Audit of RBC Transfusion in Premature Infants 2001

Audit of RBC Transfusion in Premature Infants 2001 Dr Ho Hing Tung (Paediatrics) Dr Sherman Lee (Clinical Audit) Dr Raymond Chu (Haematology) Pamela Youde Nethersole Eastern Hospital

Objectives n n To assess the compliance of RBC transfusion with new guidelines 2001 To assess the effectiveness of new guidelines in reducing the number of RBC transfusions in premature infants

Transfusion Guidelines n n Beforehand, RBC transfusion according to “Handbook of Blood Transfusion for doctors & nurses PYNEH 1998 2 nd Ed. ” New RBC transfusion guideline was implemented from Jan 2001
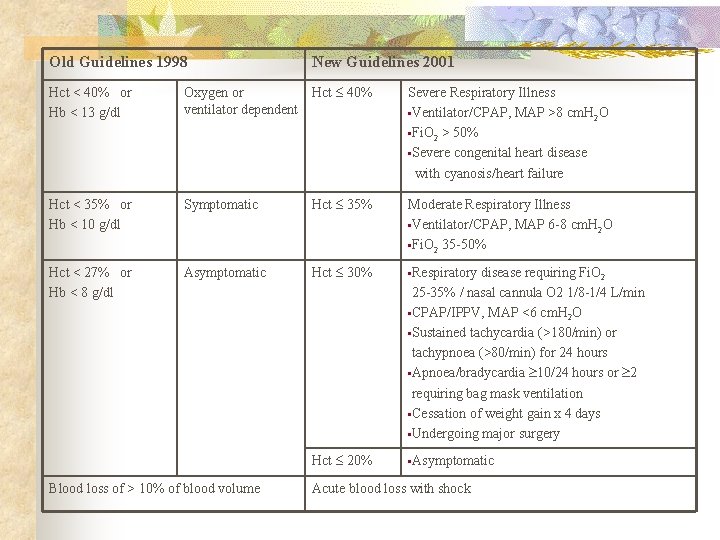
Old Guidelines 1998 New Guidelines 2001 Hct < 40% or Hb < 13 g/dl Oxygen or Hct 40% ventilator dependent Severe Respiratory Illness • Ventilator/CPAP, MAP >8 cm. H 2 O • Fi. O 2 > 50% • Severe congenital heart disease with cyanosis/heart failure Hct < 35% or Hb < 10 g/dl Symptomatic Hct 35% Moderate Respiratory Illness • Ventilator/CPAP, MAP 6 -8 cm. H 2 O • Fi. O 2 35 -50% Hct < 27% or Hb < 8 g/dl Asymptomatic Hct 30% • Respiratory Hct 20% • Asymptomatic Blood loss of > 10% of blood volume disease requiring Fi. O 2 25 -35% / nasal cannula O 2 1/8 -1/4 L/min • CPAP/IPPV, MAP <6 cm. H 2 O • Sustained tachycardia (>180/min) or tachypnoea (>80/min) for 24 hours • Apnoea/bradycardia 10/24 hours or 2 requiring bag mask ventilation • Cessation of weight gain x 4 days • Undergoing major surgery Acute blood loss with shock

Method n Inclusion criteria: n n Inborn baby with birth weight 1500 g Date of birth from 1 Sep 1998 to 31 Aug 2001 Date of discharge from 1 Sep 1998 to 30 Oct 2001 Exclusion criteria: n n n Babies not born in PYNEH All perinatal & neonatal deaths before discharge Infants required transfer out of hospital

Method n Data collection – Retrospective n n All neonatal RBC transfusion episodes were retrieved from blood bank Selection of cases according to inclusion & exclusion criteria Trace all old records Subgroup n n n BW < 1000 g BW 1001 – 1500 g Data collection as listed

Data Collection n Demographic n n n n n Name ID Sex Gestation Birth weight DOB DODay 14 DODischarge Length of stay Age attain 2. 2 kg n Clinical n n n n First Hct % at birth Ventilation Days O 2 Days BPD (36 wks PCA) IVH ( G 3) ROP ( G 3) Total no. /vol. Transfusion at 2 weeks & > 2 weeks to discharge No. of donor exposure

Data Collection n Compliance n n Indication for transfusion Compliance n n Old guideline New guideline

Data Analysis n n n Using SPSS 9. 0 Chi-square tests for discrete variables Student’s t tests for group means Linear regression for prediction estimation of transfusion Differences considered significant at a p value of < 0. 05
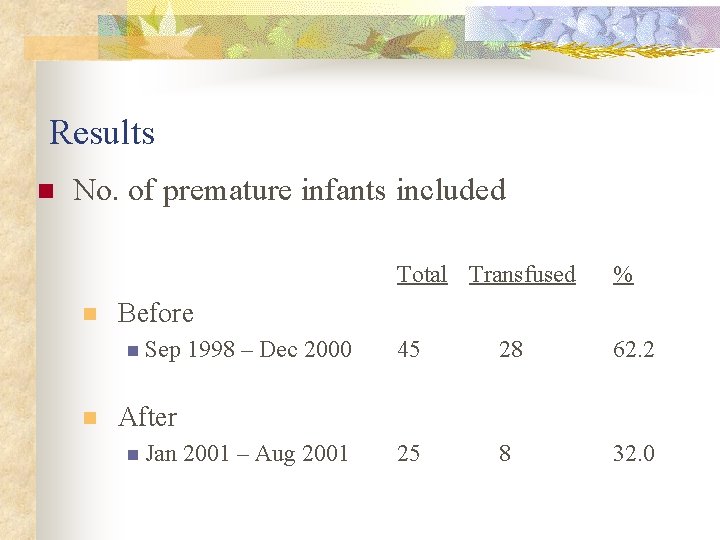
Results n No. of premature infants included n % 1998 – Dec 2000 45 28 62. 2 2001 – Aug 2001 25 8 32. 0 Before n Sep n Total Transfused After n Jan
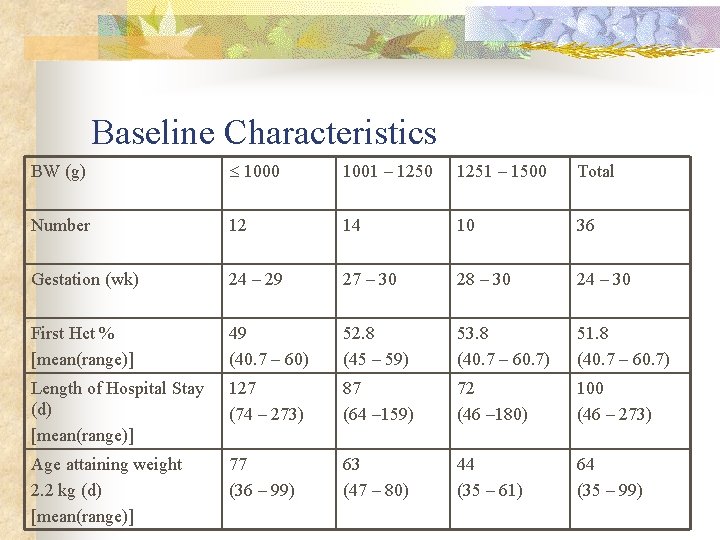
Baseline Characteristics BW (g) 1000 1001 – 1250 1251 – 1500 Total Number 12 14 10 36 Gestation (wk) 24 – 29 27 – 30 28 – 30 24 – 30 First Hct % [mean(range)] 49 (40. 7 – 60) 52. 8 (45 – 59) 53. 8 (40. 7 – 60. 7) 51. 8 (40. 7 – 60. 7) Length of Hospital Stay (d) [mean(range)] 127 (74 – 273) 87 (64 – 159) 72 (46 – 180) 100 (46 – 273) Age attaining weight 2. 2 kg (d) [mean(range)] 77 (36 – 99) 63 (47 – 80) 44 (35 – 61) 64 (35 – 99)
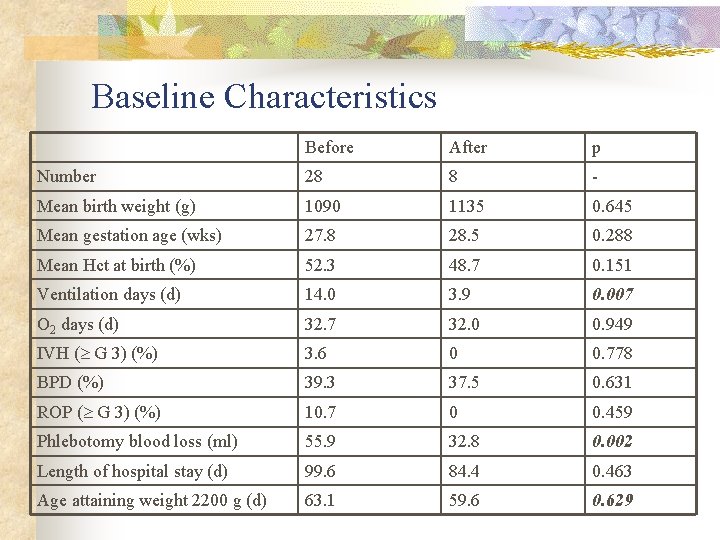
Baseline Characteristics Before After p Number 28 8 - Mean birth weight (g) 1090 1135 0. 645 Mean gestation age (wks) 27. 8 28. 5 0. 288 Mean Hct at birth (%) 52. 3 48. 7 0. 151 Ventilation days (d) 14. 0 3. 9 0. 007 O 2 days (d) 32. 7 32. 0 0. 949 IVH ( G 3) (%) 3. 6 0 0. 778 BPD (%) 39. 3 37. 5 0. 631 ROP ( G 3) (%) 10. 7 0 0. 459 Phlebotomy blood loss (ml) 55. 9 32. 8 0. 002 Length of hospital stay (d) 99. 6 84. 4 0. 463 Age attaining weight 2200 g (d) 63. 1 59. 6 0. 629
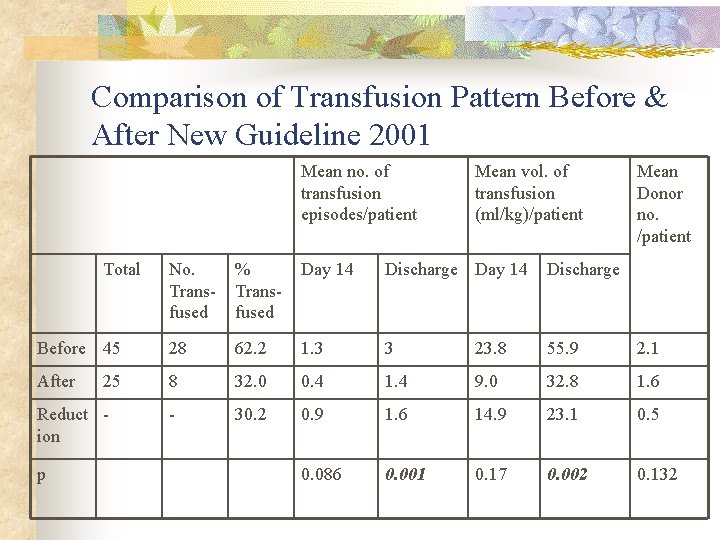
Comparison of Transfusion Pattern Before & After New Guideline 2001 Mean no. of transfusion episodes/patient Total Mean vol. of transfusion (ml/kg)/patient Mean Donor no. /patient No. Transfused % Transfused Day 14 Discharge Before 45 28 62. 2 1. 3 3 23. 8 55. 9 2. 1 After 8 32. 0 0. 4 1. 4 9. 0 32. 8 1. 6 - 30. 2 0. 9 1. 6 14. 9 23. 1 0. 5 0. 086 0. 001 0. 17 0. 002 0. 132 25 Reduct ion p
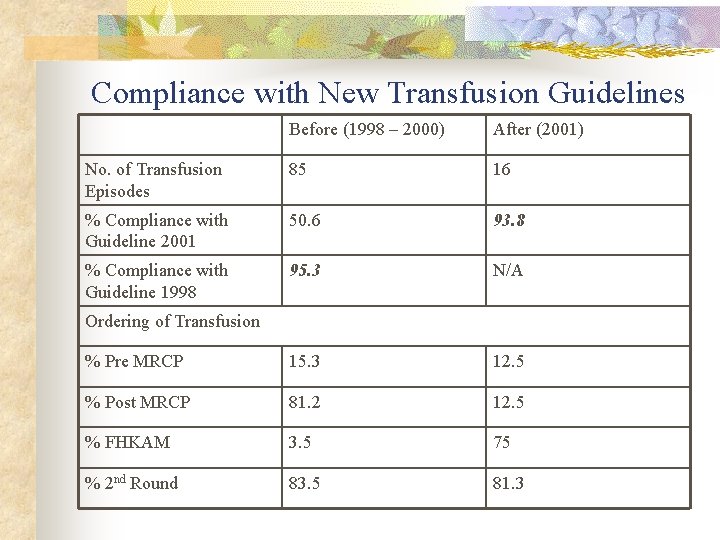
Compliance with New Transfusion Guidelines Before (1998 – 2000) After (2001) No. of Transfusion Episodes 85 16 % Compliance with Guideline 2001 50. 6 93. 8 % Compliance with Guideline 1998 95. 3 N/A % Pre MRCP 15. 3 12. 5 % Post MRCP 81. 2 12. 5 % FHKAM 3. 5 75 % 2 nd Round 83. 5 81. 3 Ordering of Transfusion

Non-compliance n Old guideline n n n Pre-MRCP Post-MRCP FHKAM 2 nd Round New guideline n n FHKAM 2 nd Round 4 episodes 1 1 2 3 1 episode 1 1
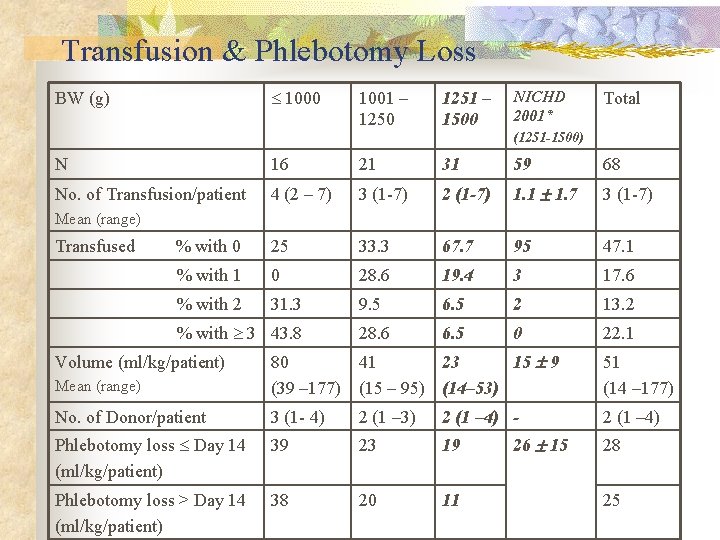
Transfusion & Phlebotomy Loss 1000 BW (g) 1001 – 1250 1251 – 1500 NICHD 2001* Total (1251 -1500) N 16 21 31 59 68 No. of Transfusion/patient 4 (2 – 7) 3 (1 -7) 2 (1 -7) 1. 1 1. 7 3 (1 -7) % with 0 25 33. 3 67. 7 95 47. 1 % with 1 0 28. 6 19. 4 3 17. 6 % with 2 31. 3 9. 5 6. 5 2 13. 2 28. 6 6. 5 0 22. 1 Mean (range) Transfused % with 3 43. 8 Mean (range) 80 41 23 15 9 (39 – 177) (15 – 95) (14– 53) 51 (14 – 177) No. of Donor/patient 3 (1 - 4) 2 (1 – 3) 2 (1 – 4) - 2 (1 – 4) Phlebotomy loss Day 14 (ml/kg/patient) 39 23 19 Phlebotomy loss > Day 14 (ml/kg/patient) 38 20 11 Volume (ml/kg/patient) 26 15 28 25
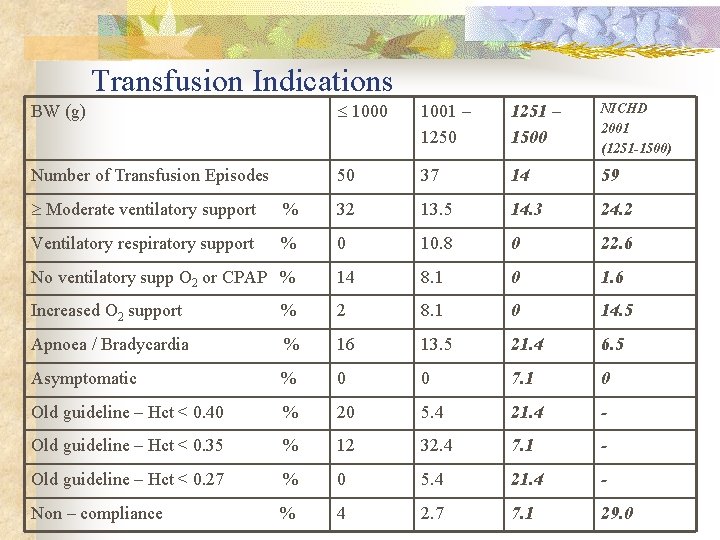
Transfusion Indications BW (g) 1000 1001 – 1250 1251 – 1500 NICHD 2001 (1251 -1500) Number of Transfusion Episodes 50 37 14 59 Moderate ventilatory support % 32 13. 5 14. 3 24. 2 Ventilatory respiratory support % 0 10. 8 0 22. 6 No ventilatory supp O 2 or CPAP % 14 8. 1 0 1. 6 Increased O 2 support % 2 8. 1 0 14. 5 Apnoea / Bradycardia % 16 13. 5 21. 4 6. 5 Asymptomatic % 0 0 7. 1 0 Old guideline – Hct < 0. 40 % 20 5. 4 21. 4 - Old guideline – Hct < 0. 35 % 12 32. 4 7. 1 - Old guideline – Hct < 0. 27 % 0 5. 4 21. 4 - Non – compliance % 4 2. 7 7. 1 29. 0
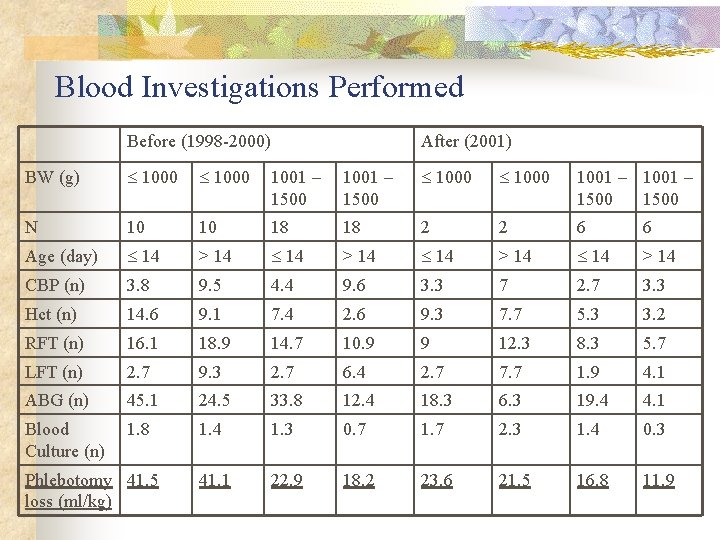
Blood Investigations Performed Before (1998 -2000) After (2001) BW (g) 1000 1001 – 1500 N 10 10 18 18 2 2 6 6 Age (day) 14 > 14 CBP (n) 3. 8 9. 5 4. 4 9. 6 3. 3 7 2. 7 3. 3 Hct (n) 14. 6 9. 1 7. 4 2. 6 9. 3 7. 7 5. 3 3. 2 RFT (n) 16. 1 18. 9 14. 7 10. 9 9 12. 3 8. 3 5. 7 LFT (n) 2. 7 9. 3 2. 7 6. 4 2. 7 7. 7 1. 9 4. 1 ABG (n) 45. 1 24. 5 33. 8 12. 4 18. 3 6. 3 19. 4 4. 1 Blood Culture (n) 1. 8 1. 4 1. 3 0. 7 1. 7 2. 3 1. 4 0. 3 41. 1 22. 9 18. 2 23. 6 21. 5 16. 8 11. 9 Phlebotomy 41. 5 loss (ml/kg)
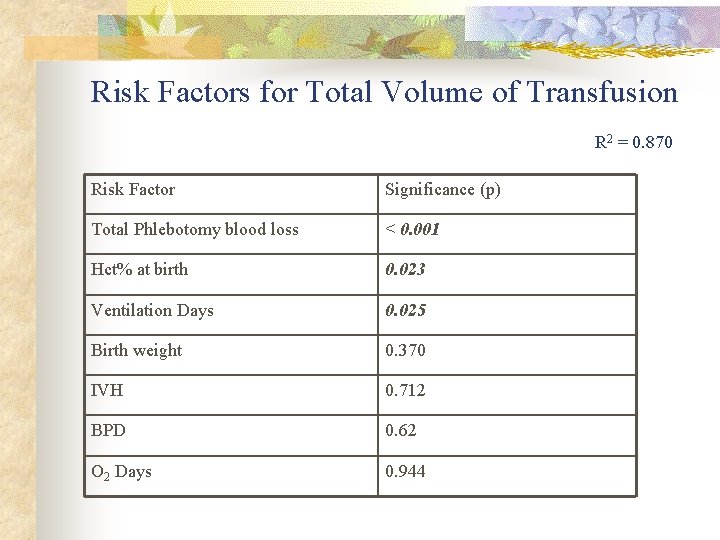
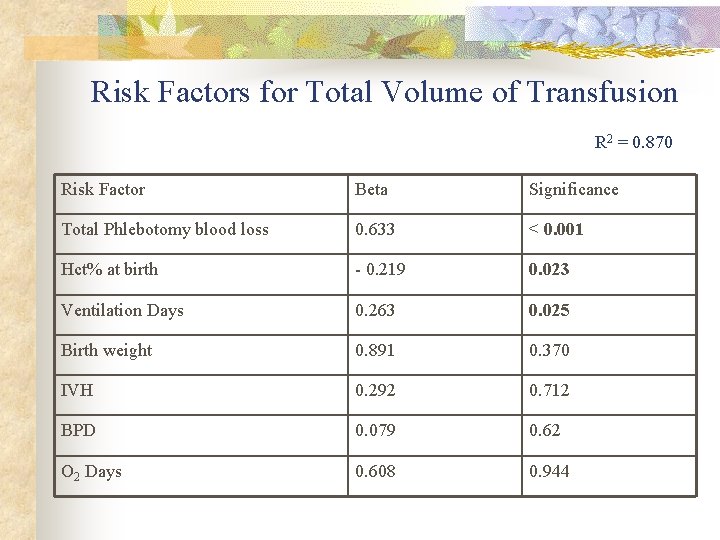
Risk Factors for Total Volume of Transfusion R 2 = 0. 870 Risk Factor Significance (p) Total Phlebotomy blood loss < 0. 001 Hct% at birth 0. 023 Ventilation Days 0. 025 Birth weight 0. 370 IVH 0. 712 BPD 0. 62 O 2 Days 0. 944

Discussion n Compliance n n Generally is good Before n n n Old Guideline New Guideline 95. 3% 50. 6% After n n Old Guideline New Guideline NA 93. 8%

Discussion n Reduction of RBC Transfusion n Before 62. 2% After 32% Reduction of transfusion may due to n n More conservative new guideline Decreased phlebotomy blood loss Infants were less ill RBC transfusion rate still higher if comparing with National Institute of Child Health and Human Development (NICHD) 2001

Discussion n Reduction of Phlebotomy blood loss (ml/kg) n Before • 1000 • 1001 -1500 n Day 15 - Discharge 41. 1 18. 2 23. 6 16. 8 25. 1 11. 9 After • 1000 • 1001 -1500 n Day 14 41. 5 22. 9 Comparable to NICHD 2001

Discussion n n Implementation of new guidelines can largely reduce the number of blood transfusions in premature infants Reduction of phlebotomy loss contribute significantly to reduction in transfusion requirements Morbidities (BPD, ROP), length of stay & age to attain weight 2. 2 kg (growth) were not significantly different with the implementation of new guideline Although our phlebotomy blood loss is comparable to NICHD, our transfusion rate is still higher The total volume of transfusion was largely accounted by phlebotomy blood loss & ventilation days

Discussion n n Speculation – with modification of guideline, limiting blood loss & use of micro-methods (POCT), non-invasive laboratory monitoring, further more conservative transfusion approach may be adopted ? Use of Erythropoietin for preterm infants in PYNEH
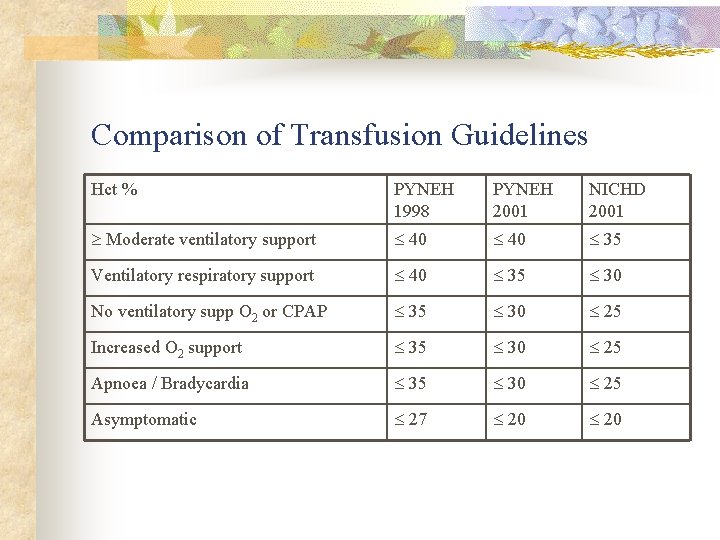
Comparison of Transfusion Guidelines Hct % PYNEH 1998 PYNEH 2001 NICHD 2001 Moderate ventilatory support 40 35 Ventilatory respiratory support 40 35 30 No ventilatory supp O 2 or CPAP 35 30 25 Increased O 2 support 35 30 25 Apnoea / Bradycardia 35 30 25 Asymptomatic 27 20

Thank you

Old guidelines 1998 n Hct < 40% or Hb < 13 g/dl n n if oxygen or ventilator dependent if asymtomatic Hct 40%, severe respiratory illness n n if symptomatic Hct < 27% or Hb < 8 g/dl n n n Hct < 35% or Hb < 10 g/dl n n New guidelines 2001 n n Hct 35% n Blood loss of > 10% of blood volume n n n n Ventilator/CPAP, MAP 6 -8 cm. H 2 O Fi. O 2 35 -50% Hct 30% n n Ventilator/CPAP, MAP >8 cm. H 2 O Fi. O 2 > 50% Severe congenital heart disease with cyanosis/heart failure Respiratory disease requiring Fi. O 2 2535% / nasal cannula O 2 1/8 -1/4 L/min CPAP/ IPPV, MAP <6 cm. H 2 O Sustained tachycardia (>180/min) or tachypnoea (>80/min) for 24 hours Apnoea/bradycardia 10/24 hours or 2 requiring bag mask ventilation Cessation of wt gain x 4 days Undergoing major surgery Hct 20% asymptomic Acute blood loss with shock
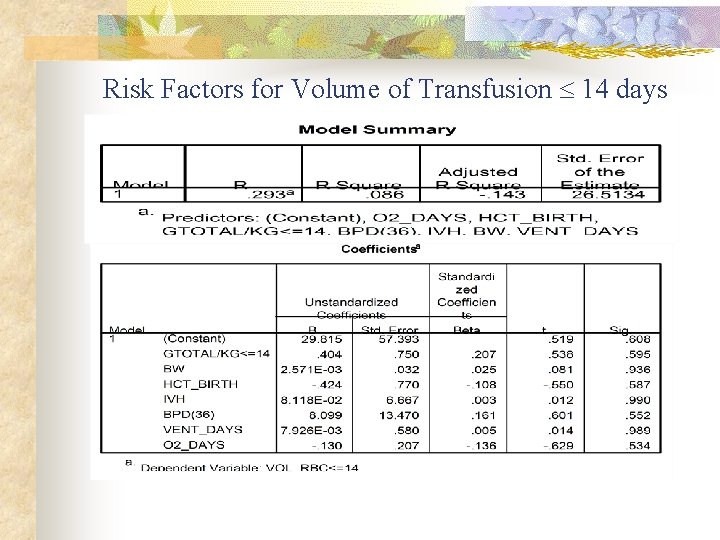
Risk Factors for Volume of Transfusion 14 days
Risk Factors for Total Volume of Transfusion R 2 = 0. 870 Risk Factor Beta Significance Total Phlebotomy blood loss 0. 633 < 0. 001 Hct% at birth - 0. 219 0. 023 Ventilation Days 0. 263 0. 025 Birth weight 0. 891 0. 370 IVH 0. 292 0. 712 BPD 0. 079 0. 62 O 2 Days 0. 608 0. 944

Discussion n n n Implementation of new guidelines can largely reduce the number of blood transfusions in premature infants Reduction of phlebotomy loss contribute significantly to reduction in transfusion requirements Morbidities (CLD, ROP), length of stay & age to attain weight 2200 g (growth) were not significantly different with the implementation of new guideline Although our phlebotomy blood loss is comparable to NICHD, our transfusion rate is still higher The total volume of transfusion was largely accounted by phlebotomy blood loss & ventilation days The inverse relationship between Hct% at birth and total volume of transfusion need further analysis
- Slides: 30