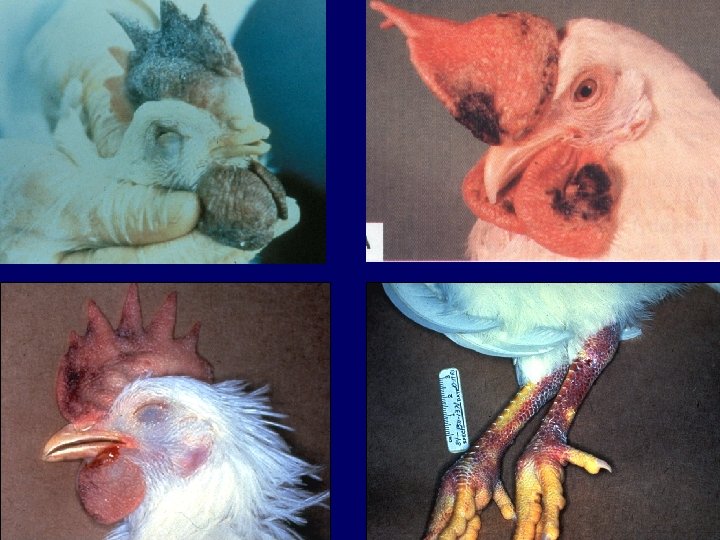
Signalment & Clinical Signs (Highly pathogenic AI disease) a) Sudden onset of high mortality (up to 100%) b) Depression c) +/- nervous signs




Gross Lesions (Highly pathogenic AI disease) a) Edematous to necrotic comb and wattles b) Edema, necrosis, and hemorrhages in different organs

High Path Avian Influenza Diagnosis 1. Clinical features in commercial poultry give a tentative diagnostic Sudden death and high mortality rate 2. RT-PCR and sequencing 3. Virus isolation and identification is the gold standard but very few laboratories in the world can handle such a virus capable of infecting humans. 4. Commercial antigen capture ELISA (lack sensitivity and will cross react with other endemic subtypes) 5. Serology: AGP, ELISA, HI, NI

RT-PCR 1. Testing can be performed in one day for multiple agents. 2. Sensitivity is very high and comparable to virus isolation. 3. Can be applied on samples from any species. 4. Decrease the chance of contamination with live virus.

RT-PCR Diagnostics in JUST Safety Considerations
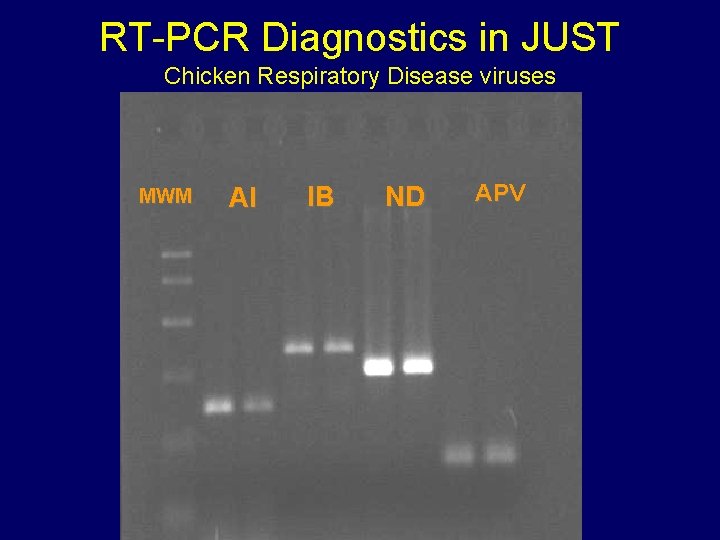
RT-PCR Diagnostics in JUST Chicken Respiratory Disease viruses MWM AI IB ND APV

Serologic Testing and Surveillance 1. 2. 3. 4. 5. 6. AGPT: Type specific (available at JUST) ELISA: Type specific (available at JUST) HI: Subtype specific (available at JUST) NI: Subtype specific Antigen capture ELISA (available at JUST) RT-PCR: Surveillance and diagnosis (available at JUST)
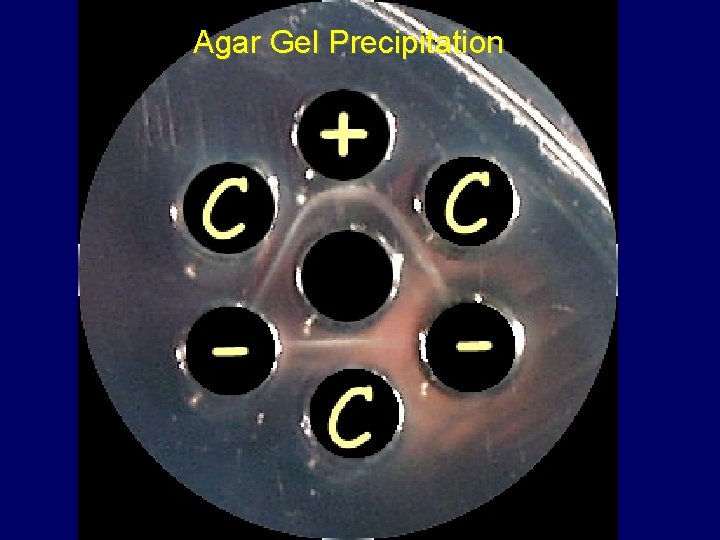
Agar Gel Precipitation
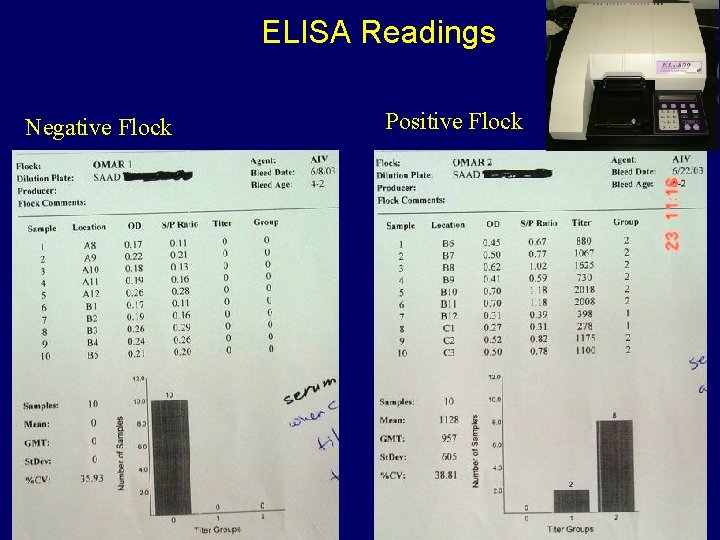
ELISA Readings Negative Flock Positive Flock

Hemagglutination Inhibition

Control & Prevention 1. Biosecurity 2. Stamping out infected flocks 3. Vaccination of flocks at high risk : a) Killed vaccines b) Viral vector vaccines c) Live attenuated vaccines are not licensed for poultry



Drastic measures in some Asian countries www. animalactivism. org/ documents




AI crossed Species Barrier into Humans A/chicken/Hong Kong/220/97 (H 5 N 1)

What conditions favor AI spread?

Densely populated countries


Very popular Live-bird markets


Free Range Poultry
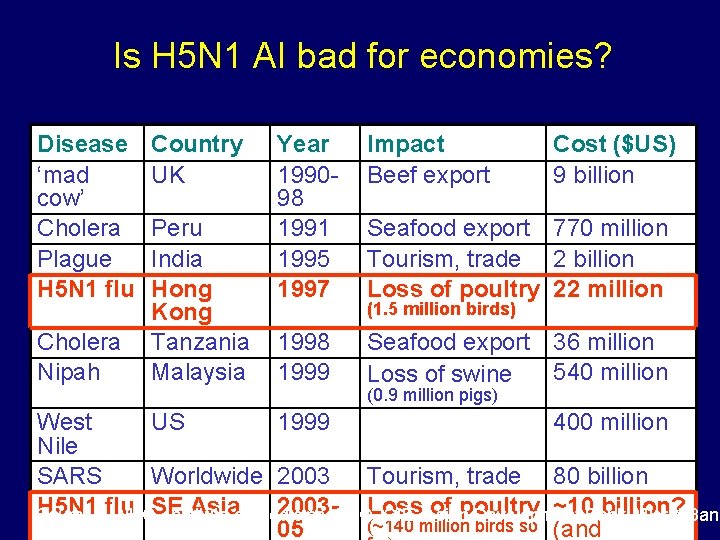
Is H 5 N 1 AI bad for economies? Disease ‘mad cow’ Cholera Plague H 5 N 1 flu Cholera Nipah Country UK Peru India Hong Kong Tanzania Malaysia Year 199098 1991 1995 1997 Impact Beef export Cost ($US) 9 billion 1998 1999 Seafood export 36 million 540 million Loss of swine Seafood export 770 million Tourism, trade 2 billion Loss of poultry 22 million (1. 5 million birds) (0. 9 million pigs) West US 1999 400 million Nile SARS Worldwide 2003 Tourism, trade 80 billion H 5 N 1 flu SE Asiaof Medicine, 2003 -FAO, Loss of poultry ~10 Bank, billion? Sources: WHO, Institute OIE, Asian Development World Bank (~140 million birds so 05 (and
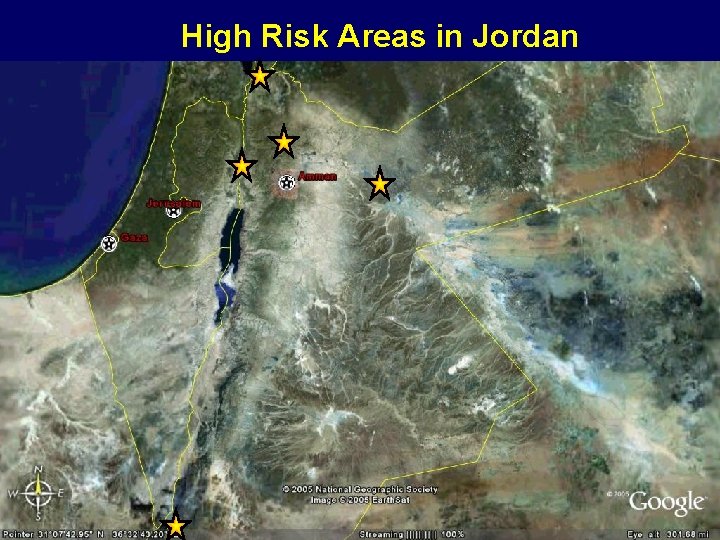
High Risk Areas in Jordan

Should we start vaccinating poultry against H 5 N 1?

Killed Vaccines 1. 2. 3. 4. Will result in only humoral antibody response against all viral proteins except NS 1. Will significantly reduce shedding of the challenge virus. Will interfere with AGP, ELISA, HI, and NI if (homologous). If sequence of the HA gene is identical to the challenge virus it may eliminate shedding completely.
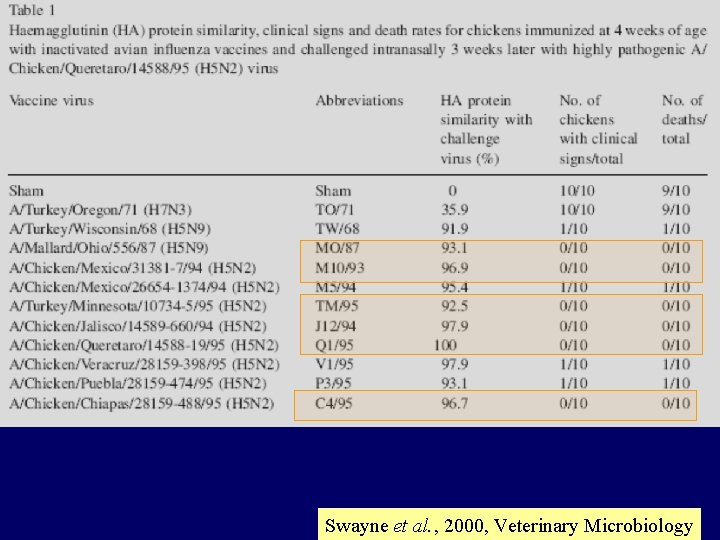
Swayne et al. , 2000, Veterinary Microbiology

Advantages of Vaccines • Reduces the number of chickens from which AI challenge virus could be reisolated. • Decreased the titers of virus detected in the cloaca and oropharynx (up to 99. 99%) • Reduced environmental contamination and prevented subsequent bird to bird transmission. • The use of killed H 5 N 2 vaccine in the face of HPAI H 5 N 1 virus challenge was able to protect chickens from disease and interrupt virus transmission.

DIVA Differentiating Infected from Vaccinated Individuals • The use of killed vaccine and unvaccinated sentinels: leaving 0. 5 -1% of the flock unvaccinated and marked (wing band) and these individuals will be subjected to serological monitoring. • Heterogonous killed vaccine: Screen for field infection using NI. • Measuring serological response to NS 1 by ELISA or western blot.

Vaccine / Industry / Politics • • • The use of vaccine to aid in the control of AI is a political issue and different people have a different say on this. In some countries financial constrains preclude stamping out policy. In some countries, export markets are not an issue to prevent vaccination. In some countries, stamping out attempt may be unsuccessful. “With the ubiquitous nature of AI in wild birds it may be vaccination the most feasible tool to soften the sting of AI” Beard 1981

Vaccine / Industry / Politics 6. 7. 8. 9. “Field results have not shown vaccine to increase the risk of undetected infection; in fact, field experience has shown that vaccination greatly enhances a control program. ” Halvorson, 2002, Avian Pathology There is no way a vaccinated flock can be a greater threat to disease control than a non-vaccinated flock that breaks with AI. Halvorson, 2002, Avian Pathology Epidemiological observations have shown that serologically positive birds are not associated with AI transmission. (Kradel, 1992) Should the government set the rules when no indemnity (compensation) is paid?

- Slides: 54