ATTRACTIONS BETWEEN MOLECULES Intermolecular Forces The forces of
ATTRACTIONS BETWEEN MOLECULES Intermolecular Forces
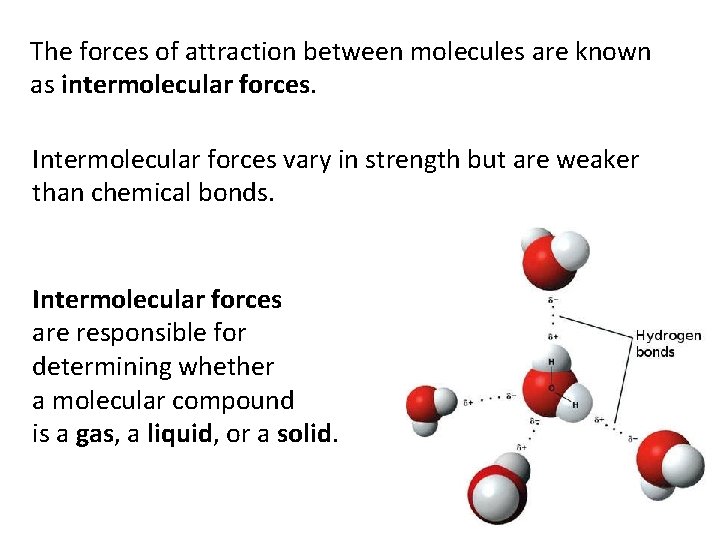
The forces of attraction between molecules are known as intermolecular forces. Intermolecular forces vary in strength but are weaker than chemical bonds. Intermolecular forces are responsible for determining whether a molecular compound is a gas, a liquid, or a solid.

Types of Intermolecular Forces: • Van der Waals Dispersion Forces (or London Dispersion Forces) • Van der Waals Dipole-Dipole Interactions • Hydrogen Bonding

Van der Waals Dispersion Forces (or London Dispersion Forces) WEAKEST weakest Nonpolar molecules have no charges, have no dipole. Nonpolar molecules contain electrons that are in constant motion. Most of the time, the electrons are symmetrically distributed within the atom.
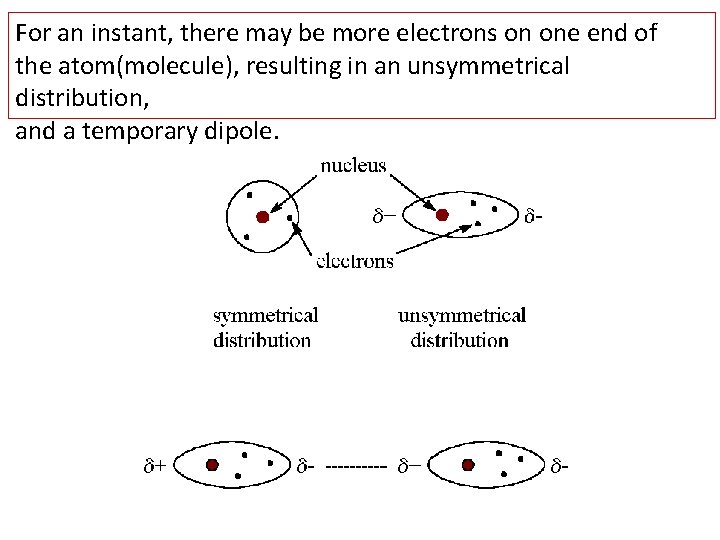
For an instant, there may be more electrons on one end of the atom(molecule), resulting in an unsymmetrical distribution, and a temporary dipole.
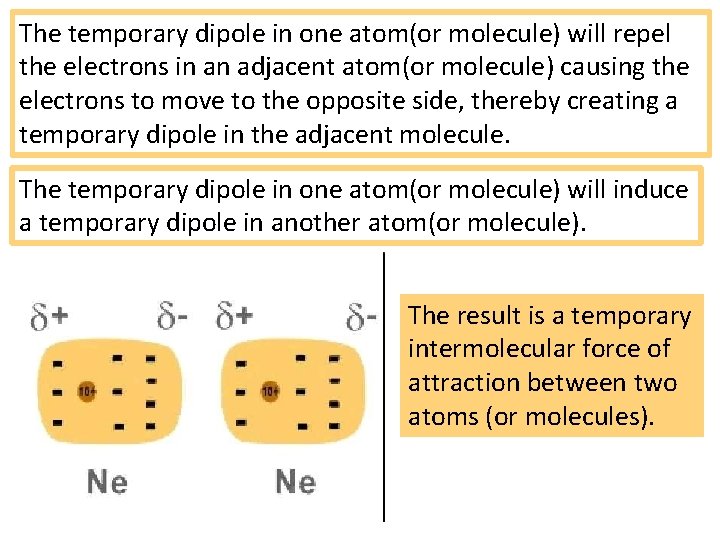
The temporary dipole in one atom(or molecule) will repel the electrons in an adjacent atom(or molecule) causing the electrons to move to the opposite side, thereby creating a temporary dipole in the adjacent molecule. The temporary dipole in one atom(or molecule) will induce a temporary dipole in another atom(or molecule). The result is a temporary intermolecular force of attraction between two atoms (or molecules).

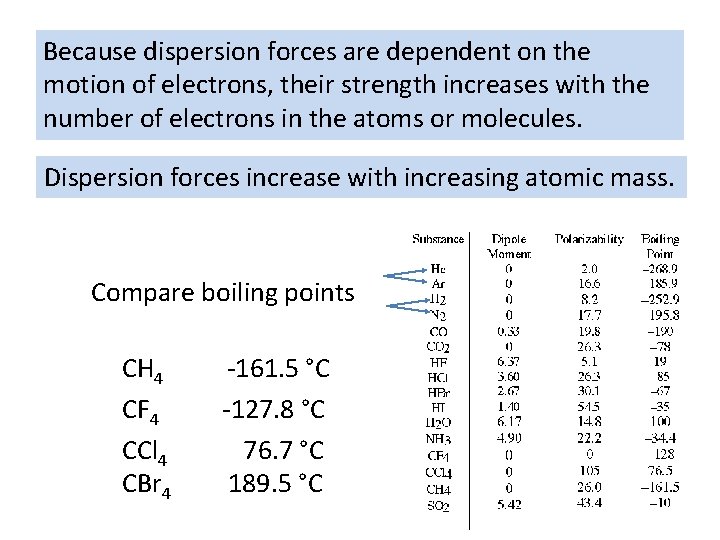
Because dispersion forces are dependent on the motion of electrons, their strength increases with the number of electrons in the atoms or molecules. Dispersion forces increase with increasing atomic mass. Compare boiling points CH 4 -161. 5 °C CF 4 -127. 8 °C CCl 4 76. 7 °C CBr 4 189. 5 °C

Explain the following trend: Boiling point F 2 gas -188 °C Cl 2 gas -35 °C Br 2 liquid 59 °C I 2 solid 184 °C Intermolecular forces are responsible for determining whether a molecular compound is a gas, a liquid, or a solid. Dispersion forces increase with increasing atomic mass.
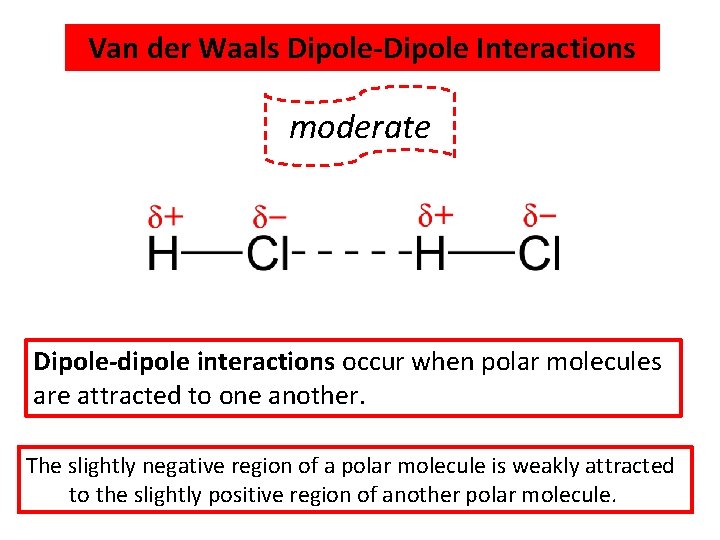
Van der Waals Dipole-Dipole Interactions WEAKEST moderate Dipole-dipole interactions occur when polar molecules are attracted to one another. The slightly negative region of a polar molecule is weakly attracted to the slightly positive region of another polar molecule.

A dipole moment is a measure of the strength of the dipole. The larger the dipole moment, the stronger the intermolecular forces, and consequently, the higher the melting point and boiling point.

The increase in boiling point happens because the molecules are getting larger with more electrons, and so van der Waals dispersion forces become greater.
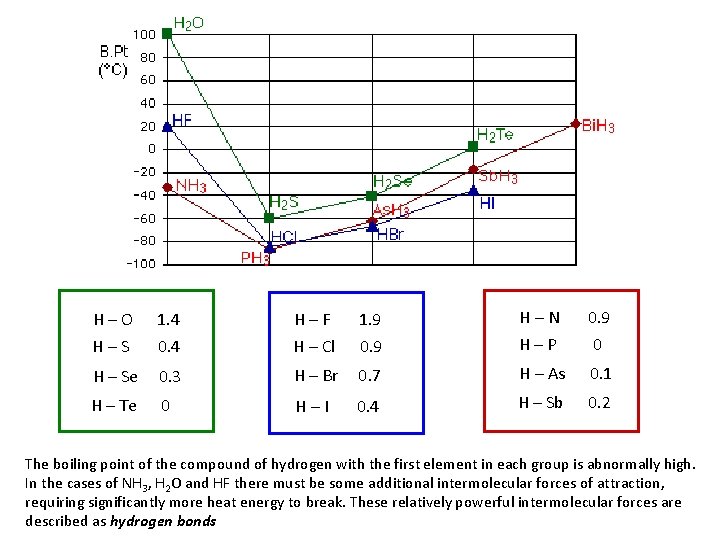
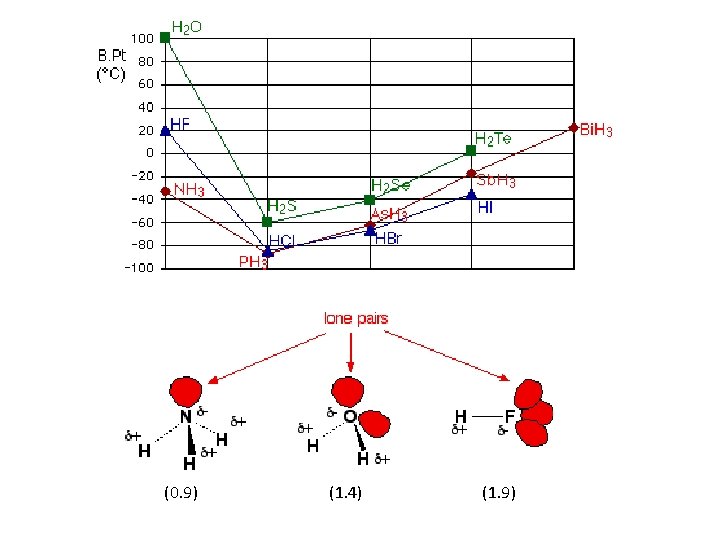
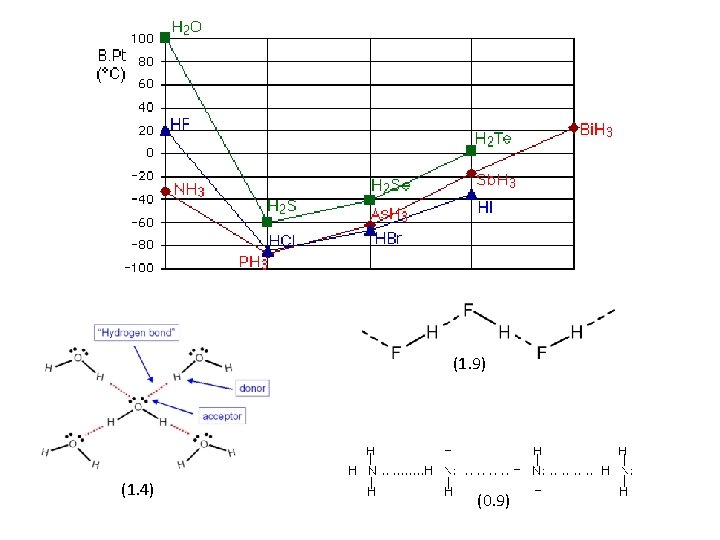
H – O 1. 4 H – F 1. 9 H – N 0. 9 H – S 0. 4 H – Cl 0. 9 H – P 0 H – Se 0. 3 H – Br 0. 7 H – As 0. 1 H – Te 0 H – I 0. 4 H – Sb 0. 2 The boiling point of the compound of hydrogen with the first element in each group is abnormally high. In the cases of NH 3, H 2 O and HF there must be some additional intermolecular forces of attraction, requiring significantly more heat energy to break. These relatively powerful intermolecular forces are described as hydrogen bonds
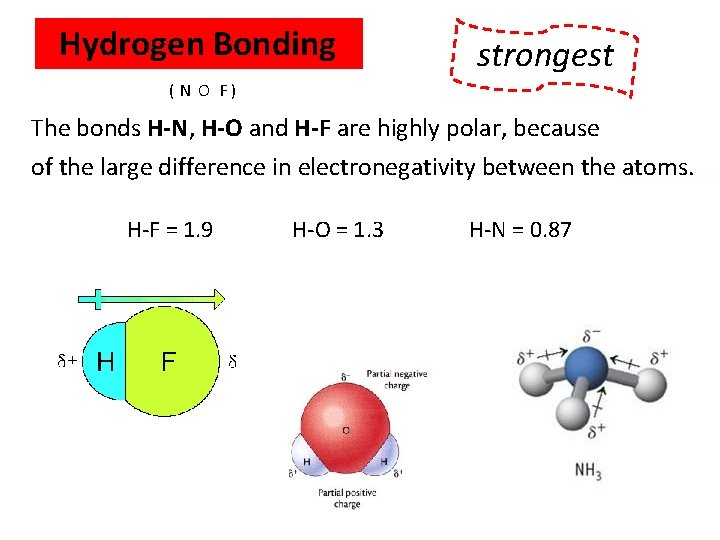
Hydrogen Bonding WEAKEST strongest ( N O F ) The bonds H-N, H-O and H-F are highly polar, because of the large difference in electronegativity between the atoms. H-F = 1. 9 H-O = 1. 3 H-N = 0. 87
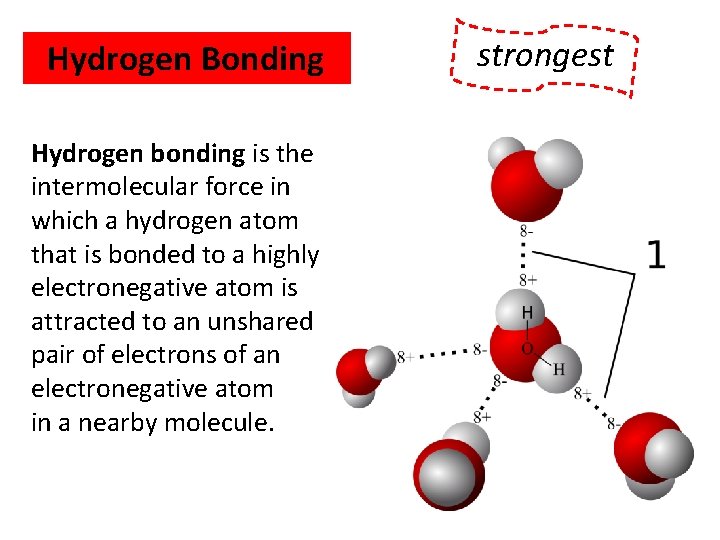
Hydrogen Bonding Hydrogen bonding is the intermolecular force in which a hydrogen atom that is bonded to a highly electronegative atom is attracted to an unshared pair of electrons of an electronegative atom in a nearby molecule. WEAKEST strongest
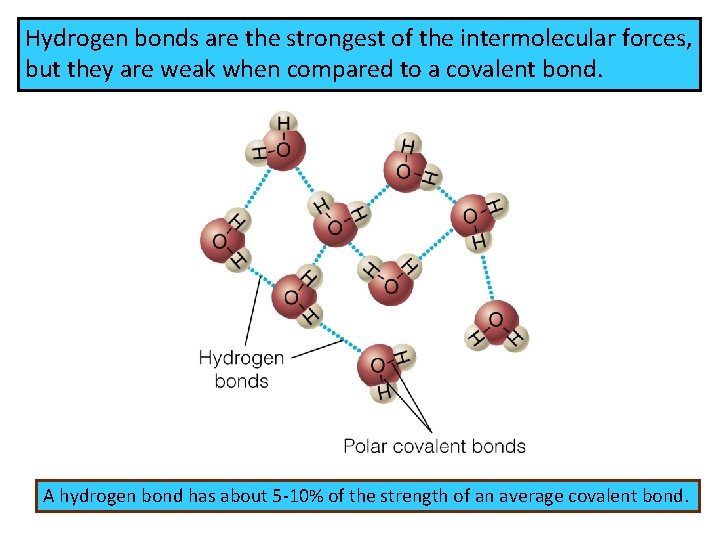
Hydrogen bonds are the strongest of the intermolecular forces, but they are weak when compared to a covalent bond. A hydrogen bond has about 5 -10% of the strength of an average covalent bond.
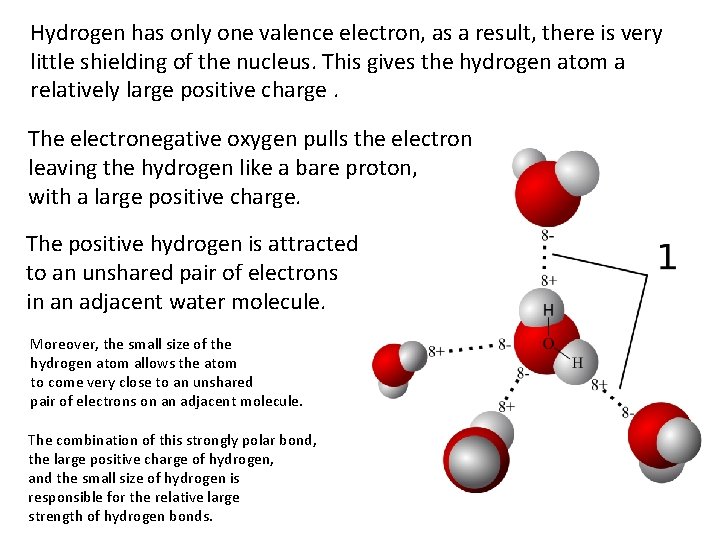
Hydrogen has only one valence electron, as a result, there is very little shielding of the nucleus. This gives the hydrogen atom a relatively large positive charge. The electronegative oxygen pulls the electron leaving the hydrogen like a bare proton, with a large positive charge. The positive hydrogen is attracted to an unshared pair of electrons in an adjacent water molecule. Moreover, the small size of the hydrogen atom allows the atom to come very close to an unshared pair of electrons on an adjacent molecule. The combination of this strongly polar bond, the large positive charge of hydrogen, and the small size of hydrogen is responsible for the relative large strength of hydrogen bonds.

Hydrogen bonding occurs when H is bonded to N, O, or F: (0. 9) (1. 4) (1. 9) Notice that in each of these molecules: • The hydrogen is attached directly to one of the most electronegative elements, causing the hydrogen to acquire a significant amount of positive charge. • Each of the elements to which the hydrogen is attached is not only significantly negative, but also has at least one "active" lone pair. Lone pairs at the 2 -level have the electrons contained in a relatively small volume of space which therefore has a high density of negative charge. Lone pairs at higher levels are more diffuse and not so attractive to positive things.
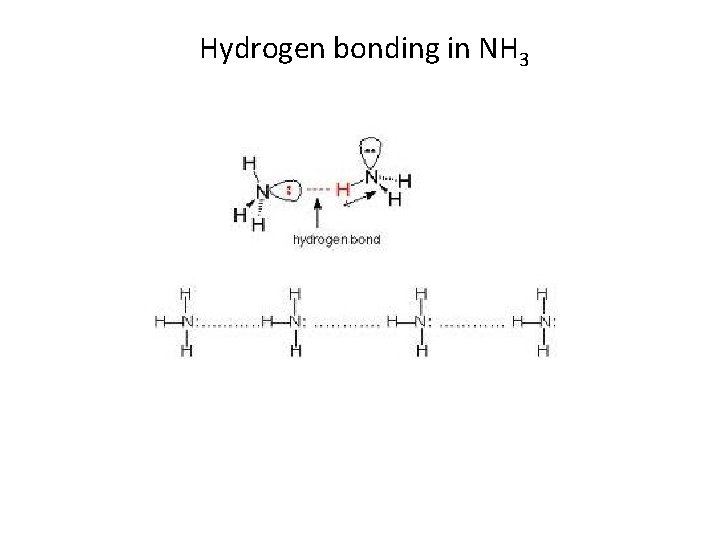
Hydrogen bonding in NH 3
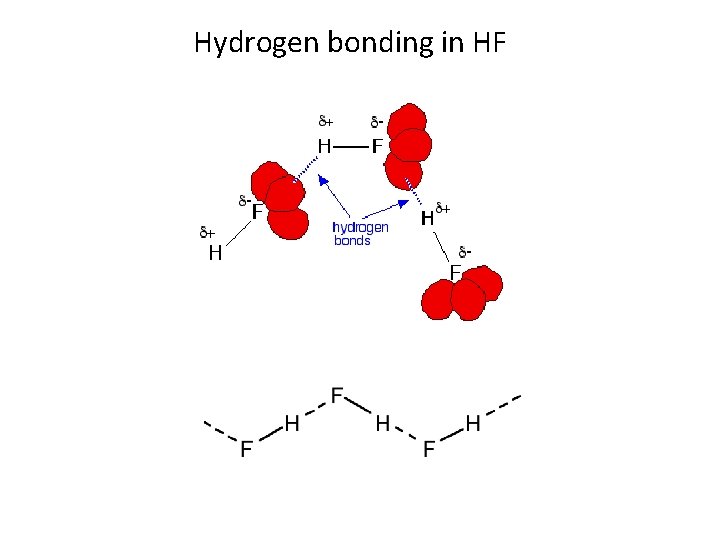
Hydrogen bonding in HF
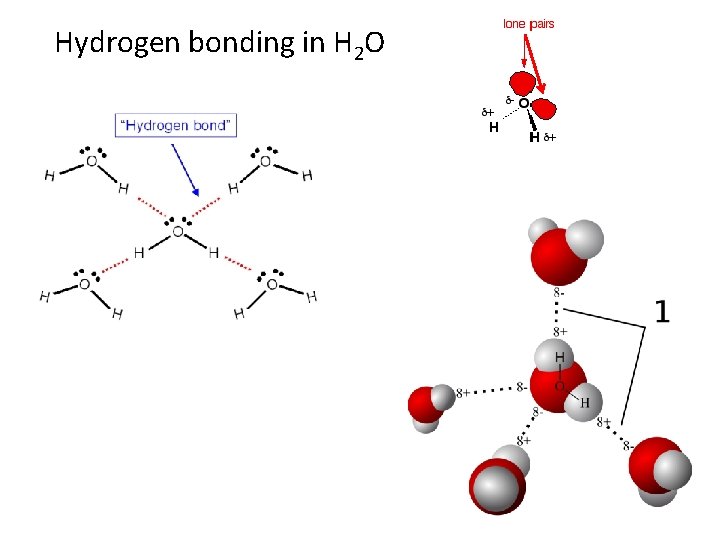
Hydrogen bonding in H 2 O

Consider two water molecules coming close together The δ+ hydrogen is so strongly attracted to the lone pair that it is almost as if you were beginning to form a co-ordinate (dative covalent) bond. It doesn't go that far, but the attraction is significantly stronger than an ordinary dipole-dipole interaction. Hydrogen bonds have about a tenth of the strength of an average covalent bond, and are being constantly broken and reformed in liquid water. If you liken the covalent bond between the oxygen and hydrogen to a stable marriage, the hydrogen bond has "just good friends" status. Water as a "perfect" example of hydrogen bonding Notice that each water molecule can potentially form four hydrogen bonds with surrounding water molecules. There are exactly the right numbers of δ+ hydrogens and lone pairs so that every one of them can be involved in hydrogen bonding. This is why the boiling point of water is higher than that of ammonia or hydrogen fluoride.
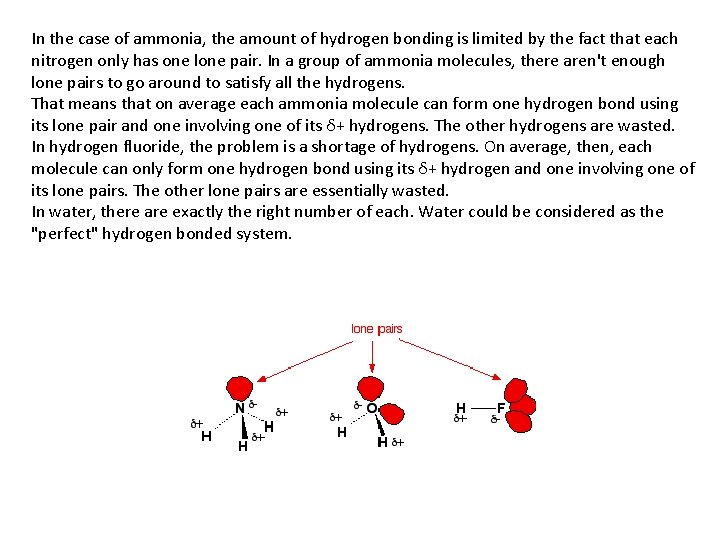
In the case of ammonia, the amount of hydrogen bonding is limited by the fact that each nitrogen only has one lone pair. In a group of ammonia molecules, there aren't enough lone pairs to go around to satisfy all the hydrogens. That means that on average each ammonia molecule can form one hydrogen bond using its lone pair and one involving one of its δ+ hydrogens. The other hydrogens are wasted. In hydrogen fluoride, the problem is a shortage of hydrogens. On average, then, each molecule can only form one hydrogen bond using its δ+ hydrogen and one involving one of its lone pairs. The other lone pairs are essentially wasted. In water, there are exactly the right number of each. Water could be considered as the "perfect" hydrogen bonded system.

Intermolecular Forces and Boiling Point At room temperature, the intermolecular forces are attracting water molecules to each other, thereby allowing water to be a liquid.
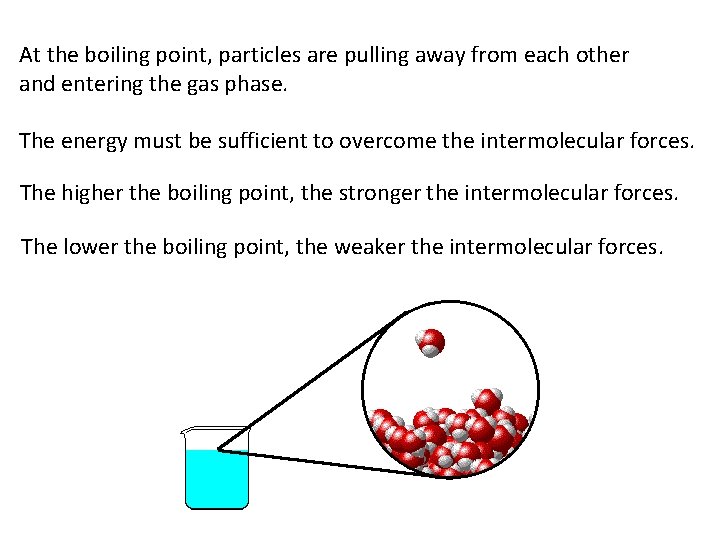
At the boiling point, particles are pulling away from each other and entering the gas phase. The energy must be sufficient to overcome the intermolecular forces. The higher the boiling point, the stronger the intermolecular forces. The lower the boiling point, the weaker the intermolecular forces.
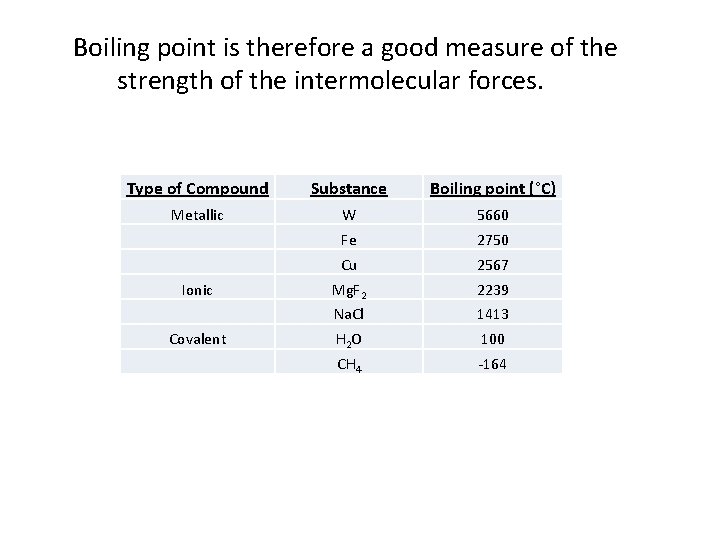
Boiling point is therefore a good measure of the strength of the intermolecular forces. Type of Compound Substance Boiling point (°C) Metallic W 5660 Fe 2750 Cu 2567 Ionic Mg. F 2 2239 Na. Cl 1413 Covalent H 2 O 100 CH 4 -164
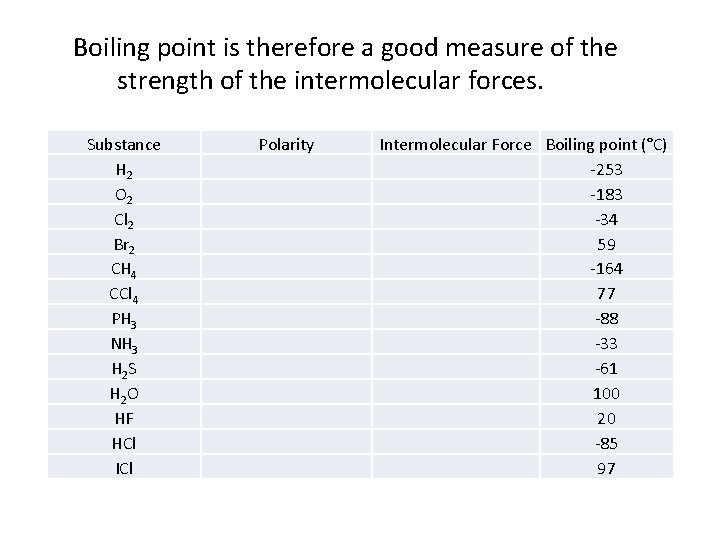
Boiling point is therefore a good measure of the strength of the intermolecular forces. Substance H 2 O 2 Cl 2 Br 2 CH 4 CCl 4 PH 3 NH 3 H 2 S H 2 O HF HCl ICl Polarity Intermolecular Force Boiling point (°C) -253 -183 -34 59 -164 77 -88 -33 -61 100 20 -85 97
- Slides: 29