Automatic pipettes use and calibration O Automatic pipets

Automatic pipettes use and calibration

O Automatic pipets are used in the biological, biochemical, and microbiological teaching and research laboratories to accurately transfer small liquid volumes. Glass pipets which are used in chemical laboratories are not highly accurate for volumes less than 1 milliliter (1 ml), but the automatic pipets are both accurate and precise. O Each pipet can be set to transfer any volume within its own volume range. Although the complete set of seven Pipetman models covers the volume range from 0. 1 l to 5000 l (5 ml), you will most often use the P-20, P-200, and P-1000 models which are recommended for volume transfers of from 2 to 20 l, 50 to 200 l, and 100 to 1000 l (1 ml), respectively

Pipetman model available


Using a Micropipettor: O Never exceed the upper or lower limits of these pipettors. O Set the desired volume by turning the centrally located rings clockwise to increase volume or counterclockwise to decrease volume O The examples below show to read the micropipettor:

O Place a tip on the discharge end of the pipettor. O The plunger will stop at two different positions when it is depressed. The first of these stopping points is the point of initial resistance and is the level of depression that will result in the desired volume of solution being transferred. O The second stopping point can be found when the plunger is depressed beyond the initial resistance until it is in contact with the body of the pipettor. At this point the plunger cannot be further depressed. This second stopping point is used for the complete discharging of solutions from the plastic tip. You should not reach this second stop when drawing liquid into the pipettor, only when expelling the last drop.

Methods of pipetting O Forward pipetting This is the most frequently used technique. In forward pipetting, an exactly set volume of liquid is aspired to the tip and then it is delivered to a new vessel. This technique is recommended for pipetting of diluted aqueous solutions, buffers, diluted acids and bases. O Reverse technique In reverse technique, a larger volume of solution is aspired to the tip. Then, an exact volume is delivered (and some liquid remains in the tip). This technique is suitable for very viscous or volatile liquids, biologic fluids, foaming solutions and for measuring very small volumes.

Forward method O Depress the plunger until you feel the initial resistance and insert tip into the solution, just barely below the surface of the liquid and not as deep as possible. O Carefully and slowly release plunger

O Discharge the solution into the appropriate container by depressing plunger. This time, depress the plunger to the point of initial resistance, wait one second, and then continue pressing the plunger as far as it will go in order to discharge the entire volume of solution. O Remove tip by pressing down on the tip discarder

Reverse method O Push the button to the second position. O Dip the tip 2– 5 mm bellow the level of the solution. Slowly aspire the solution to the tip. O Remove the tip slowly from the solution. If necessary, wipe droplets from the external surface of the tip. O Deliver the liquid to a new vessel, pushing the button to the first position only. O Hold the button in the first position and remove the tip from the vessel. O The liquid remaining in the tip can be returned to the stock vessel or thrown away.

NOTES: O Never point a pipettor up. This may cause liquid to run down into the pipettor destroying it. O When withdrawing liquids with the pipettor, always release the plunger slowly. This prevents liquid from rushing into the end of the pipette and clogging it up. This is especially important with large volume pipettors (200 -1000 μl). O Be sure you use the proper size tip for each pipettor O Always use a new tip for each different liquid. O Use the correct pipettor for the volume that is to be dispensed. Never use the 200 -1000 μl pipette to dispense volumes below 200 μl. going below or above the range of the micropipettor may damage the instrument.

O withdraw the pipet from the receiving vessel carefully, sliding the tip along the wall of the vessel. Holding the tip against the side of vessel is especially important when transferring small volumes of liquid. O Remove the tip from the sample liquid. No liquid should remain on the OUTSIDE of the tip. Wipe away any droplets on the outside of the tip with a lint-free tissue, such as KIMWIPES, but only wipe droplets from the side of the tip. NEVER TOUCH THE TIP ORIFICE (opening) or you may absorb part of your sample

O Pre-rinsing minimizes intertip or intratip variances due to any coating of the inner tip wall. When pipetting some solution, such as serum or other protein-containing solutions and organic solvents, a significant film may be retained on the inside of the wall of the disposable tips, resulting in an excessive volume error. Since this film remains relatively constant in successive pipettings with the same tip, excellent precision may be obtained by discarding the first filling of the tip and refilling the tip a second time or more times to use as the sample(s). This procedure is recommended when critical reproducibility (precision) is required.

O When pipetting concentrated acids of highly corrosive solutions such as halide salts, it is recommended that the pipet be disassembled after use to inspect the piston, shaft, and seal assemblies and clean them, if necessary. O Air bubbles in the tip can greatly reduce pipetting accuracy. If an air bubble is trapped within the tip during intake, dispense the sample into the original vessel, check the tip immersion depth, and pipet more slowly. If an air bubble appears a second time, discard the tip and use a new one.

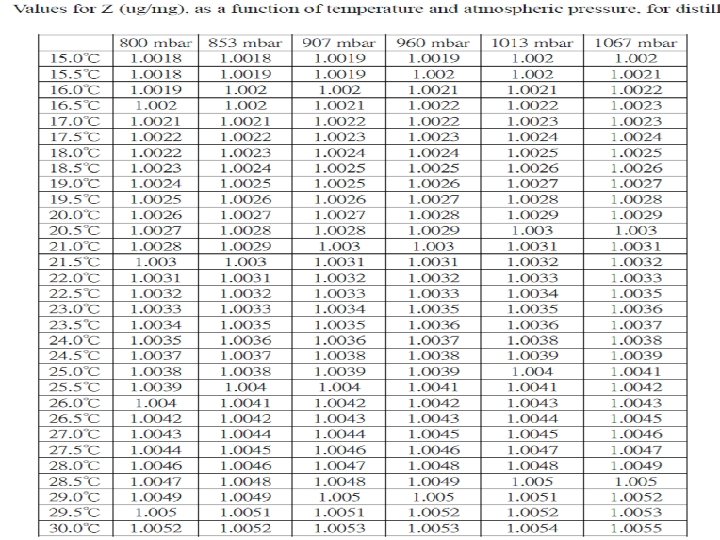
Pipette calibration Principle: O Under a constant temperature and atmospheric pressure, the density of distilled water is constant. O The volume of water can be determined by weighting dispensed water. O The calibration of pipette is carried out determining the volume of water, the accuracy of measurements is effected by ambient temperature, atmospheric pressure and relative humidity. These factors are usually combined to give the Z factor, used in calculation of volume of water. O Then the calculated volume of water is compared with theoretical volume to determine the accuracy and precision of the pipette.

Material and equipment: (1) pipette and tips (2) 50 ml beaker and plastic medicine cup (3) distilled water (4) temperature meter ( ± 0. 1℃ ) (5) Analytical balance ( ± 1. 0 mg ) (6) Atmospheric pressure meter

Procedure 1. Determine the water temperature and record it. 2. Place a beaker filled with distilled water into analytic balance and close the door of balance waiting for equilibrium of inner vapor 3. Place a plastic medicine cup on the pan and adjust the weight to zero. 4. Put a tip onto the pipette and set the volume which is to be tested. 5. Pre-rinse the tip: aspirate and dispense the setting volume three times and press the push – button on the second stop to remove any remaining liquid. 6. Press the push-button to the first positive stop. Hold the pipette vertically, immerse the tip in the liquid and release the push button slowly and smoothly to aspirate the liquid.

7. 8. 9. 10. 11. 12. 13. 14. 15. Wait one second and withdraw the tip from the liquid. Wipe any droplets away from the outside of the tip using a kimwipe. Place the end of the tip against the inside wall of the plastic cup at an angle of 10 -40 °. Press the push-button smoothly to the first stop. Wait one second, change new site and press the push-button on the second stop. Keeping the push-button press to the end, remove the pipette by drawing the tip along the inside surface of the plastic cup and release the push button. Close the door of balance and record the value on the balance display after it has stabilized. Repeat step (6) to (12) 14 times Eject the tip. Pipette should be calibrated every three months routinely.


Calculation 1. Calculate the difference in weight between one readin an the next 2. Convert the weight unit of measured value into the volume unit of measured value using the following formula: Volume ( ml or ul ) = Weight ( mg or ug ) x Z Z value : conversion factor, which is conversion of density

3. Calculate the average ( Mean ), accuracy, standard deviation (S. D. ) and imprecision ( C. V. ) using the following formula:
- Slides: 22