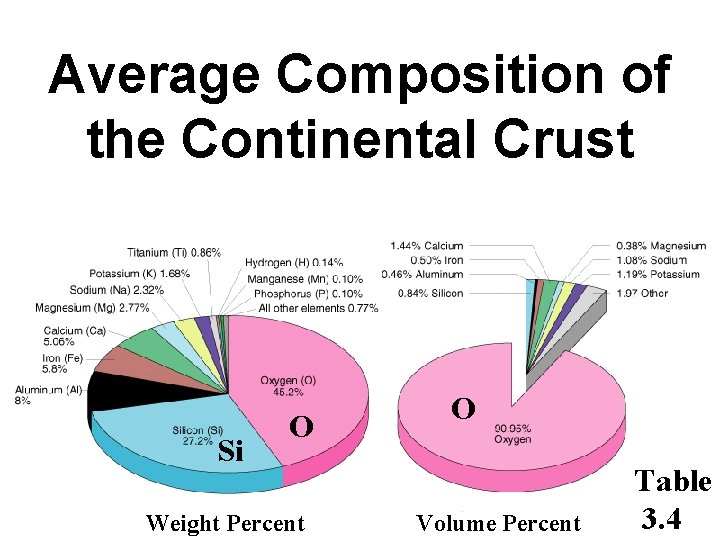
Average Composition of the Continental Crust Si O









- Slides: 21

Average Composition of the Continental Crust Si O Weight Percent O Volume Percent Table 3. 4

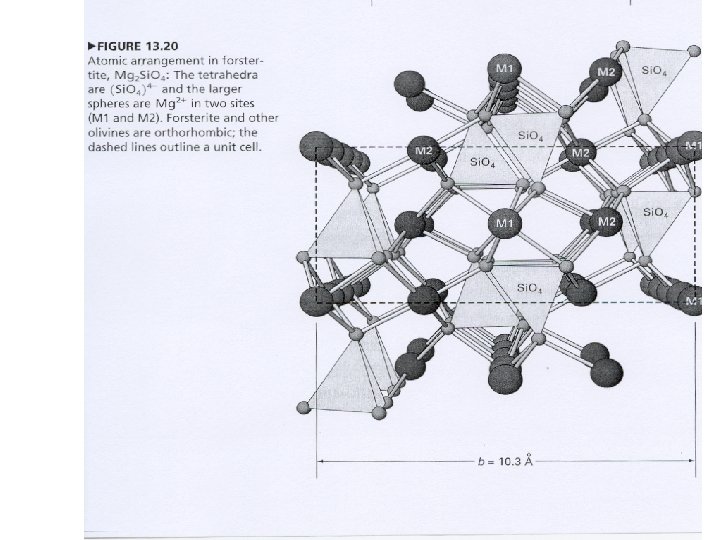
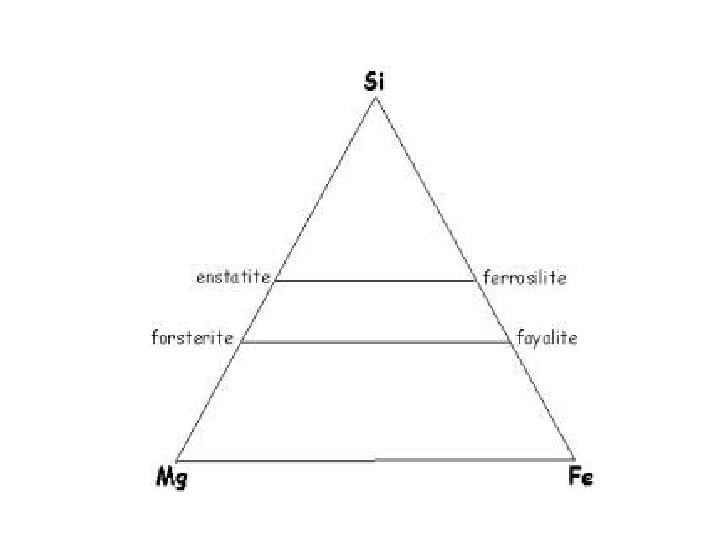
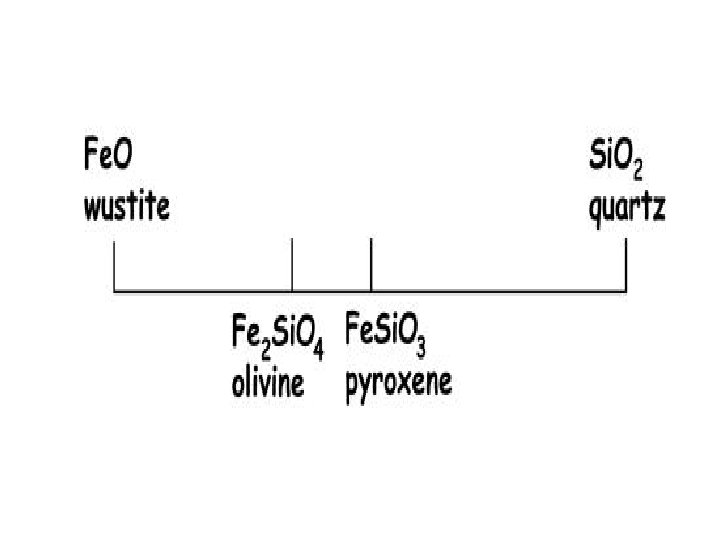
Olivine: formed from single silica tetrahedra


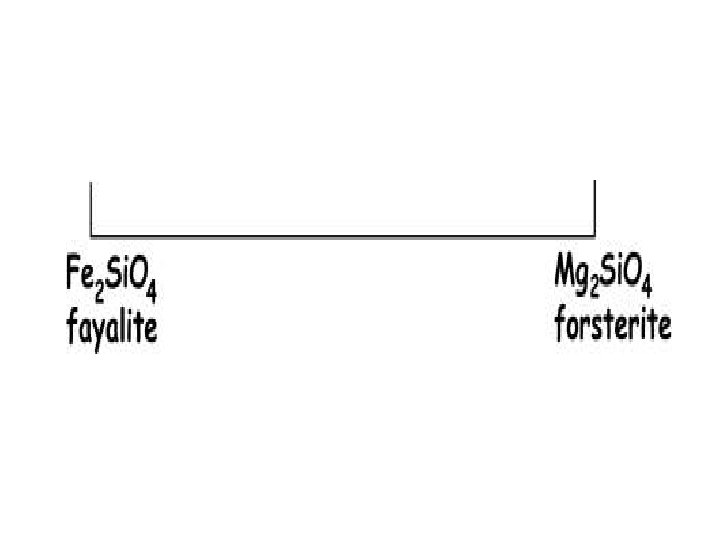
Phase – olivine ; Mg 2 Si. O 4 Components: Mg(2), Si, O(4) Can be reduced to Mg. O(2) and Si. O 2 Which is 66. 67 mole percent Mg. O and 33. 33 mole percent Si. O 2 This can be converted to weight by: . 6667 moles Mg. O X 40. 31 g/mole = 26. 875 g. 3333 moles Si. O 2 X 60. 09 g/mole = 20. 028 g 46. 903 g Giving 57. 3 weight % Mg. O and 42. 7 weight % Si. O 2

Given: 57. 3 wt. % Mg. O and 42. 7 wt. % Si. O 2 57. 3 g / 40. 31 g/mole = 1. 4215 moles Mg. O 42. 7 g / 60. 09 g/mole = 0. 7106 moles Si. O 2 1. 4215/(1. 4215 + 0. 7106) x 100 = 66. 67 mole % Mg. O 0. 7106/(1. 4215 + 0. 7106) x 100 = 33. 33 mole % Si. O 2 Or a mole ratio ( sometimes called “atomic”) of 2 Mg. O to 1 Si. O 2 The mineral formula would then be based on 4 oxygens giving Mg 2 Si. O 4, which is olivine ( specifically forsterite)

Actual analysis (wt. ) would give 34. 5 % Mg, 20. 0 % Si (with the remaining 45. 5 % assumed to be oxygen) Mg (24. 31 g/mole), Si (28. 09 g/mole) O ( 16. 00 g/mole) 34. 5 g / 24. 31 g/mole = 1. 42 moles 20. 0 g / 28. 09 g/mole = 0. 71 moles 45. 5 g / 16 g/mole = 2. 84 moles Mg 1. 42 Si 0. 71 O 2. 84 converted to 1 mole O gives Mg 0. 5 Si 0. 25 O 1 converted to whole numbers gives Mg 2 Si O 4









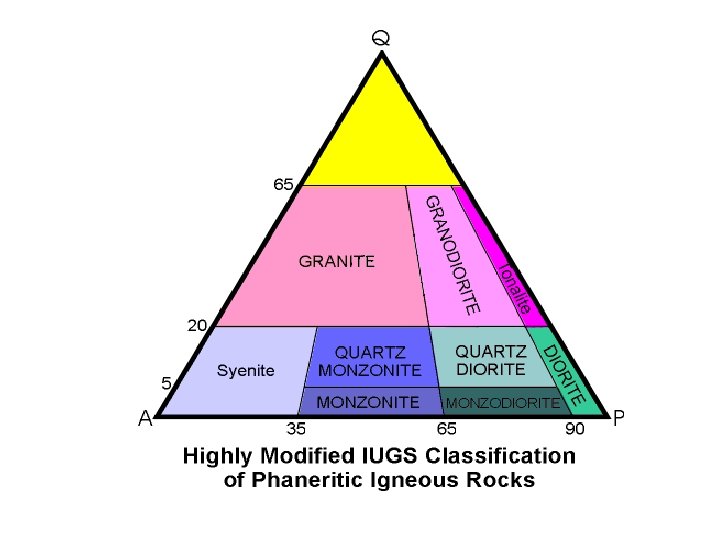
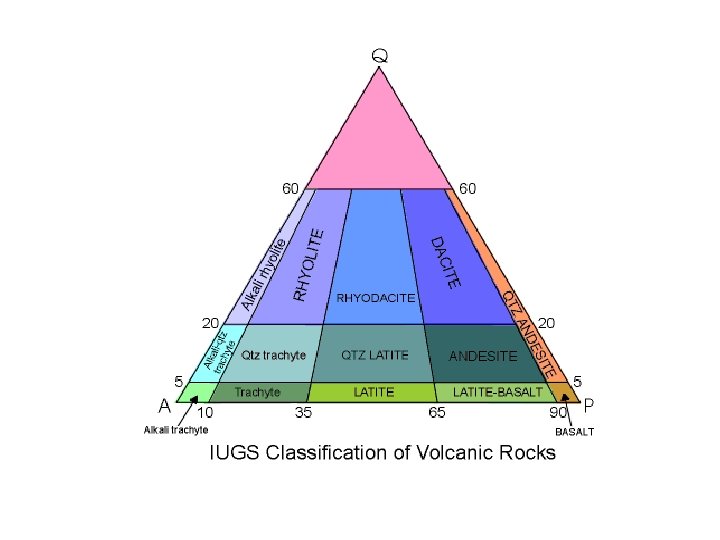
Classification of Igneous Rocks • Rock with 70% X, 20% Y, 10% Z • Plot mineral composition of triangle diagram Figure 2 -1 a. Method #1 for plotting a point with the components: 70% X, 20% Y, and 10% Z on triangular diagrams. An Introduction to Igneous and Metamorphic Petrology, John Winter, Prentice Hall.

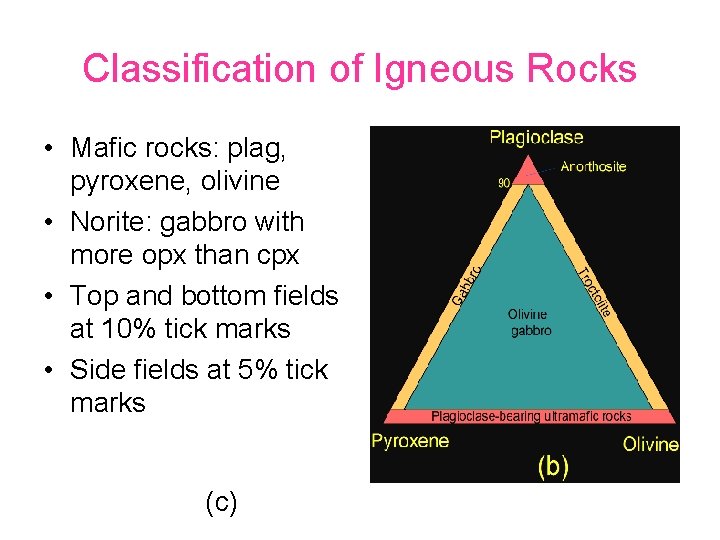
Classification of Igneous Rocks • Mafic rocks: plag, pyroxene, olivine • Norite: gabbro with more opx than cpx • Top and bottom fields at 10% tick marks • Side fields at 5% tick marks (c)




G 225 – Review For Exam 1 Models of the atom Rutherford, Bohr, de. Broglie Electronic configuration Quantum numbers Periodic Table Valence electrons ; oxidationreduction Z and A numbers Isotopes ; stable and radiogenic Bonding Ionic Covalent Vander. Waals Metallic Polar versus non-polar Electronegativity Atomic structures Ionic radii – bond lengths

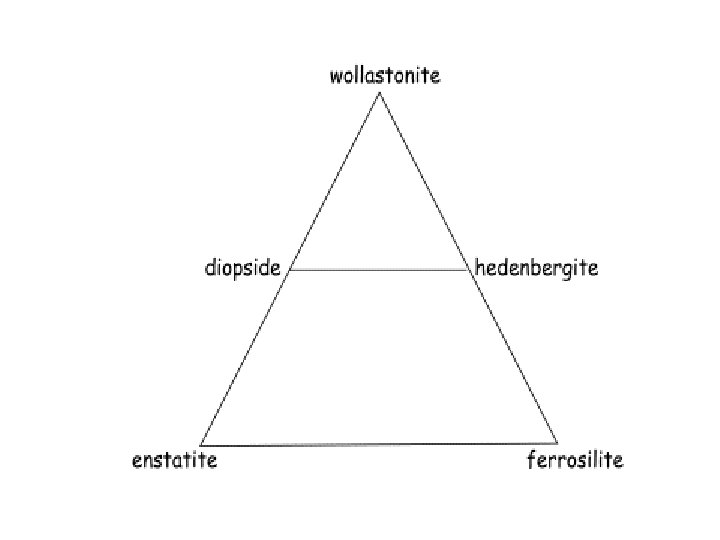
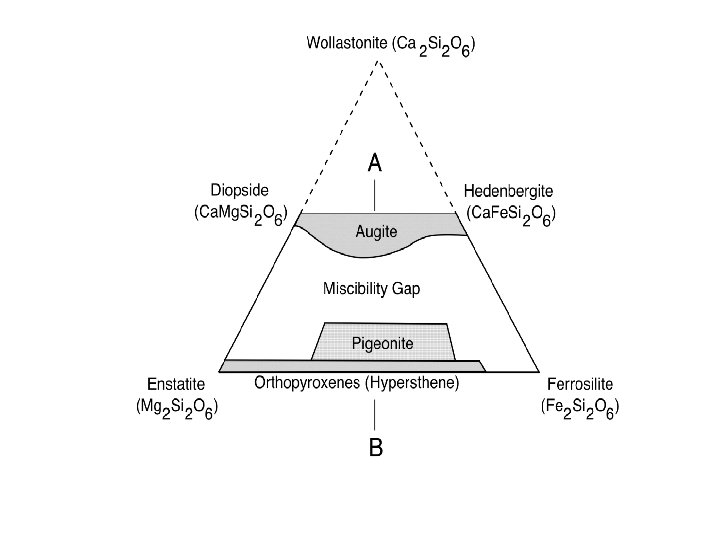
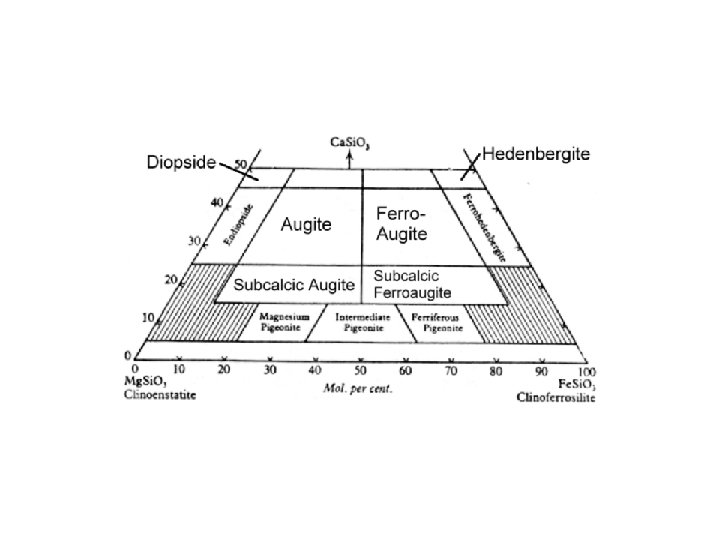
Coordination numbers Hexagonal and cubic closest packing Pauling’s Rules Radicals Isodesmic, anisodesmic, mesodesmic Silicate tetrahedral linkages; subclasses; polymerization Crystal Systems Mineral compositions and plotting procedures; solid solution