AVERT Trial Atorvastatin Versus Revascularization Treatments AVERT Trial











- Slides: 17

AVERT Trial Atorvastatin Versus Revascularization Treatments (AVERT) Trial Presented at The American Heart Association Scientific Sessions 1998 Presented by Dr. Bertram Pitt

AVERT Trial: Background • The goal of the AVERT trial was to assess the effect of aggressive lipid-lowering therapy on ischemic events in low-risk patients with single- or doublevessel CAD. Presented at AHA 1998

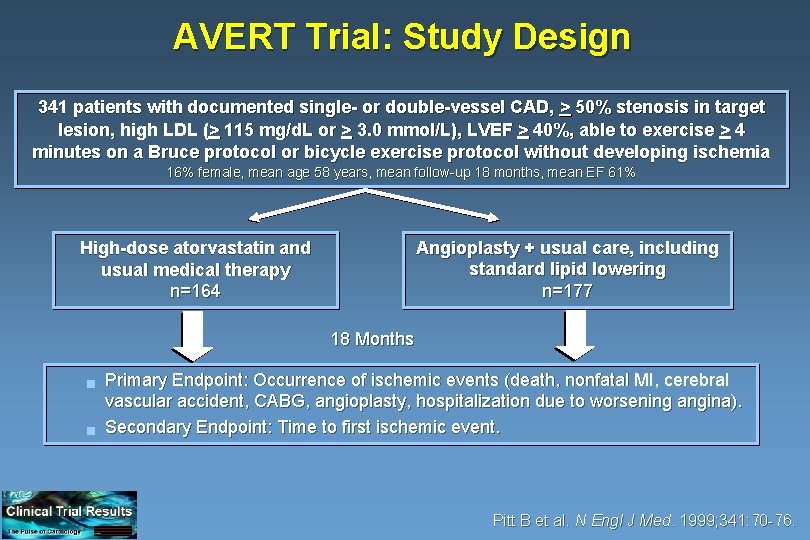
AVERT Trial: Study Design 341 patients with documented single- or double-vessel CAD, > 50% stenosis in target lesion, high LDL (> 115 mg/d. L or > 3. 0 mmol/L), LVEF > 40%, able to exercise > 4 minutes on a Bruce protocol or bicycle exercise protocol without developing ischemia 16% female, mean age 58 years, mean follow-up 18 months, mean EF 61% Angioplasty + usual care, including standard lipid lowering n=177 High-dose atorvastatin and usual medical therapy n=164 18 Months g g Primary Endpoint: Occurrence of ischemic events (death, nonfatal MI, cerebral vascular accident, CABG, angioplasty, hospitalization due to worsening angina). Secondary Endpoint: Time to first ischemic event. Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

AVERT: Major Exclusion Criteria • Left main disease or 3 -vessel disease • Unstable angina • MI within previous 14 days • Known ejection fraction <40% or NYHA Class III or IV heart failure • Previous CABG, unless grafts were patent and patient did not have 3 -vessel disease • CABG recommended based on current angiogram • Percutaneous revascularization in previous 6 months • Known hypersensitivity to HMG-Co. A reductase inhibitors • AST/ALT >2 x ULN • CPK >3 x ULN or unexplained elevations Pitt B et al. N Engl J Med. 1999; 341: 70 -76. Mc. Cormick LS et al. Am J Cardiol. 1997; 80: 1130 -1133.

AVERT: Overview of Study Procedures Treatment phase • Patients randomized to atorvastatin – discontinued other lipid-lowering medication and immediately began atorvastatin 80 mg/d • Patients randomized to angioplasty/usual care (UC) – underwent angioplasty followed by “usual care” • usual care may or may not have included lipid-lowering therapy (eg, diet, behavior modification, or medication) • angioplasty may or may not have included stenting • usual care was determined by investigator or patient’s primary physician Pitt B et al. N Engl J Med. 1999; 341: 70 -76. Mc. Cormick LS et al. Am J Cardiol. 1997; 80: 1130 -1133.

AVERT: Primary Efficacy Assessment • Incidence of an ischemic event in each treatment group • Ischemic event was defined as occurrence of one of the following: – cardiac death – resuscitation after cardiac arrest – nonfatal MI – CVA – CABG – angioplasty (other than the original procedure in angioplasty/usual care group) – worsening angina verified by objective evidence resulting in hospitalization CVA=cerebrovascular accident. Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

AVERT: Secondary Efficacy Assessments • Time from randomization to ischemic event • Percent change from baseline in TC, LDL-C, HDL-C, TG, apo A 1, apo B, and Lp(a) • All-cause mortality • Change from baseline in angina class • Worsening angina with objective evidence • Change in quality of life • Economic assessment Pitt B et al. N Engl J Med. 1999; 341: 70 -76. Mc. Cormick LS et al. Am J Cardiol. 1997; 80: 1130 -1133.

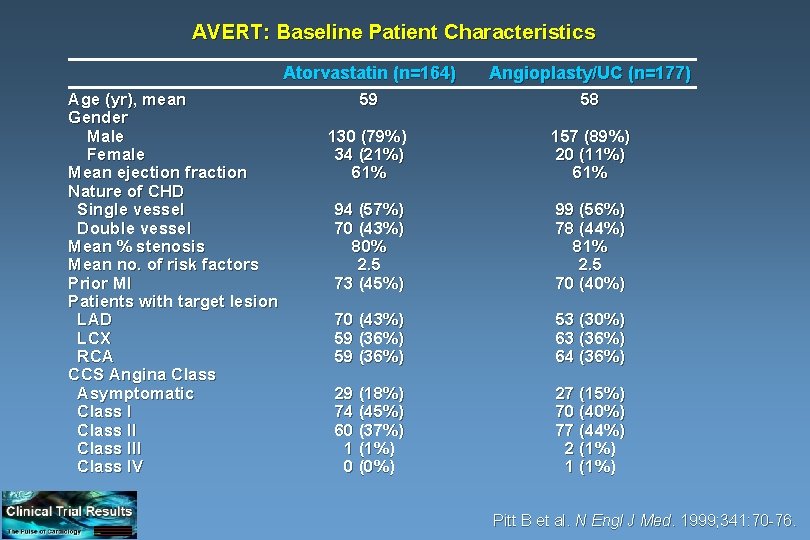
AVERT: Baseline Patient Characteristics Age (yr), mean Gender Male Female Mean ejection fraction Nature of CHD Single vessel Double vessel Mean % stenosis Mean no. of risk factors Prior MI Patients with target lesion LAD LCX RCA CCS Angina Class Asymptomatic Class III Class IV Atorvastatin (n=164) Angioplasty/UC (n=177) 59 58 130 (79%) 34 (21%) 61% 157 (89%) 20 (11%) 61% 94 (57%) 70 (43%) 80% 2. 5 73 (45%) 99 (56%) 78 (44%) 81% 2. 5 70 (40%) 70 (43%) 59 (36%) 53 (30%) 63 (36%) 64 (36%) 29 (18%) 74 (45%) 60 (37%) 1 (1%) 0 (0%) 27 (15%) 70 (40%) 77 (44%) 2 (1%) 1 (1%) Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

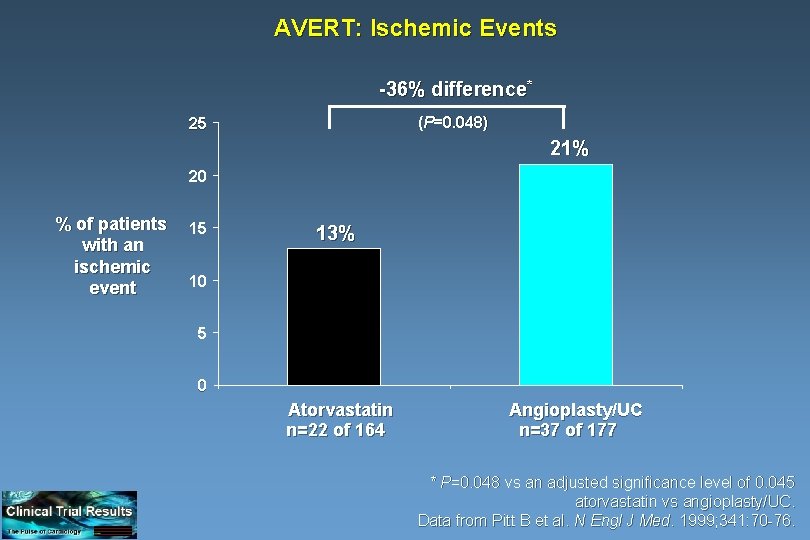
AVERT: Ischemic Events Number (%) of patients experiencing an ischemic event Any Ischemic event Atorvastatin n=164 Angioplasty/UC n=177 % 22 (13) 37 (21) -36* Death 1 (0. 6) Resuscitated cardiac arrest 0 (0. 0) Nonfatal MI 4 (2. 4) CVA 0 (0. 0) CABG 2 (1. 2) Revascularization 18 (11. 0) Worsening angina with objective evidence & hospitalization 11 (6. 7) 1 (0. 6) 0 (0. 0) 5 (2. 8) 0 (0. 0) 9 (5. 1) 21 (11. 9) 25 (14. 1) *P=0. 048 vs an adjusted significance level of 0. 045. Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

AVERT: Ischemic Events -36% difference* (P=0. 048) 25 21% 20 % of patients with an ischemic event 15 13% 10 5 0 Atorvastatin n=22 of 164 Angioplasty/UC n=37 of 177 * P=0. 048 vs an adjusted significance level of 0. 045 atorvastatin vs angioplasty/UC. Data from Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

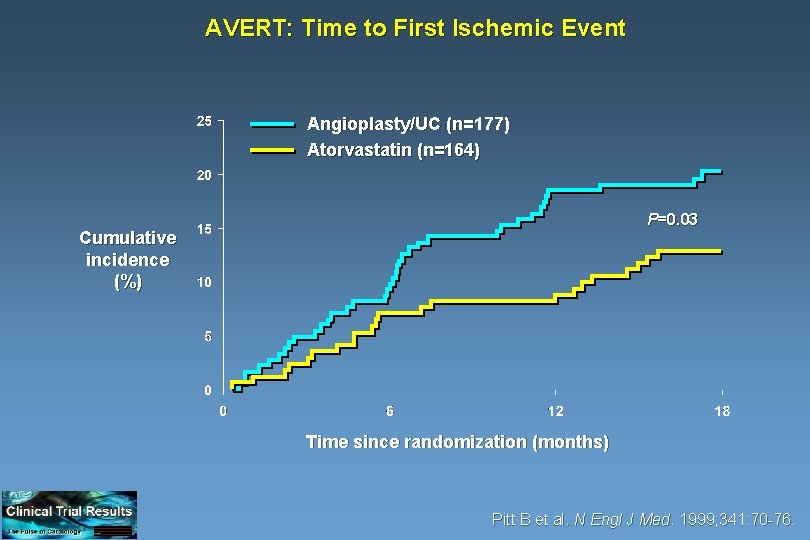
AVERT: Time to First Ischemic Event Angioplasty/UC (n=177) Atorvastatin (n=164) P=0. 03 Cumulative incidence (%) Time since randomization (months) Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

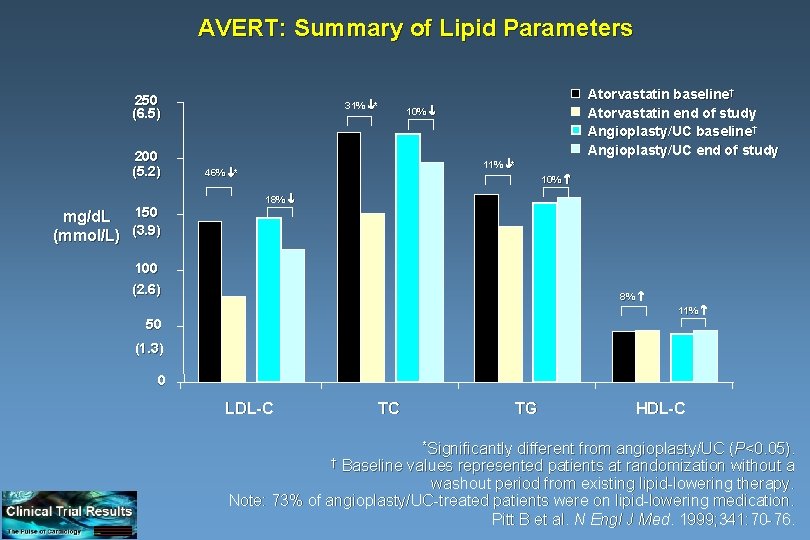
AVERT: Summary of Lipid Parameters 250 (6. 5) 200 (5. 2) mg/d. L 150 (mmol/L) (3. 9) 31% * Atorvastatin baseline† Atorvastatin end of study Angioplasty/UC baseline† Angioplasty/UC end of study 10% 11% * 46% * 10% 18% 100 (2. 6) 8% 11% 50 (1. 3) 0 LDL-C TC TG different from angioplasty/UC (P<0. 05). Baseline values represented patients at randomization without a washout period from existing lipid-lowering therapy. Note: 73% of angioplasty/UC-treated patients were on lipid-lowering medication. Pitt B et al. N Engl J Med. 1999; 341: 70 -76. † *Significantly HDL-C

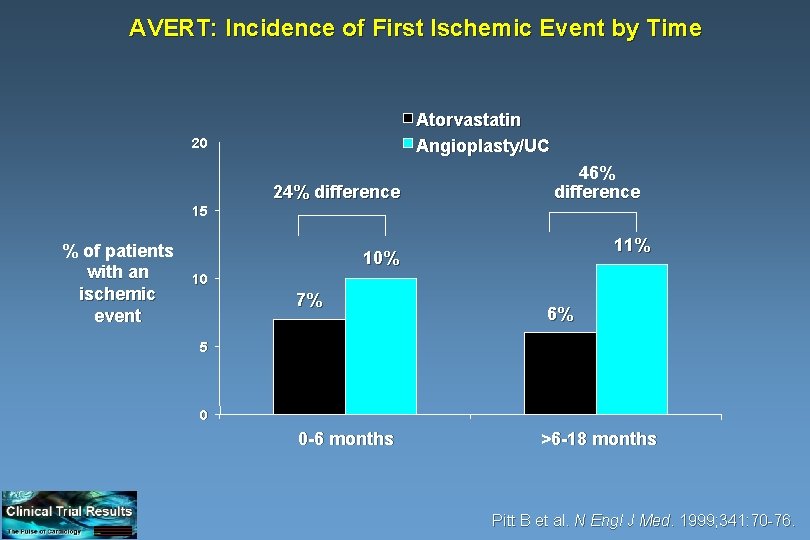
AVERT: Incidence of First Ischemic Event by Time Atorvastatin Angioplasty/UC 20 24% difference 46% difference 15 % of patients with an ischemic event 11% 10 7% 6% 5 0 0 -6 months >6 -18 months Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

AVERT: Safety Evaluation • Elevations in AST or ALT (consecutive elevations >3 x ULN) – 4 (2. 4%) atorvastatin-treated patients – none in angioplasty/UC-treated patients • Elevations in CPK (>10 x ULN) – none in either treatment group • There were no clinically significant differences in adverse event rates between the two treatment groups – in this study, eight patients discontinued atorvastatin treatment due to an adverse event, seven of which remained in the study Pitt B et al. N Engl J Med. 1999; 341: 70 -76.

AVERT Trial: Limitations • The patients enrolled in the AVERT trial were extremely low -risk, with stable CAD, 1 or 2 vessel CAD, and normal ventricular function. • The low-risk patient population randomized to angioplasty in this study may not reflect the population of patients that receive percutaneous interventions in clinical practice. Presented at AHA 1998

AVERT Trial: Limitations (cont. ) • Although the primary endpoint was clinically interesting, it was not statistically significant after alpha adjustment for interim analyses. • Looking at the KM curves, it appears that the benefit of atorvastatin for ischemic events is not apparent until six months from the onset of therapy • It could be speculated that this is due to the time required for plaque stabilization and a reduction in new lesion development. Presented at AHA 1998

AVERT: Conclusions Aggressive lipid lowering with atorvastatin in stable CAD patients: • Reduces ischemic events by 36% • Delays the time to first event • Is safe • Can delay or prevent the need for percutaneous revascularization Pitt B et al. N Engl J Med. 1999; 341: 70 -76.