Authenticity of Sugar Speciality Sugars Analysis of water




![Beet/Cane Sugar Blending (2 5 samples) Cane/Beet Sugar Mixture: Content of Cane Sugar [%] Beet/Cane Sugar Blending (2 5 samples) Cane/Beet Sugar Mixture: Content of Cane Sugar [%]](https://slidetodoc.com/presentation_image_h2/fb292514a0b8fab8b8dd6083fff67bad/image-10.jpg)

![Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Cane Sugar: Sucrose [H+] H 2 Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Cane Sugar: Sucrose [H+] H 2](https://slidetodoc.com/presentation_image_h2/fb292514a0b8fab8b8dd6083fff67bad/image-16.jpg)






- Slides: 25

Authenticity of Sugar & Speciality Sugars Analysis of water by isotopes in order to determine the authenticity of sugar & speciality sugars – Determination of carbohydrate source & geographical origin 10 th EUROFOODWATER Conference on Water in Food Prague, Czech Republic, 21 st September 2018 Dr Andreas Georg Degenhardt

Introduction / Purpose Scientific Interest § new strategies/methods of instrumental analysis § knowledge about biological and geographical relationships and differences in natural sources of sucrose § relationships between sugar as raw material and processed speciality sugars asparagus eggs wine caviar wood Developments in the analytical determination of source/origin during the last 20 years 3

Agenda I. Photosynthesis / stable isotopes / principle of determination II. Determination of plant sources for sucrose III. Determination of geographical origin IV. Speciality Sugars: Cane (Crystal) vs. Invert vs. Burnt Sugar V. Summary & Conclusions 4

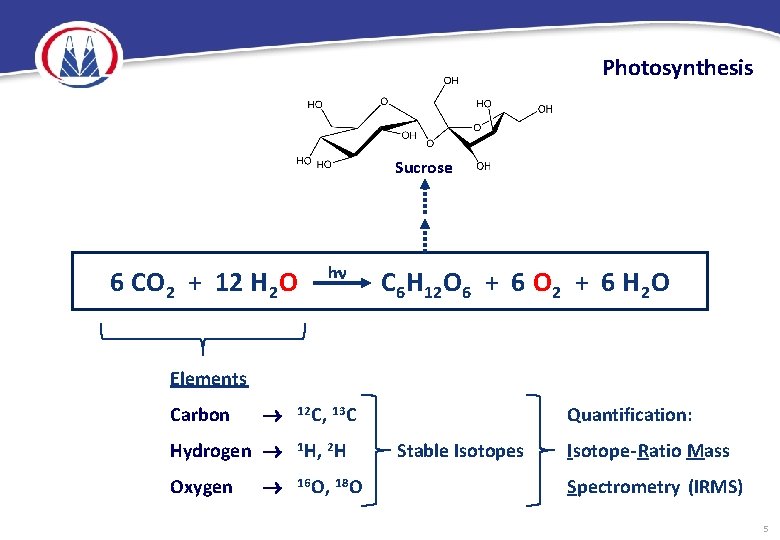
Photosynthesis Sucrose 6 CO 2 + 12 H 2 O h C 6 H 12 O 6 + 6 O 2 + 6 H 2 O Elements Carbon Hydrogen Oxygen 12 C, 13 C 1 H, 2 H 16 O, 18 O Quantification: Stable Isotopes Isotope-Ratio Mass Spectrometry (IRMS) 5

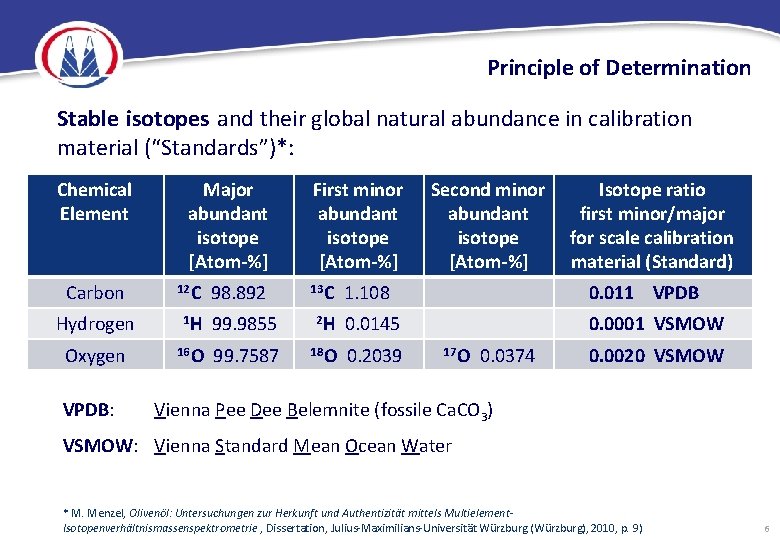
Principle of Determination Stable isotopes and their global natural abundance in calibration material (“Standards”)*: Chemical Element Major abundant isotope [Atom-%] Carbon 12 C Hydrogen 1 H 99. 9855 Oxygen 16 O 99. 7587 VPDB: 98. 892 First minor abundant isotope [Atom-%] 13 C Second minor abundant isotope [Atom-%] Isotope ratio first minor/major for scale calibration material (Standard) 1. 108 0. 011 VPDB 2 H 0. 0145 0. 0001 VSMOW 18 O 0. 2039 17 O 0. 0374 0. 0020 VSMOW Vienna Pee Dee Belemnite (fossile Ca. CO 3) VSMOW: Vienna Standard Mean Ocean Water * M. Menzel, Olivenöl: Untersuchungen zur Herkunft und Authentizität mittels Multielement. Isotopenverhältnismassenspektrometrie , Dissertation, Julius-Maximilians-Universität Würzburg (Würzburg), 2010, p. 9) 6

Principle of Determination 13 CO 2 / 12 CO 2 Sugar Isotopologs 2 H 1 H / 1 H 1 H 12 C 18 O 16 O 13 C, 12 C 2 H, 1 H / 12 C 16 O 18 O, 16 O Sample 2 H/1 H [‰] 18 O/16 O [‰] 13 C/12 C [‰] Beet Sugar Germany (Rhineland) -66. 5 28. 6 -27. 4 Cane Sugar Brasil (São Paulo State) -8. 6 34. 2 -11. 7 (VSMOW) (VPDB) 7

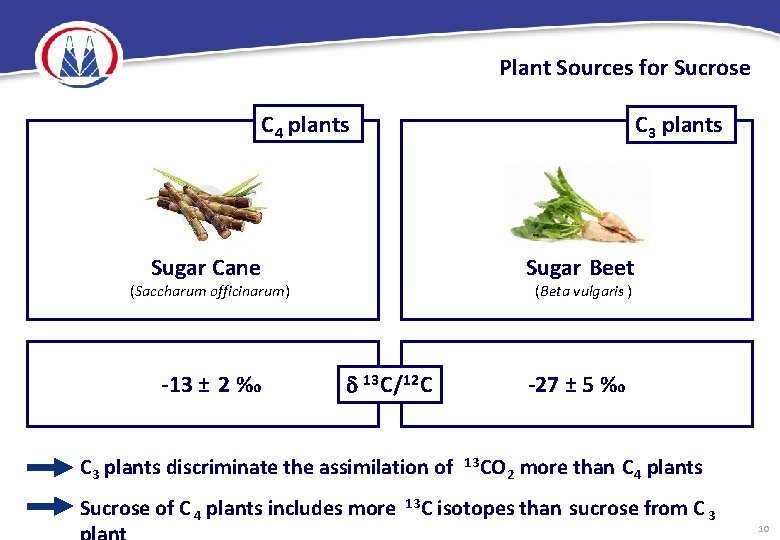
Plant Sources for Sugar Beet C 3 plants (Beta vulgaris ) Maple (Acer Saccharum, Acer nigrum) Rice (Oryza sativa) Sugar Cane C 4 plants (Saccharum officinarum) Corn (Zea mays) Sorghum (Sorghum bicolor) Palm (Phoenix silvestris , Borassus flabilliformis, Coccus nucifera, Caryota urens) 9

Plant Sources for Sucrose C 4 plants C 3 plants Sugar Cane Sugar Beet (Saccharum officinarum) -13 ± 2 ‰ (Beta vulgaris ) 13 C/12 C -27 ± 5 ‰ C 3 plants discriminate the assimilation of Sucrose of C 4 plants includes more 13 CO 2 more than C 4 plants isotopes than sucrose from C 3 10

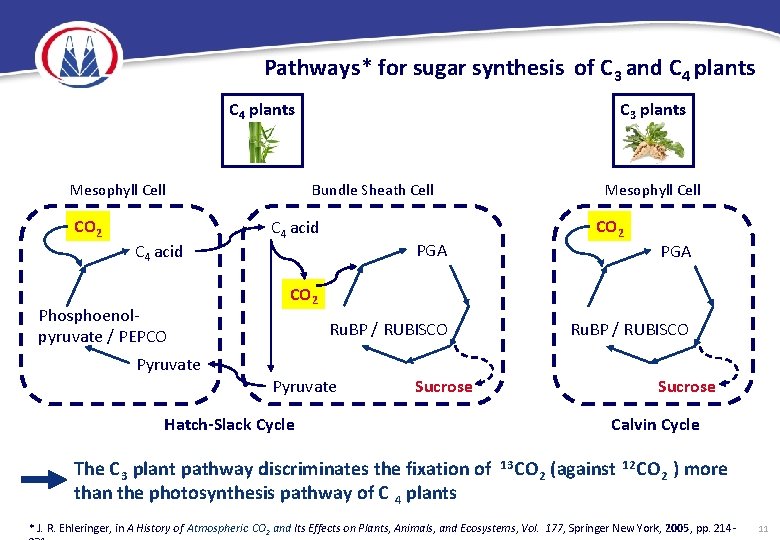
Pathways* for sugar synthesis of C 3 and C 4 plants Mesophyll Cell CO 2 C 4 acid Phosphoenolpyruvate / PEPCO Pyruvate C 3 plants Bundle Sheath Cell C 4 acid Mesophyll Cell CO 2 PGA CO 2 Ru. BP / RUBISCO Pyruvate Ru. BP / RUBISCO Sucrose Hatch-Slack Cycle The C 3 plant pathway discriminates the fixation of than the photosynthesis pathway of C 4 plants Calvin Cycle 13 CO 2 (against 12 CO 2 ) more * J. R. Ehleringer, in A History of Atmospheric CO 2 and Its Effects on Plants, Animals, and Ecosystems, Vol. 177, Springer New York, 2005 , pp. 214 - 11
![BeetCane Sugar Blending 2 5 samples CaneBeet Sugar Mixture Content of Cane Sugar Beet/Cane Sugar Blending (2 5 samples) Cane/Beet Sugar Mixture: Content of Cane Sugar [%]](https://slidetodoc.com/presentation_image_h2/fb292514a0b8fab8b8dd6083fff67bad/image-10.jpg)
Beet/Cane Sugar Blending (2 5 samples) Cane/Beet Sugar Mixture: Content of Cane Sugar [%] Pure Cane Mixtures of cane/beet sugar are distinguishable by Pure Beet 13 C/12 C isotope analysis Exact quantification is difficult, an “estimation” of cane/beet ratio is possible 12

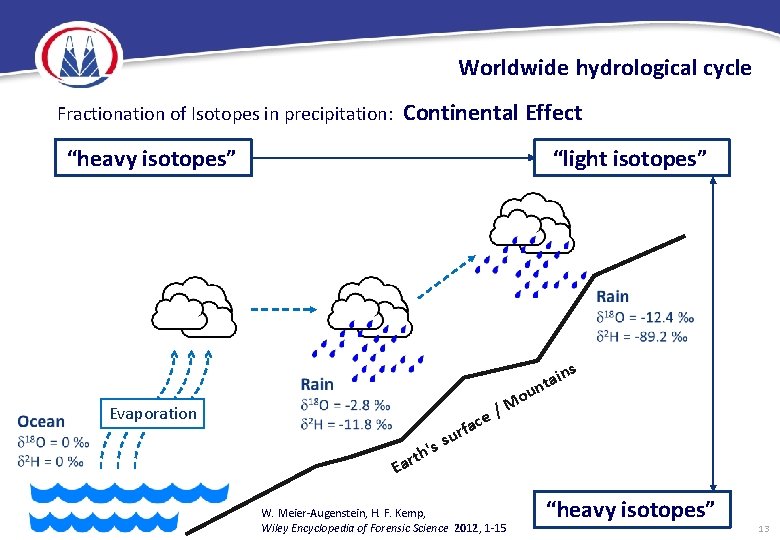
Worldwide hydrological cycle Fractionation of Isotopes in precipitation: Continental Effect “heavy isotopes” “light isotopes” ns i a nt u Evaporation 's th r a E fa sur ce o M / W. Meier-Augenstein, H. F. Kemp, Wiley Encyclopedia of Forensic Science 2012 , 1 -15 “heavy isotopes” 13

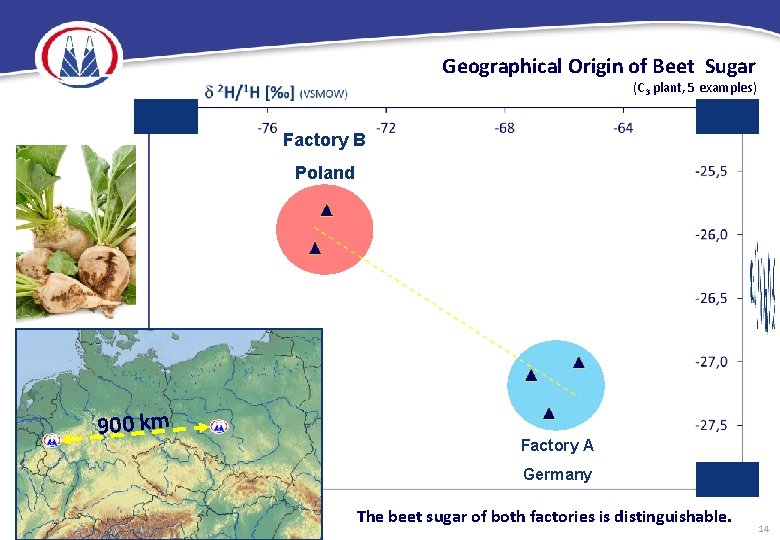
Geographical Origin of Beet Sugar (C 3 plant, 5 examples) Factory B Poland 900 km Factory A Germany The beet sugar of both factories is distinguishable. 14

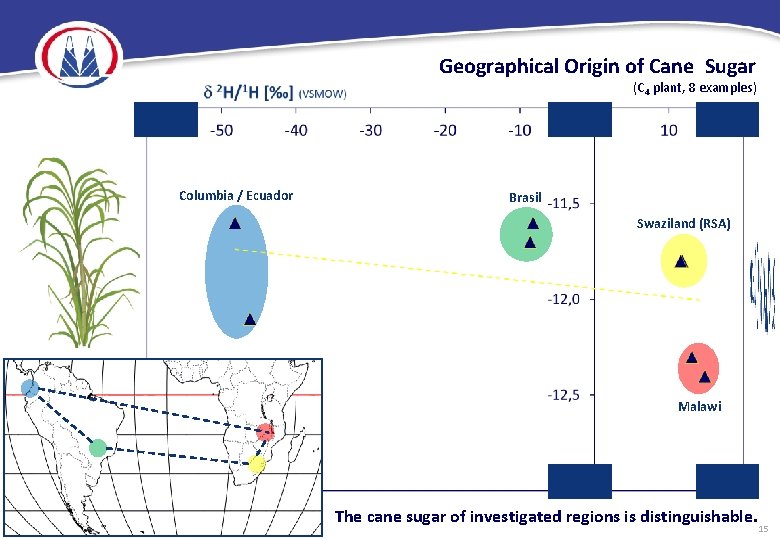
Geographical Origin of Cane Sugar (C 4 plant, 8 examples) Columbia / Ecuador Brasil Swaziland (RSA) Malawi The cane sugar of investigated regions is distinguishable. 15

Speciality Sugars Elsdorf, Germany Manufacturing Invert & Burnt Sugar from Cane Sugar (Cane vs. Invert vs. Burnt Sugar) Can we determine the plant source and the geographical origin of invert and burnt sugar syrups in the same way? Origin of Cane Sugar Columbia Brazil Swaziland 16

Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Cane Sugar Invert Sugar Syrup Burnt Sugar Syrup 17
![Speciality Sugars Cane vs Invert vs Burnt Sugar Cane Sugar Sucrose H H 2 Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Cane Sugar: Sucrose [H+] H 2](https://slidetodoc.com/presentation_image_h2/fb292514a0b8fab8b8dd6083fff67bad/image-16.jpg)
Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Cane Sugar: Sucrose [H+] H 2 O Invert Sugar Syrup: Glucose Cane Sugar Fructose Invert Sugar Syrup 18

Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Invert Sugar Syrup: Sucrose + Glucose + Fructose T = 140 °C t = 120 min Burnt Sugar Syrup: Melanoidins + Organic acids + HMF + … Invert Sugar Syrup Burnt Sugar Syrup 19

Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Swaziland Cane Sugar Columbia Invert Sugar Syrup Brazil Burnt Sugar Syrup 20

Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) Swaziland Columbia Brazil Determination 13 C/12 C, 2 H/1 H and 18 O/16 O by IRMS analysis 2 H/1 H (D/H) analysis of ethanol after yeast fermentation 21

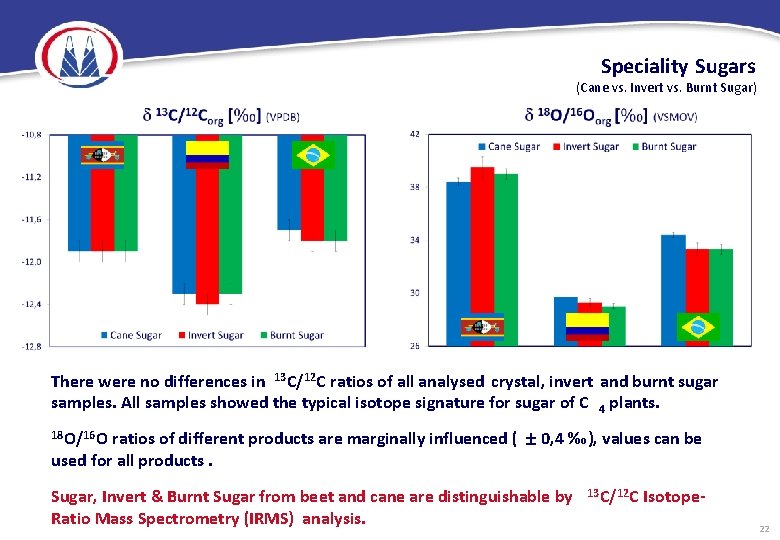
Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) There were no differences in 13 C/12 C ratios of all analysed crystal, invert and burnt sugar samples. All samples showed the typical isotope signature for sugar of C 4 plants. ratios of different products are marginally influenced ( 0, 4 ‰), values can be used for all products. 18 O/16 O Sugar, Invert & Burnt Sugar from beet and cane are distinguishable by Ratio Mass Spectrometry (IRMS) analysis. 13 C/12 C Isotope 22

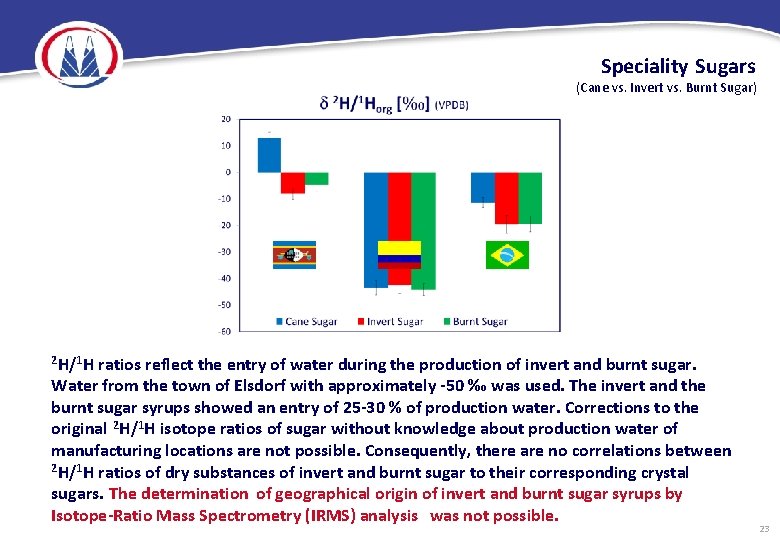
Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) 2 H/1 H ratios reflect the entry of water during the production of invert and burnt sugar. Water from the town of Elsdorf with approximately -50 ‰ was used. The invert and the burnt sugar syrups showed an entry of 25 -30 % of production water. Corrections to the original 2 H/1 H isotope ratios of sugar without knowledge about production water of manufacturing locations are not possible. Consequently, there are no correlations between 2 H/1 H ratios of dry substances of invert and burnt sugar to their corresponding crystal sugars. The determination of geographical origin of invert and burnt sugar syrups by Isotope-Ratio Mass Spectrometry (IRMS) analysis was not possible. 23

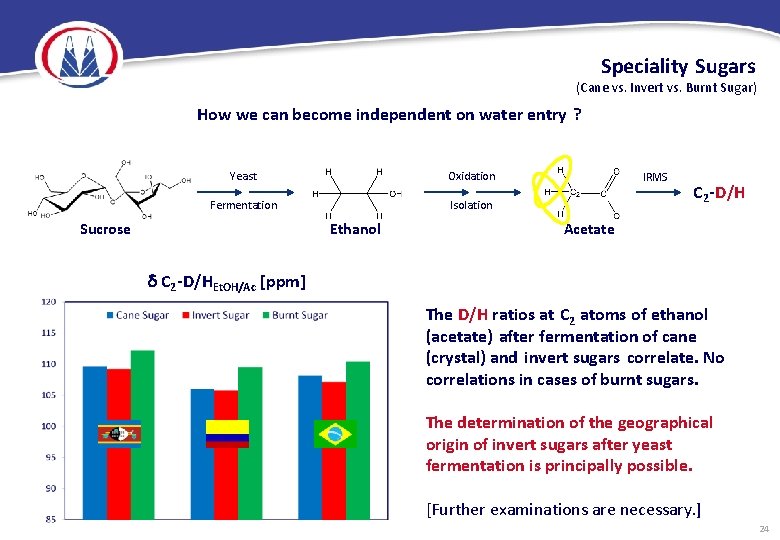
Speciality Sugars (Cane vs. Invert vs. Burnt Sugar) How we can become independent on water entry ? Yeast Oxidation Fermentation Isolation Sucrose Ethanol IRMS C 2 -D/H Acetate C 2 -D/HEt. OH/Ac [ppm] The D/H ratios at C 2 atoms of ethanol (acetate) after fermentation of cane (crystal) and invert sugars correlate. No correlations in cases of burnt sugars. The determination of the geographical origin of invert sugars after yeast fermentation is principally possible. [Further examinations are necessary. ] 24

Authenticity of Sugar Summary & Conclusions I Sucrose from beet and cane are distinguishable by 13 C/12 C Isotope-Ratio Mass Spectrometry (IRMS) analysis caused by different pathways of photosynthesis. Contents of cane and beet in cane/beet sugar mixtures are not exactly quantifiable, but assessable. The geographical origin of sucrose can be determined by IRMS analysis of 18 O/16 O and 2 H/1 H isotope ratios – qualified data base is pre-condition. 25

Authenticity of Speciality Sugars Summary & Conclusions II Sugar, Invert & Burnt Sugar from beet and cane are distinguishable by 13 C/12 C Isotope-Ratio Mass Spectrometry ( IRMS) analysis caused by different pathways of photosynthesis. The 2 H/1 H ratios of analysed invert and burnt sugar syrups showed an entry of production water. Without knowledge about production water of manufacturing locations, the determination of geographical origin of invert and burnt sugar syrups by conventional Isotope-Ratio Mass Spectrometry ( IRMS) analysis was not possible. The D/H ratios at C 2 atoms of ethanol after fermentation of cane (crystal) and invert sugars showed correlations. Taking into consideration of the uncorrected 13 C/12 C , 18 O/16 O ratios in combination with C 2 -D/H ratios after ethanol-fermentation, a differentiation between different origins of crystal and invert sugars is possible. 26

Thank you for listening! 27