Automotive Batteries Automotive Batteries n WHAT AND AUTOMOTIVE

Automotive Batteries

Automotive Batteries n WHAT AND AUTOMOTIVE BATTERY DOES n A battery supplies power to the starter and ignition system to start the engine. n A battery also supplies the extra power necessary when the vehicle's electrical load exceeds the supply from the charging system.

Automotive Batteries n Automotive batteries acts as a voltage stabilizer in the electrical system. n An automotive battery converts chemical energy into electrical energy n An automotive battery evens out voltage spikes and prevents them from damaging other components in the electrical system

Automotive Batteries n What the standby battery does: n Supplies electrical power to critical systems in the event of a power outage. n Acts as a voltage stabilizer, to smooth out fluctuations in the electrical generation systems. n Temporarily holds large electrical loads as electric utilities switch from one generation system to another.

How a Battery is Made n Batteries are made of five basic components: n 1. A resilient Plastic container. n 2. Positive and negative internal plates made of lead.
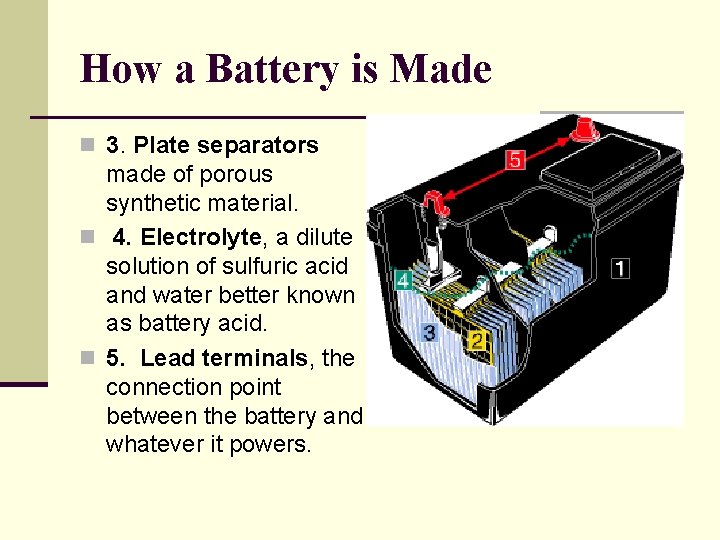
How a Battery is Made n 3. Plate separators made of porous synthetic material. n 4. Electrolyte, a dilute solution of sulfuric acid and water better known as battery acid. n 5. Lead terminals, the connection point between the battery and whatever it powers.
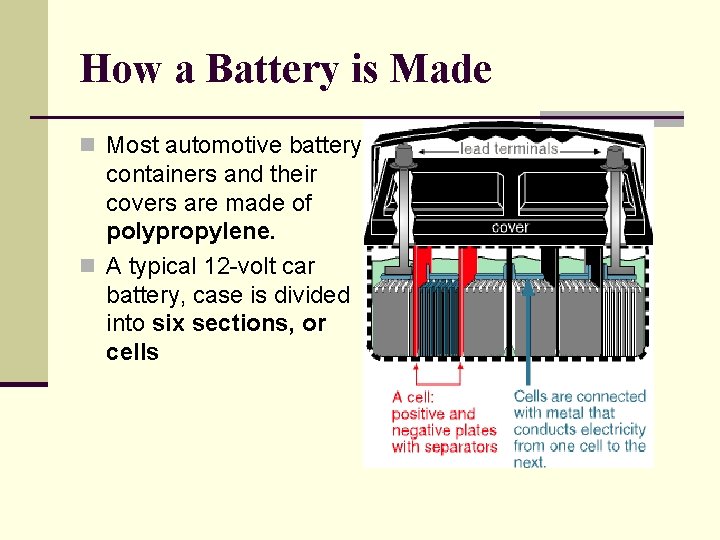
How a Battery is Made n Most automotive battery containers and their covers are made of polypropylene. n A typical 12 -volt car battery, case is divided into six sections, or cells
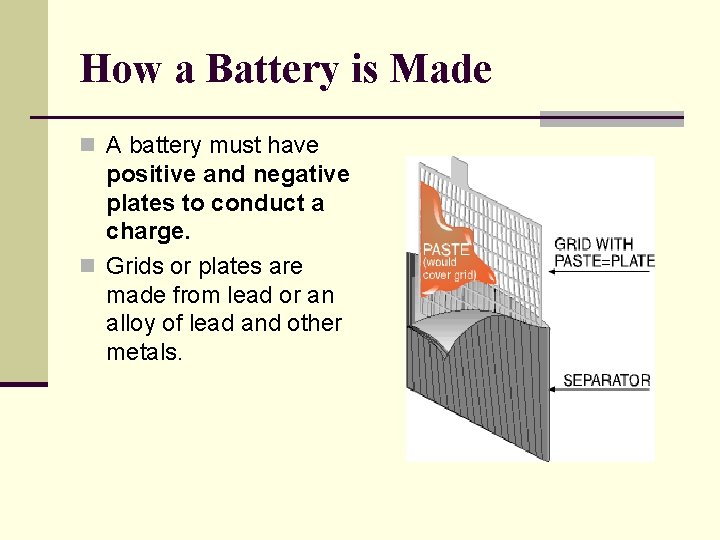
How a Battery is Made n A battery must have positive and negative plates to conduct a charge. n Grids or plates are made from lead or an alloy of lead and other metals.

How a Battery is Made n A mixture of lead oxide -- which is powdered lead, sulfuric acid and water is applied to the grids n Expander material made of powdered sulfates is added to the paste to produce negative plates n The expander material is not added to the positive plates.
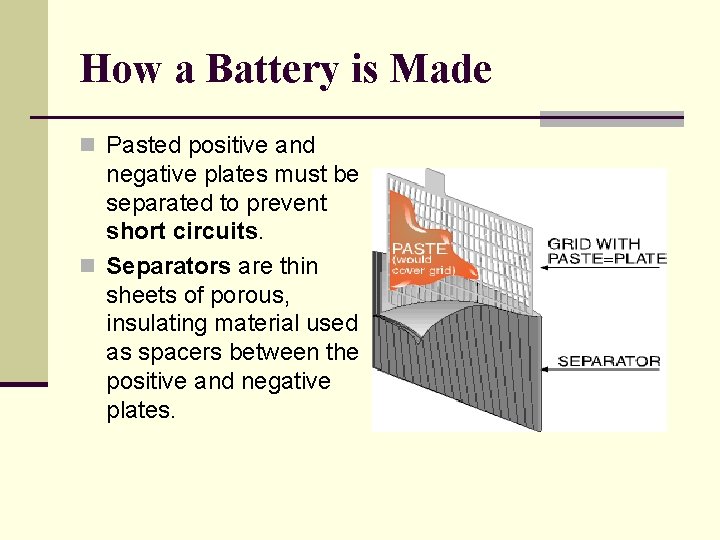
How a Battery is Made n Pasted positive and negative plates must be separated to prevent short circuits. n Separators are thin sheets of porous, insulating material used as spacers between the positive and negative plates.
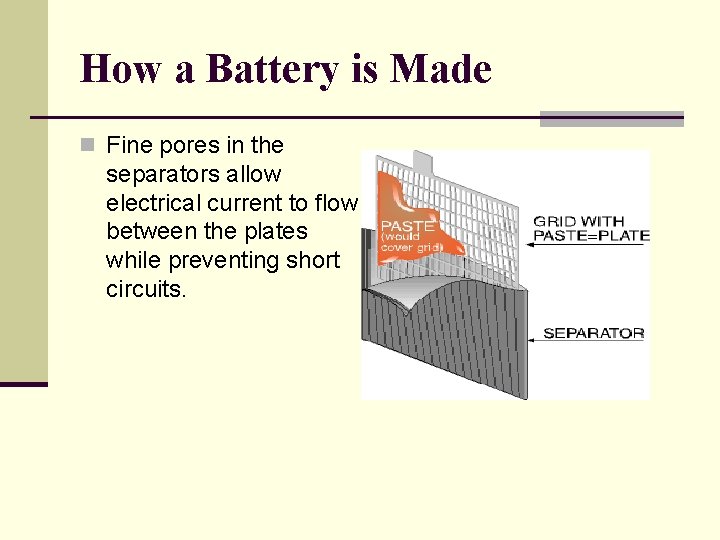
How a Battery is Made n Fine pores in the separators allow electrical current to flow between the plates while preventing short circuits.

How a Battery is Made n A positive plate is paired with a negative plate and a separator to make an element n There is one element per battery cell n Elements are dropped into the cells in the battery case n The cells are connected with a metal that conducts electricity.
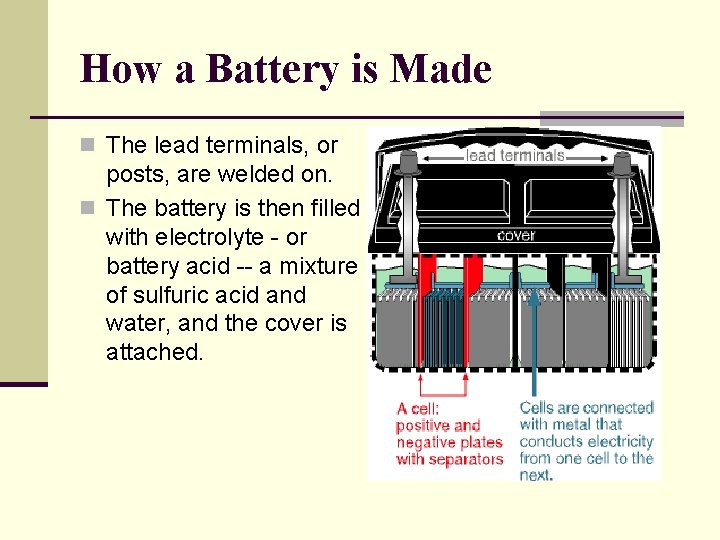
How a Battery is Made n The lead terminals, or posts, are welded on. n The battery is then filled with electrolyte - or battery acid -- a mixture of sulfuric acid and water, and the cover is attached.

How a Battery is Made n After a battery is made its is n Checked for leaks. n Charged for several hours. n Labels added

Battery Quiz 1. What supplies power to the ignition and the starting system A. Starter motor n B. Battery n C. Alternator n D. None of the above n

Battery Quiz 2. What component acts as a voltage stabilizer in the electrical system? 1. Voltage regulator n 2. Current regulator n 3. Battery n 4. Alternator n

Battery Quiz 3. How many cell is in an automotive battery A. 12 n B. 6. n C. 4 n D. 2 n

Battery Quiz 4. Each cell of an automotive battery will supply how many volts? A. 2 n B. 4 n C. 10 n D. 12 n

Battery Quiz 5. A mixture of sulfuric acid and water is called: A. Battery liquid n B. Battery activator n C. Electrical charge n D. Electrolyte n

Battery Quiz 6. There is how many elements element per battery cell? A. 1 n B. 2 n C. 4 n D. 6 n

Battery Quiz 7. A battery must have _______ to conduct electricity. A. Negative plates only n B. Positive plates only n C. Positive and Neutral plates n D. Positive and Negative plates n

Battery Quiz 8. Most automotive battery containers and their covers are made of: A. Steel n B. Wood n C. Soft plastic n D. polypropylene n

Battery Quiz 9. After a battery is made its is: A. Charged for several hours n B. Check for leaks n C. Has labels added n D. All the above n

Battery Quiz 10. A _____ supplies the extra power necessary when the vehicle's electrical load exceeds the supply from the charging system. n A. Alternator n B. Starter n C. Fuel cell n D. Battery

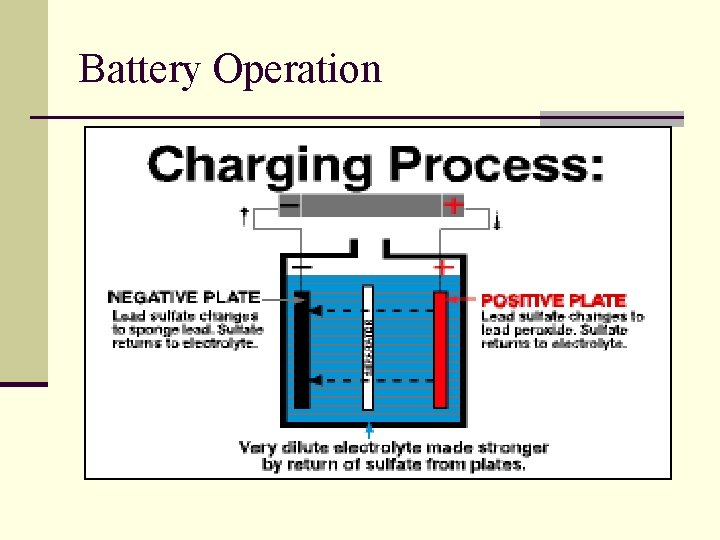
Battery Operation n A battery convert chemical energy into electrical energy n When an atom loses and electron it become a positively charge Ion n When am atom gains an electron it become a negatively charge ion. n Battery produce Voltage and Amperage. Voltage is defined and electrical pressure n Amperage is defined as electron flow from one atom to another. n

Battery Operation n When you have and abundance of negatively charge ions and a abundance of positively charged ions connected together in an electrical circuit current will begin to flow. n The larger the difference between the positive ions and the negative ions the greater the voltage or electrical pressure difference. n The greater the flow electrons from one atom to another the greater the current flow

Battery Operation n Battery have two different ratings. n 1. Cold cranking amperage (CCA): Is the ability of a battery to maintain 7. 3 volts for 30 seconds at 0 degrees F n 2. Reserve Capacity (RC): Is the ability of a battery to supply 10. 5 volts at 25 amps at 80 degrees F.

Battery Operation n A battery stores electricity for future use. n A battery develops voltage from the chemical reaction produced when two unlike materials, such as the positive and negative plates, are immersed in electrolyte, a solution of sulfuric acid and water. n In a typical lead-acid battery, the voltage is approximately 2 volts per cell, for a total of 12 volts.

Battery Operation n Electricity flows from the battery as soon as there is a circuit between the positive and negative terminals. This happens when any load that needs electricity, such as the radio, is connected to the battery.

Battery Operation

Battery Operation n When a battery is discharge the plates have the same amount of electrical charge. n As a battery discharges, the lead plates become more chemically alike, the acid becomes weaker and less voltage is produced.

Battery Operation n You can recharge a discharged battery by feeding electrical current back into the battery. n A full charge restores the chemical difference between the plates and leaves the battery ready to deliver its full power.
Battery Operation

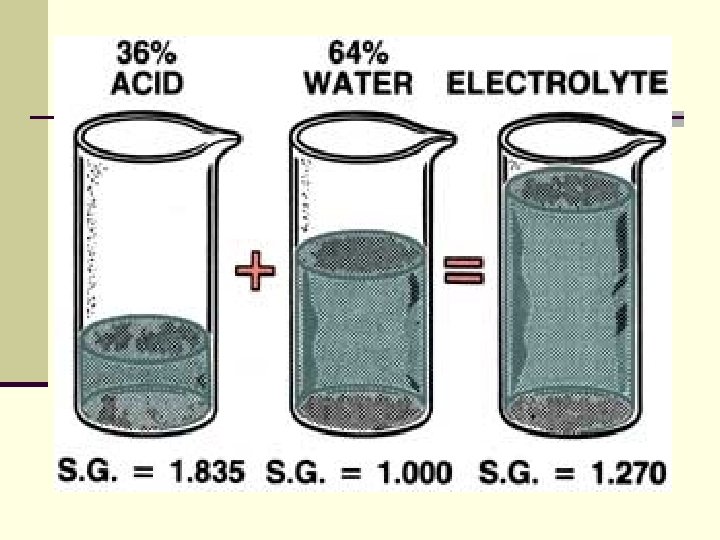
Batteries n SPECIFIC GRAVITY Specific gravity means exact weight. n A "Hydrometer" or a "Refractometer" compares the exact weight of electrolyte with that of water. n Strong electrolyte in a charged battery is heavier than weak electrolyte in a discharged battery. n By weight, the electrolyte in a fully charged battery is about 36% acid and 64% water.

Batteries n The specific gravity of water is 1. 000. n Acid is 1. 835 times heavier than water, so its specific gravity is 1. 835. n The electrolyte mixture of water and acid has a specific gravity of 1. 270, usually stated as "twelve and seventy. "

Battery Operation n This unique process of discharging and charging in the lead-acid battery means that energy can be discharged and restored over and over again. n This is what's known as the cycling ability in a battery.

Battery Quiz 1. You can recharge a discharged battery by: A. Turning the headlights on n B. Replacing the electricity that has been removed n C. Removing the positive battery cable n D. Replacing the Negative battery cable n

Battery Quiz 2. Technician A says a fully charge battery should have a specific gravity of: A. 1. 130 n B. 1. 220 n C. 1. 260 n D. 1. 270 n

Battery Quiz 3. What tool is used to measure battery specific gravity? A. Milometer n B. Micrometer n C. Kilometer n D. Hydrometer n

Battery Quiz 4. Electrolyte is a mixture of sulfuric acid and _______ : A. Hydrogen n B. Carbon n C. Sulfuric carbon n D. Water n

Battery Quiz 5. This unique process of discharging and charging a lead-acid battery is called: A. Discharging n B. Cycling n C. Overcharging n D. None of the above n

Batteries Battery are rated in both cold cranking amps and reserve capacity. Cold Cranking Amps is the amount of amperage a battery can supply at 0 Degrees while maintaining 9. 6 volts for 30 seconds Reserve capacity: The amount of time a battery can supply 10 volts at 25 amps when not being charged

Batteries n When a battery becomes discharged it must be recharged. n A battery can be charged by the vehicle alternators or by an external battery charger n When connecting or disconnecting a battery charger care should be taken not to create a spark.

Batteries n When connecting or disconnecting a battery charger care should be taken not to create a spark. Hydrogen and oxygen can explode n Spark will cause a battery to explode n The gasses created during charging are explosive n

Batteries n When charging a battery always connect the positive cable first and the negative cable last n When removing battery cables disconnect the Negative cable first and the Positive cable last. n Most automotive manufactures recommend disconnecting a battery before its recharged.

- Slides: 47