Atrial fibrillation stroke prevention Whats new Prof John

Atrial fibrillation & stroke prevention: What’s new? Prof. John Camm St Georges University of London, United Kingdom
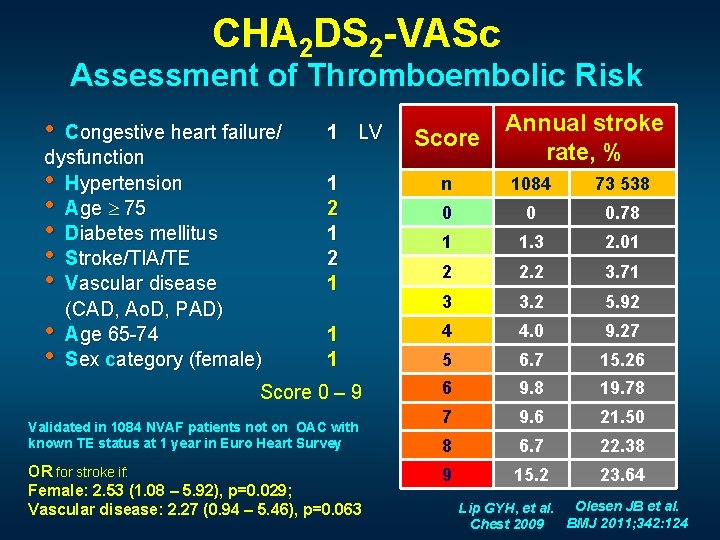
CHA 2 DS 2 -VASc Assessment of Thromboembolic Risk • Congestive heart failure/ dysfunction • Hypertension • Age 75 • Diabetes mellitus • Stroke/TIA/TE • Vascular disease (CAD, Ao. D, PAD) • Age 65 -74 • Sex category (female) 1 LV Score Annual stroke rate, % 1 2 1 n 1084 73 538 0 0 0. 78 1 1. 3 2. 01 2 2. 2 3. 71 3 3. 2 5. 92 1 1 4 4. 0 9. 27 5 6. 7 15. 26 6 9. 8 19. 78 7 9. 6 21. 50 8 6. 7 22. 38 9 15. 2 23. 64 Score 0 – 9 Validated in 1084 NVAF patients not on OAC with known TE status at 1 year in Euro Heart Survey OR for stroke if: Female: 2. 53 (1. 08 – 5. 92), p=0. 029; Vascular disease: 2. 27 (0. 94 – 5. 46), p=0. 063 Lip GYH, et al. Olesen JB et al. BMJ 2011; 342: 124 Chest 2009
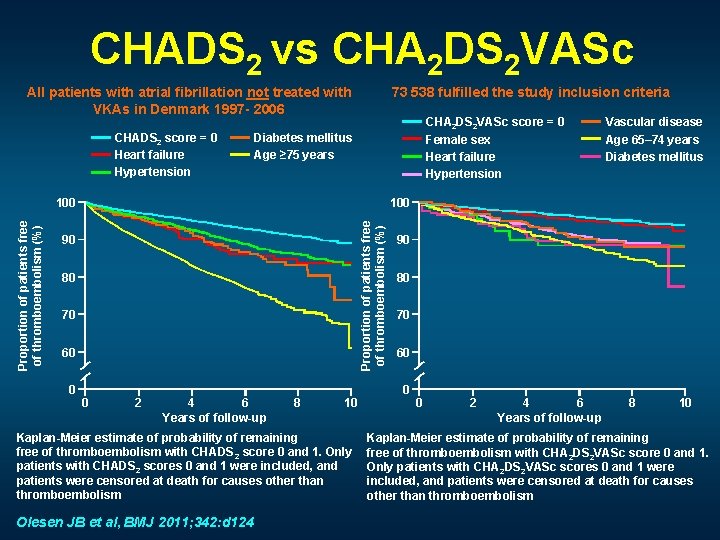
CHADS 2 vs CHA 2 DS 2 VASc All patients with atrial fibrillation not treated with VKAs in Denmark 1997 - 2006 CHADS 2 score = 0 Heart failure Hypertension 73 538 fulfilled the study inclusion criteria CHA 2 DS 2 VASc score = 0 Female sex Heart failure Hypertension Diabetes mellitus Age ≥ 75 years 100 Proportion of patients free of thromboembolism (%) 100 90 80 70 60 0 Vascular disease Age 65– 74 years Diabetes mellitus 0 2 4 6 Years of follow-up 8 10 Kaplan-Meier estimate of probability of remaining free of thromboembolism with CHADS 2 score 0 and 1. Only patients with CHADS 2 scores 0 and 1 were included, and patients were censored at death for causes other than thromboembolism Olesen JB et al, BMJ 2011; 342: d 124 90 80 70 60 0 0 2 4 6 Years of follow-up 8 10 Kaplan-Meier estimate of probability of remaining free of thromboembolism with CHA 2 DS 2 VASc score 0 and 1. Only patients with CHA 2 DS 2 VASc scores 0 and 1 were included, and patients were censored at death for causes other than thromboembolism
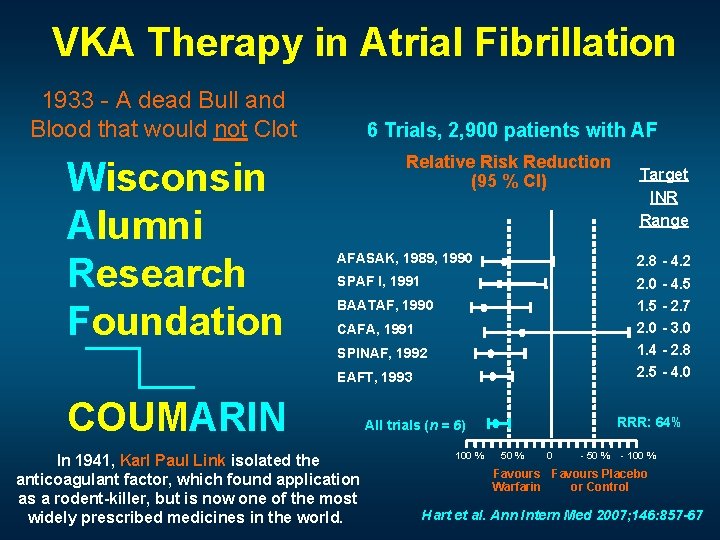
VKA Therapy in Atrial Fibrillation 1933 - A dead Bull and Blood that would not Clot Wisconsin Alumni Research Foundation COUMARIN 6 Trials, 2, 900 patients with AF Relative Risk Reduction (95 % CI) AFASAK, 1989, 1990 2. 8 - 4. 2 2. 0 - 4. 5 1. 5 - 2. 7 2. 0 - 3. 0 1. 4 - 2. 8 2. 5 - 4. 0 SPAF I, 1991 BAATAF, 1990 CAFA, 1991 SPINAF, 1992 EAFT, 1993 In 1941, Karl Paul Link isolated the anticoagulant factor, which found application as a rodent-killer, but is now one of the most widely prescribed medicines in the world. RRR: 64% All trials (n = 6) 100 % Target INR Range 50 % 0 - 50 % - 100 % Favours Placebo or Control Warfarin Hart et al. Ann Intern Med 2007; 146: 857 -67
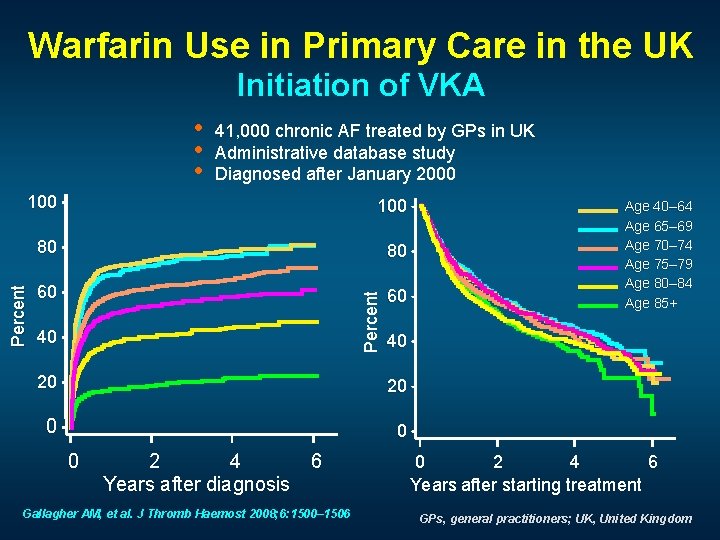
Warfarin Use in Primary Care in the UK Initiation of VKA • • • 41, 000 chronic AF treated by GPs in UK Administrative database study Diagnosed after January 2000 80 80 60 60 Percent 100 40 40 20 20 0 2 4 Years after diagnosis 6 Gallagher AM, et al. J Thromb Haemost 2008; 6: 1500– 1506 Age 40– 64 Age 65– 69 Age 70– 74 Age 75– 79 Age 80– 84 Age 85+ 0 2 4 6 Years after starting treatment GPs, general practitioners; UK, United Kingdom

Sub-optimal TTR and Risk of Stroke Warfarin TTR groupa 1. 0 Samsa, 2000 (n = 61) Cumulative survival Samsa, 2000 (n = 125) Mc. Cormick, 2001 (n = 174) Matchar, 2003 (n = 363) Matchar, 2003 (n = 317) Go, 2003 (n = 7445) Shen, 2007 (n = 11, 016) Nichol, 2008 (n = 756) 71%– 100% 61%– 70% 51%– 60% 41%– 50% 31%– 40% ≤ 30% No warfarin 0. 9 0. 8 0. 7 Overall TTR 51% 0. 6 20 40 60 % TTR (95% CI) 80 0 500 1000 1500 2000 Survival to stroke (days) ● Meta-analysis of TTR (%) of AF patients treated with warfarin in the community ● TTR >70% is necessary to reduce stroke risk in patients with CHADS 2 score ≥ 2 compared with the nonwarfarin treatment group (p=0. 025) a. No. of warfarin-treated patients in each group is defined by proportion of time spent within INR target range TTR, time in therapeutic range Baker WL et al. J Manag Care Pharm 2009; 15: 244– 252. Morgan CL et al. Thromb Res 2009; 124: 37– 41.

By any other name ……! ADIOS – Antiarrhythmic Drugs Improves Outcomes Study KAPUT ADIOS
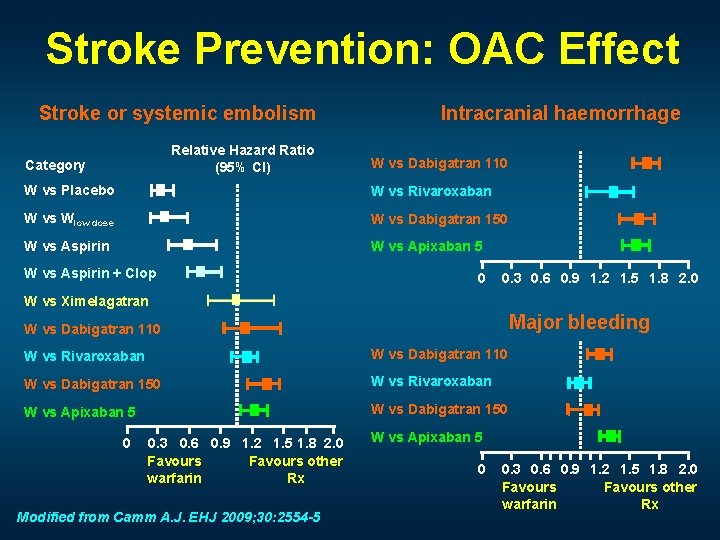
Stroke Prevention: OAC Effect Stroke or systemic embolism Relative Hazard Ratio (95% CI) Category Intracranial haemorrhage W vs Dabigatran 110 W vs Placebo W vs Rivaroxaban W vs Wlow dose W vs Dabigatran 150 W vs Aspirin W vs Apixaban 5 W vs Aspirin + Clop 0 0. 3 0. 6 0. 9 1. 2 1. 5 1. 8 2. 0 W vs Ximelagatran Major bleeding W vs Dabigatran 110 W vs Rivaroxaban W vs Dabigatran 110 W vs Dabigatran 150 W vs Rivaroxaban W vs Apixaban 5 W vs Dabigatran 150 0 0. 3 0. 6 0. 9 1. 2 1. 5 1. 8 2. 0 Favours other warfarin Rx Modified from Camm A. J. EHJ 2009; 30: 2554 -5 W vs Apixaban 5 0 0. 3 0. 6 0. 9 1. 2 1. 5 1. 8 2. 0 Favours other warfarin Rx
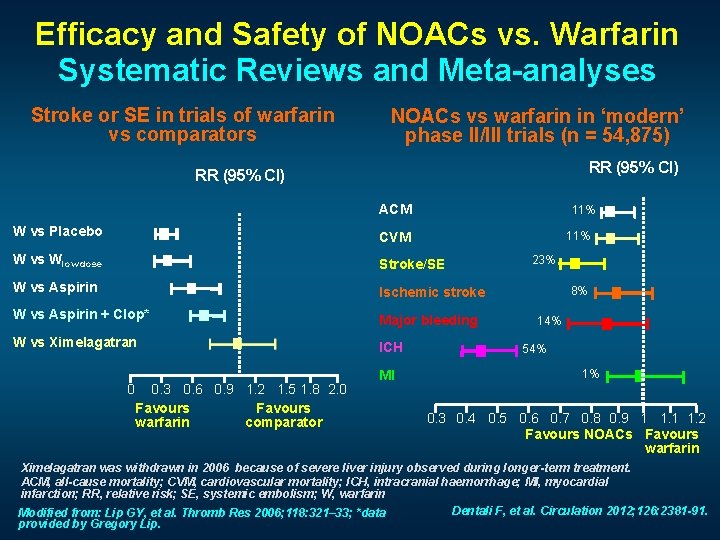
Efficacy and Safety of NOACs vs. Warfarin Systematic Reviews and Meta-analyses Stroke or SE in trials of warfarin vs comparators NOACs vs warfarin in ‘modern’ phase II/III trials (n = 54, 875) RR (95% CI) ACM 11% W vs Placebo CVM W vs Wlow dose Stroke/SE W vs Aspirin Ischemic stroke W vs Aspirin + Clop* Major bleeding W vs Ximelagatran ICH 0 0. 3 0. 6 0. 9 1. 2 1. 5 1. 8 2. 0 Favours warfarin comparator MI 11% 23% 8% 14% 54% 1% 0. 3 0. 4 0. 5 0. 6 0. 7 0. 8 0. 9 1 1. 2 Favours NOACs Favours warfarin Ximelagatran was withdrawn in 2006 because of severe liver injury observed during longer-term treatment. ACM, all-cause mortality; CVM, cardiovascular mortality; ICH, intracranial haemorrhage; MI, myocardial infarction; RR, relative risk; SE, systemic embolism; W, warfarin Dentali F, et al. Circulation 2012; 126: 2381 -91. Modified from: Lip GY, et al. Thromb Res 2006; 118: 321– 33; *data provided by Gregory Lip.
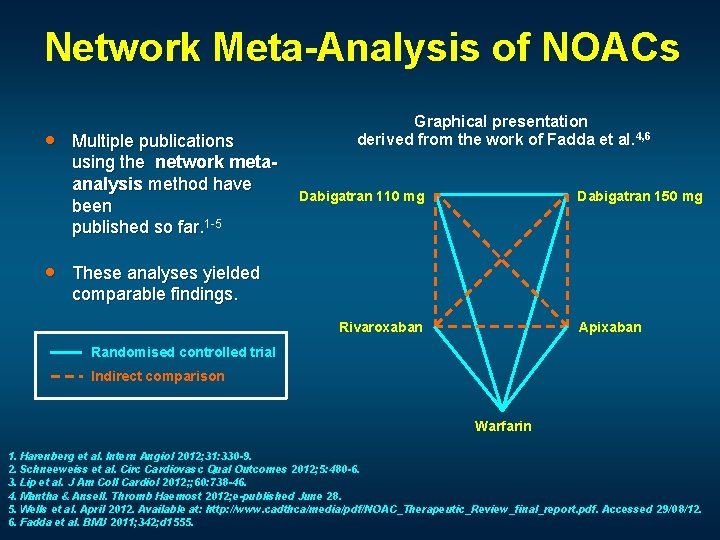
Network Meta-Analysis of NOACs · · Multiple publications using the network metaanalysis method have been published so far. 1 -5 Graphical presentation derived from the work of Fadda et al. 4, 6 Dabigatran 110 mg Dabigatran 150 mg These analyses yielded comparable findings. Apixaban Rivaroxaban Randomised controlled trial Indirect comparison Warfarin 1. Harenberg et al. Intern Angiol 2012; 31: 330 -9. 2. Schneeweiss et al. Circ Cardiovasc Qual Outcomes 2012; 5: 480 -6. 3. Lip et al. J Am Coll Cardiol 2012; ; 60: 738 -46. 4. Mantha & Ansell. Thromb Haemost 2012; e-published June 28. 5. Wells et al. April 2012. Available at: http: //www. cadthca/media/pdf/NOAC_Therapeutic_Review_final_report. pdf. Accessed 29/08/12. 6. Fadda et al. BMJ 2011; 342; d 1555.
![NOAC Rx in AF: Indirect Comparison 10 efficacy (stroke or Systemic embolus [ITT]) Dabigatran NOAC Rx in AF: Indirect Comparison 10 efficacy (stroke or Systemic embolus [ITT]) Dabigatran](http://slidetodoc.com/presentation_image_h/045ba7b021819feaef49cf5c52c9cf8d/image-11.jpg)
NOAC Rx in AF: Indirect Comparison 10 efficacy (stroke or Systemic embolus [ITT]) Dabigatran 150 HRD v W = 0. 65 HRD v A = 0. 82 (0. 62 to 1. 10) 10 efficacy in CHADS 2 ≥ 3 Dabigatran 150 HRD v W = 0. 70 HRD v A = 1. 03 (0. 69 to 1. 54) Apixaban HRA v W = 0. 79 HRA v W = 0. 68 Safety (major haemorrhage as treated) Dabigatran 150 HRD v W = 0. 98 HRD v A = 1. 42 (1. 16 to 1. 74) HRD v R = 0. 80 (0. 56 to 1. 13) Rivaroxaban HRR v W = 0. 88 HRA v R = 0. 77 (0. 56 to 1. 06) Safety (major haemorrhage in CHADS 2 ≥ 3) Dabigatran 150 HRD v W = 1. 05 HRD v A = 1. 52 (1. 11 to 2. 07) Apixaban HRA v W = 0. 69 Schneeweeisss S, et al. Circ Cardiovasc Qual Outcomes. 2012; 5: 480 -486. HRD v R = 1. 04 (0. 80 to 1. 34) Rivaroxaban HRR v W = 1. 01 HRA v R = 0. 77 (0. 52 to 0. 90)
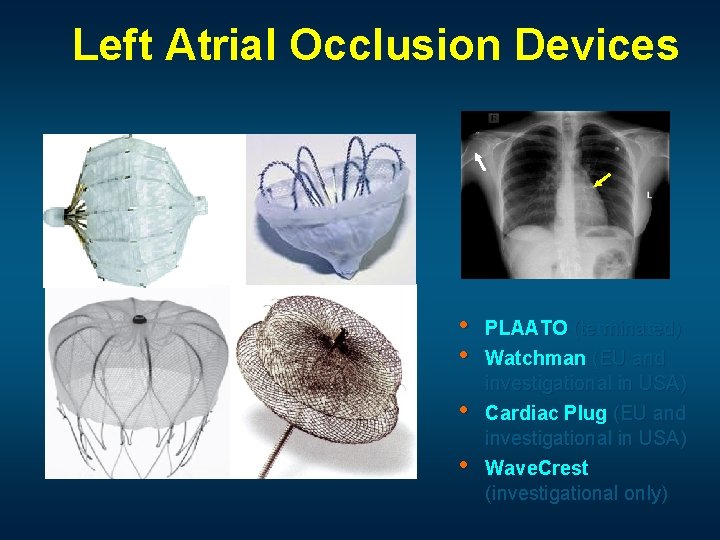
Left Atrial Occlusion Devices • • PLAATO (terminated) Watchman (EU and investigational in USA) Cardiac Plug (EU and investigational in USA) Wave. Crest (investigational only)

PROTECT-AF: Latest Results 463 patients received the Watchman and 244 warfarin management Average CHADS 2 scores 2. 2 and 2. 3, respectively Mean follow-up: 45 months Rate Ratios (95% CI) for Primary Efficacy and Safety End Points and Secondary End Points in PROTECT-AF, by Intention to Treat End points (2621 pt/years) Primary efficacy end point: Stroke, systemic embolism, or cardiovascular or unexplained death All-cause mortality Cardiovascular mortality Hemorrhagic stroke Primary safety end point: serious pericardial effusion, major bleeding, procedure-related stroke, hemorrhagic stroke, and device embolization Reddy VY, et al. Abstract HRS Denver 2013 RR (95% CI) 0. 60 (0. 41– 1. 05) 0. 66 (0. 45– 0. 98) 0. 40 (0. 21– 0. 72) 0. 15 (0. 03– 0. 49) 1. 17 (0. 78– 1. 95)

LARIAT - Percutaneous LAA Ligation Patients screened N=119 Patients excluded N=16 (13. 4%) Eligible patients 103 (86. 5%) LAA > 40 mm (8) Unsuitable LAA orientation (8) Excluded at procedure N = 14 (13. 6%) Adhesions (3) Mobile thrombus (11) Patients to be treated N=89 (86. 4%) Residual leak (3) Severe pericarditis(1) Late effusion (1) Unexplained SCD (1) Non-embolic late CVA (2) Bartus K et al. JACC 2012 Failure N=4 (4. 5%) Success N=85 (95. 5%) One year follow-up

LAA Closure/Occlusion/Excision Recommendations for LAA closure/occlusion/excision Recommendations Class Level Interventional, percutaneous LAA closure may be considered in patients with a high stroke risk and contraindications for long-term oral anticoagulation. IIb B Surgical excision of the LAA may be considered in patients undergoing open heart surgery. IIb C European Heart Journal 2012 doi: 10. 1093/eurheartj/ehs 253 European Heart Journal 2012 - doi: 10. 1093/eurheartj/ehs 253

ESC 2012 Update AF Guidelines AF Non-valvular No antithrombotic therapy Dotted lines indicate options not well validated; Dashed lines: less preferable or less validated * = mechanical or rheumatic † = not “female” only §= dual antiplatelet therapy preferred ‡ = see Summary of Product Characteristics for specific indications CHA 2 DS 2 -VASc: 1 and not suitable for, or refusing NOAC or VKA EUAPI 355 c. UK Paroxysmal, Persistent or Permanent Valvular* < 65 years, no cardiovascular disease Dose-adjusted VKA INR: 2 -3) CHA 2 DS 2 -VASc 1† ≥ 2 OAC therapy Modified from the 2012 focused update of the ESC Guidelines for the management of AF Camm AJ, et al. Europace. 2012; 14(10): 1385 -413. Assess bleeding risk (HAS-BLED) Consider patient values and preferences Suitable for OAC therapy CHA 2 DS 2 -VASc: 2 refusing OAC CHA 2 DS 2 -VASc: 2 unsuitable for OAC NOAC drugs‡ Consider Dose-adjusted LAAO, Apixaban aspirin + VKA clopidogrel or or LAA Dabigatran (INR: 2– 3) § aspirin only excision Rivaroxaban ESC, European Society of Cardiology; LAA, left atrial appendage; LAAO, left atrial appendage occlusion

Thank you for your attention
- Slides: 17