Atrial fibrillation and DM Stephen Byrne and Fiona

Atrial fibrillation and DM Stephen Byrne and Fiona Barton Cardiology CNS

Aims and objectives What is AF? How is AF diagnosed? Why do we care? Treatment options? Pharmacology of AF Case Studies Questions?
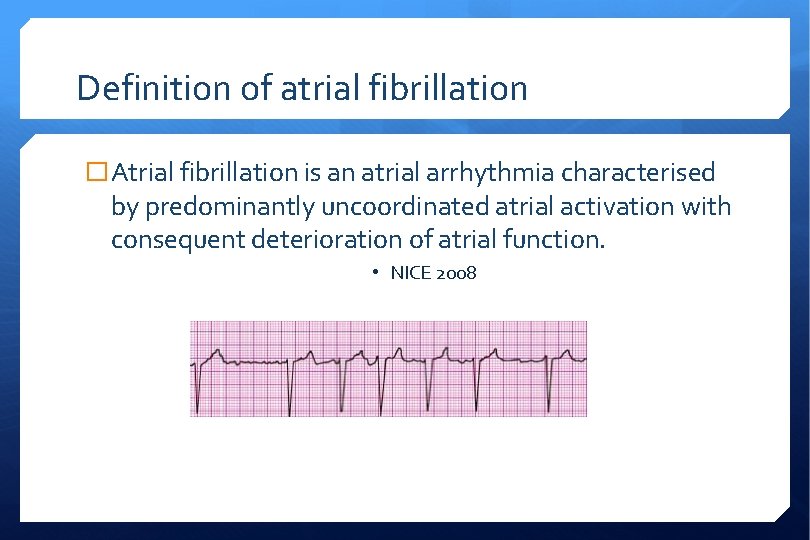
Definition of atrial fibrillation � Atrial fibrillation is an atrial arrhythmia characterised by predominantly uncoordinated atrial activation with consequent deterioration of atrial function. • NICE 2008
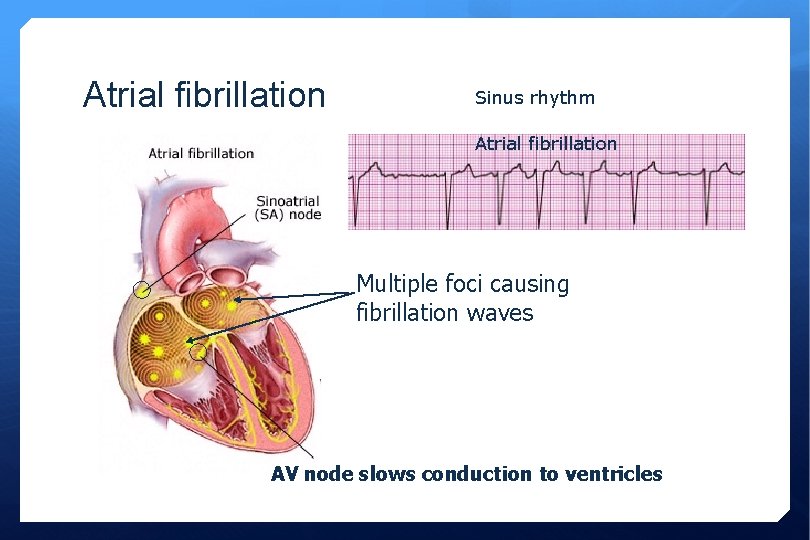
Atrial fibrillation Sinus rhythm Atrial fibrillation Multiple foci causing fibrillation waves AV node slows conduction to ventricles
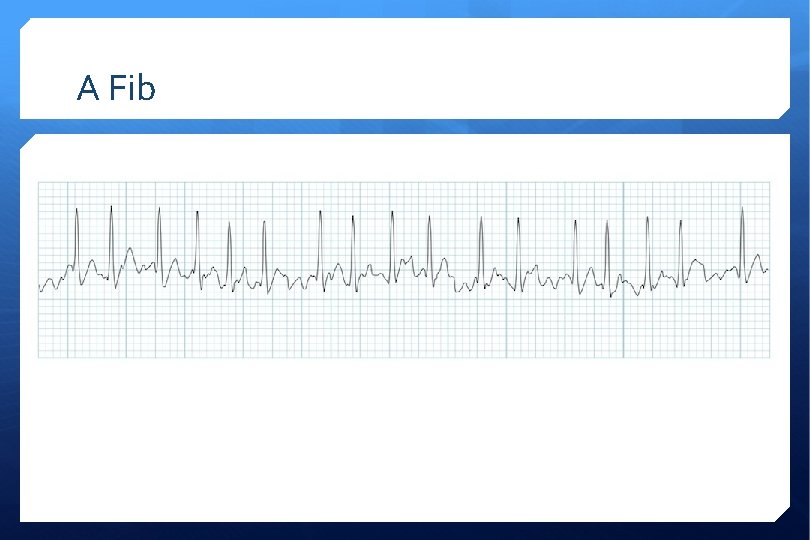
A Fib
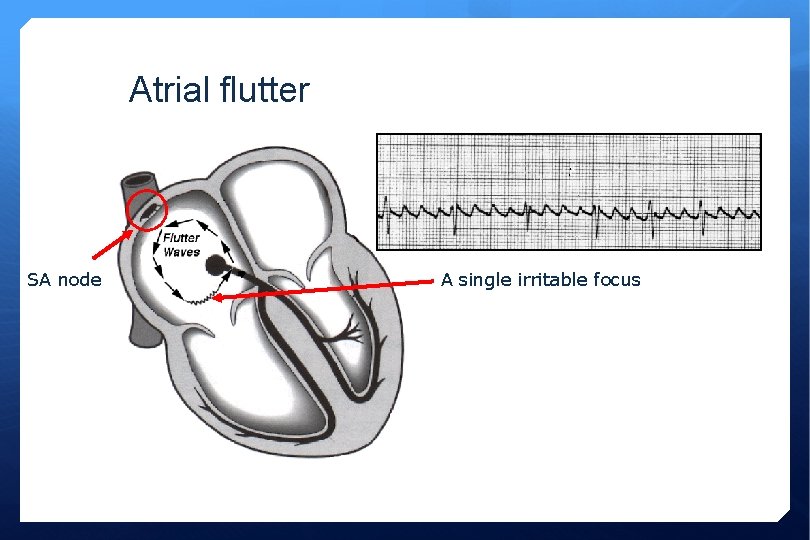
Atrial flutter SA node A single irritable focus

Diagnosing AF Pulse checks 3 lead monitor, 12 lead ECG, 24/48 hour 7 day Holter monitor If found History Clinical examination CXR ECHO Labs electrolytes thyroid renal hepatic function FBC BNP is some.
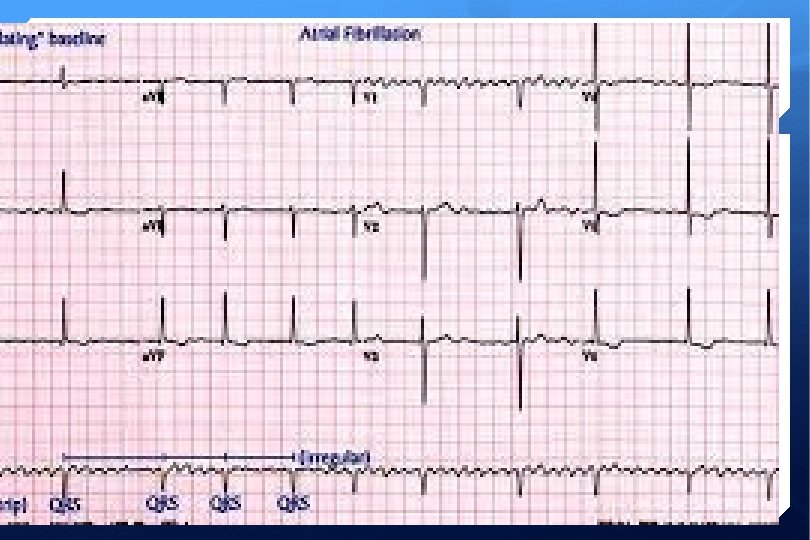
ECG

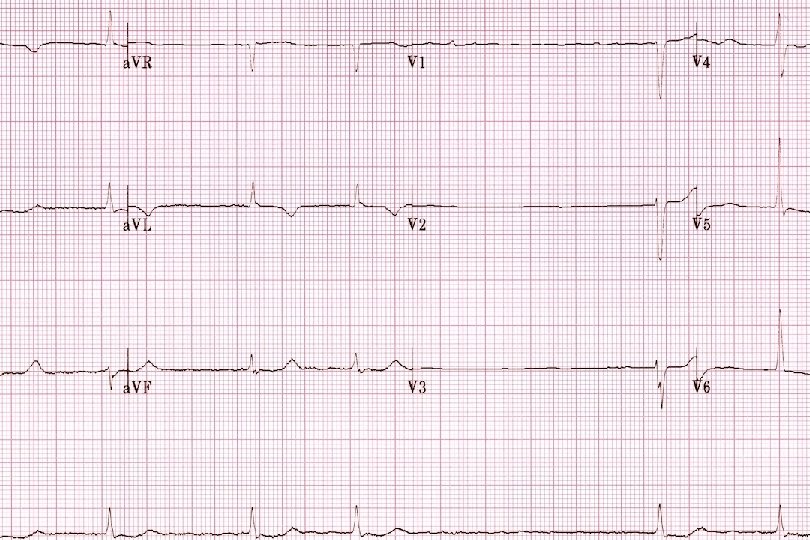

Classification Paroxysmal (23%) Self-terminating (<7 days) Persistent (38%) Lasting longer than 7 days Permanent (39%) Episodes > 1 year or unresponsive to reversal Non valvular AF is AF in the absence of rheumatic mitral stenosis, a mechanical or bioprosthetic heart valve, or mitral valve repair


Epidemiology Most common presenting arrhythmia in cardiology Affects 2% of the population Incidence of AF increasing (13% in last 20 years) Stewart et al Population, prevalence, incidence and predictors of atrial fibrillations. Heart 2001 86: 516 -521 Increasing Prevalence with age. Approx 1% of those <60 years whereas 12% of those in the 75 to 84 year age range. AHA/ASC Guidelines for management of AF JACC 2014

Why do we care? Impact of Atrial Fibrillation on the Risk of Death The Framingham Heart Study Conclusions—In subjects from the original cohort of the Framingham Heart Study, AF was associated with a 1. 5 - to 1. 9 -fold mortality risk after adjustment for the preexisting cardiovascular conditions with which AF was related. The decreased survival seen with AF was present in men and women and across a wide range of ages. Circulation 1998: 946 -952

Rotterdam Study - Ommoord 7983 participants over age 55 years 1990 – 1999 209 cases of AF noted on inception 167 developed new AF over course of study Over all incidence 5. 5% Prevalence increasing with age 55 – 60 yrs 0. 7% 85 yr and above 17. 8% Heeringa et al 2006 EHJ 27: 949 - 953

Etiology of AF Atrial dilatation and micro fibrosis are the most important factors contributing to the occurrence and maintenance of AF is associated with increase in connective tissue between cardiac cells Leads to increase deposits of collagen and fibronectin causing fibrosis Fibrosis within atrial tissues leads to the development of arrhythmias The appearance of AF is often associated with exacerbation of underlying heart disease.

Decompensation Loss of atrial contraction may markedly decrease cardiac output particularly if diastolic ventricular filling is impaired by mitral stenosis hypertension hypertrophic or restrictive cardiomyopathy. Sympathetic activation and vagal withdrawal such as with exertion or illness accelerate the ventricular response.

Contributing factors Obesity 15% increase in obesity equates to a 7. 5% increase in AF Congestive heart failure (41%) COPD (11%) Hypertension (71%) Sleep apnoea Valvular heart disease (63%) Ageing Diabetes mellitus (20%) Thyroid dysfunction (10%) Coronary artery disease (36%) Renal disease (13%) EORP-AF study Europace 2014; 16(3): 308 -319
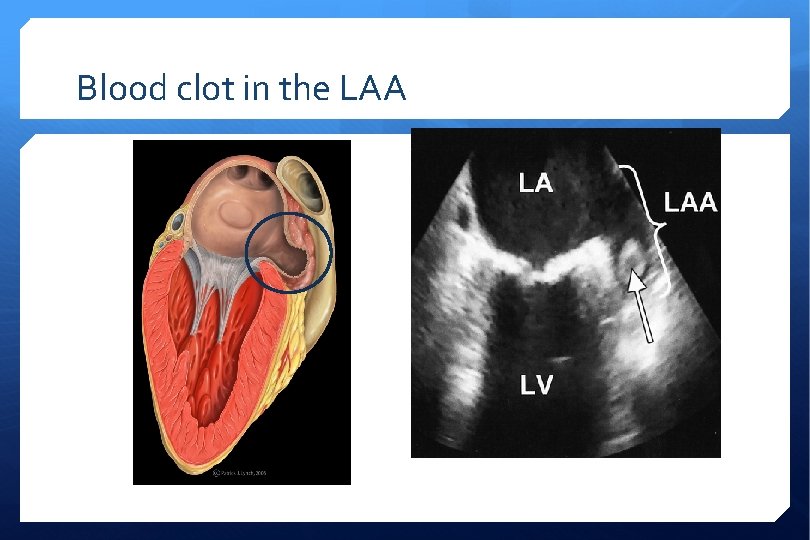
Blood clot in the LAA

Risk of Stroke. Associated with a 5 fold increase risk of stroke and stroke risk increases with age. AF related strokes are likely to be more severe then non AF strokes AHA/ACC/HRS Guidelines JACC 2014 Observed absolute stroke rates for nonanticoagulated patients with single independent risk factors were in the range of 6 to 9% per year for prior stroke/TIA, 1. 5 to 3% per year for history of hypertension, 1. 5 to 3% per year for age >75, and 2. 0 to 3. 5% per year for diabetes. Neurology August 7 2007 Vol 69 No 6 546 -554

AF non a benign issue! 5 fold increase in Stroke 3 fold increase in Heart Failure 2 fold increase in Dementia Patients with AF are hospitalized twice as often as patients without AF: Are three times more likely to have multiple admissions and 2. 1% of patients with AF died in hospital compared to 0. 1% without it.

Diabetes patients are at higher risk of AF Diabetes Mellitus is a strong independent risk factor for atrial fibrillation atrial flutter. Control DM 10. 3% A Fib 14. 9% A Fib 2. 5% A Flutter 4% A Flutter International Journal of Cardiology Vol 105 Issue 3 P 315 -318 2005

Symptoms can range from non existent to severe. Chronic fatigue Breathlessness on exertion Palpitations Syncope/ pre syncope Chest pain/ tightness on exertion Tachycardia induced cardiomyopathy Embolic stroke (5 fold risk of stroke)

Acute Management Stable or unstable? Unstable transfer to hospital Stable Rate control Consider Anticoagulation Refer for Cardiology opinion.

Initial aims of treatment Reduce symptoms Rate control Beta blockade/ calcium channel blockade Determine LV function - Echocardiogram Antiarrhythmic selection

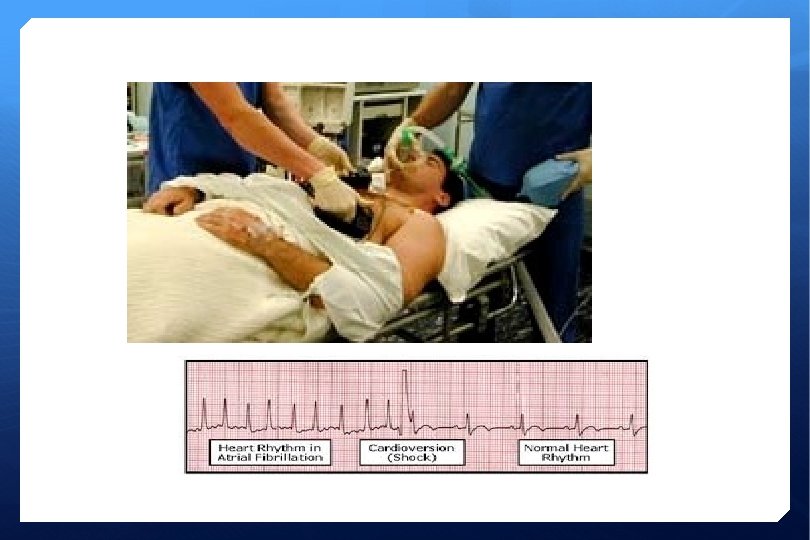
DC Cardioversion Performed under G. A. (propofol) Synchronised electrical shock AP position of pads Success rate >95% Risks 1 -2% cardiac arrest (VF/VT) – beware digoxin/ over use of rate control Stroke Skin burns Aspiration Awake within 5 -10 min, If fails – proceed to chemical then immediate DC cardioversion

Post Discharge Anticoagulation monitoring for further 6 - 8 weeks If CHADS 2 VA 2 Sc score < 2 aspirin 75 mg od If > 2 warfarin, dabigatran or rivaroxaban life-long Antiarrhythmic continues (long term) Advice for patients Avoid alcohol (specifically spirits) Avoid caffeine Avoid eating large meals If palpitations reoccur – get to hospital immediately C oronary AD H ypertension A 2 ge >75 D iabetes S 2 troke V ascular Disease A ge >65 S ex category (F)
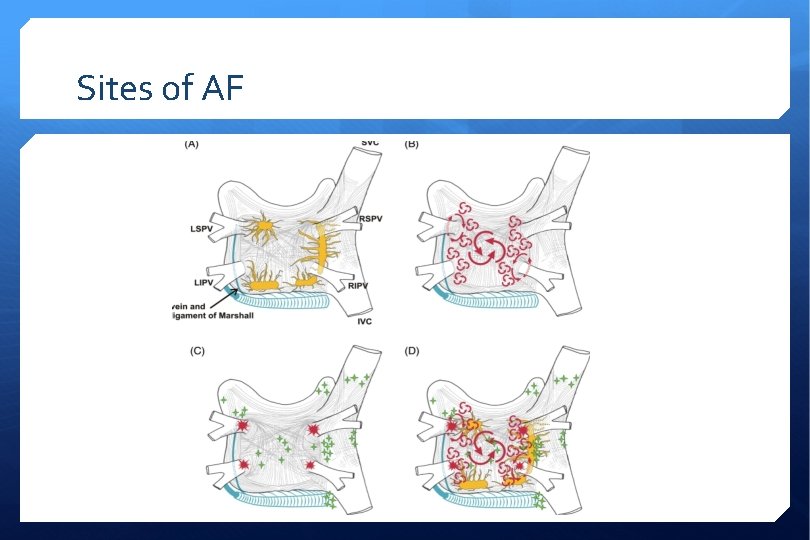
Sites of AF
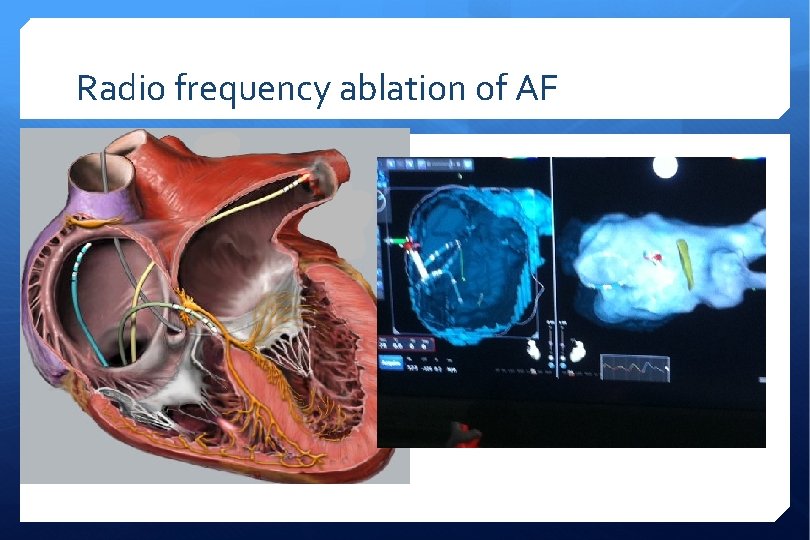
Radio frequency ablation of AF
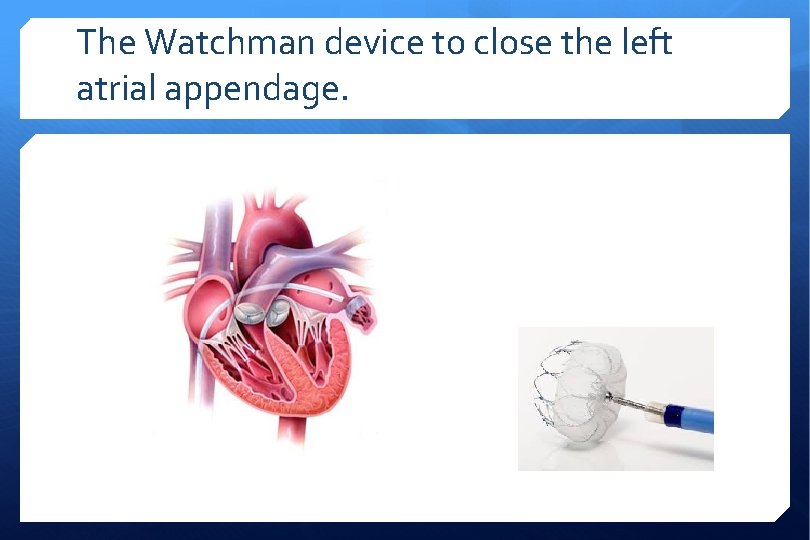
The Watchman device to close the left atrial appendage.

Rate Control and Pharmacotherpay Options
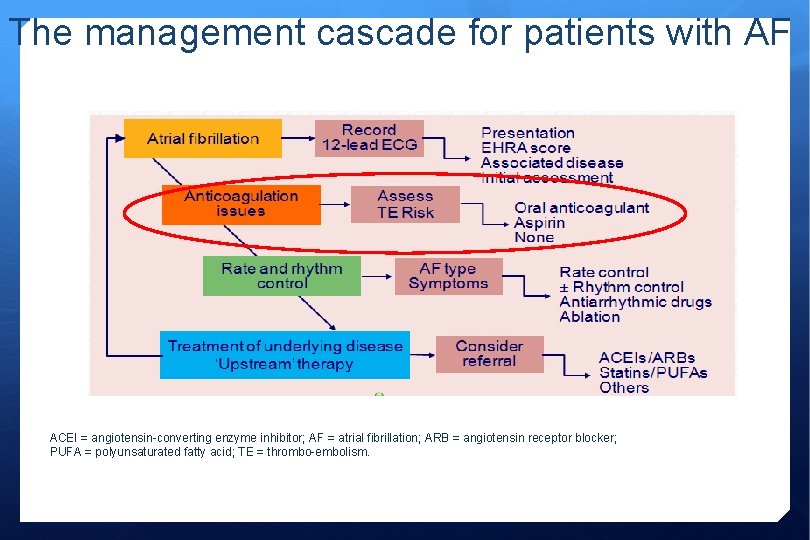
The management cascade for patients with AF ACEI = angiotensin-converting enzyme inhibitor; AF = atrial fibrillation; ARB = angiotensin receptor blocker; PUFA = polyunsaturated fatty acid; TE = thrombo-embolism.

Available Drugs Vaughan Williams Classification Class I-Sodium channel blockers quinidine, disopyramide, lignocaine flecainide, propafenone Class II-Beta blockers propranolol, atenolol, metoprolol, bisoprolol, nebivolol Class III- Potassium channel blockers sotalol, amiodarone, ibutilide Class IV-Calcium channel blockers verapamil, diltiazem Others-Digoxin, Adenosine

Class 1 drugs-Sodium channel blockers 1 a-Disopyramide-anticholinergic side effects, occasionally torsades pointes-dose is 250 mg SR bd Quinidine-not used anymore as high chance of torsades pointes. Procainamide-lupus like syndrome without renal involvement, torsades. 1 b-Lignocaine-toxicity usually in elderly patients with heart failure manifest by confusion and convulsions

Class 2. Beta blockers Class 3. -Potassium channel blockers Amiodarone, Sotalol, Ibutilide, Bretylium • Nausea, hypothyroidism, less commonly hyperthyroidism, skin discolouration-blue-gray, photosensitive rash-use sunblock, corneal depositsnight driving, hepatitis-cumulative with oral preparation, allergic with intravenous preparation, pulmonary fibrosis, torsades (unusual) Use minimum dose, usually 200 mg/d Check TFTs and LFTs and Cx. R at intervals

Class 4 -Calcium channel blockers Verapamil-constipation, hypotension, ankle and finger oedema, facial flushing, headache Diltiazem-as above but less likely to cause side-effects Digoxin, and adenosine are not classified in Vaughan. Williams classification

Types of rhythm correction DC cardioversion (20%) Chemical cardioversion (36%) Ibutilide Vernakalant Amiodarone Radiofrequency ablation (7%) EORP-AF study Europace 2014; 16(3): 308 -319

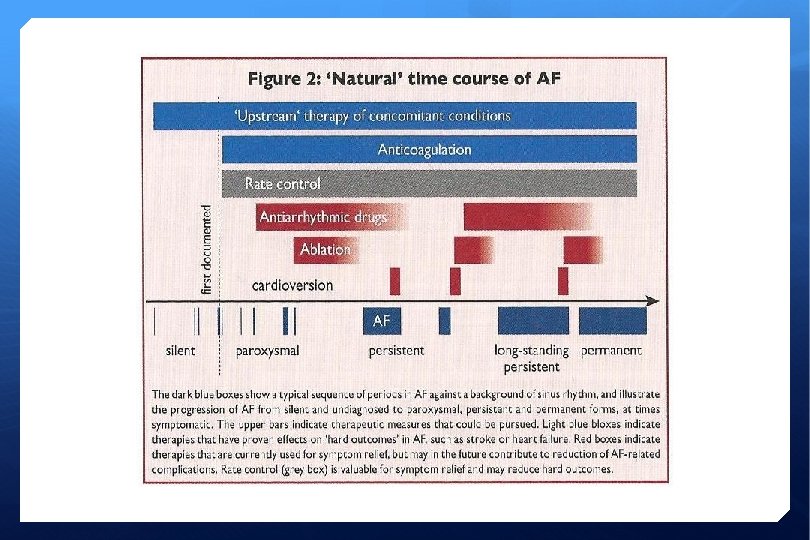
Atrial Fibrillation Treatment Depends on Whether paroxysmal, persistent or chronic Presence or absence of other heart disease Presence of diabetes, hypertension, CVA, age > 75 years Severity of symptoms Reversible precipitant Response to medication
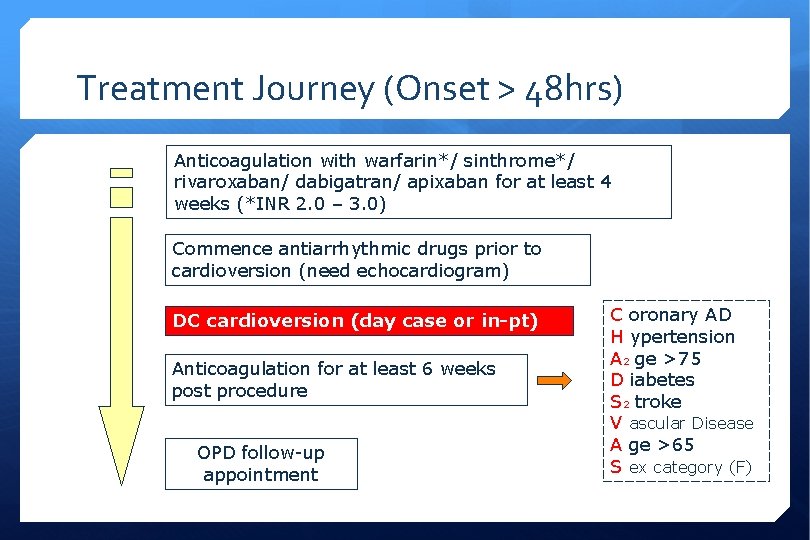
Treatment Journey (Onset > 48 hrs) Anticoagulation with warfarin*/ sinthrome*/ rivaroxaban/ dabigatran/ apixaban for at least 4 weeks (*INR 2. 0 – 3. 0) Commence antiarrhythmic drugs prior to cardioversion (need echocardiogram) DC cardioversion (day case or in-pt) Anticoagulation for at least 6 weeks post procedure OPD follow-up appointment C oronary AD H ypertension A 2 ge >75 D iabetes S 2 troke V ascular Disease A ge >65 S ex category (F)
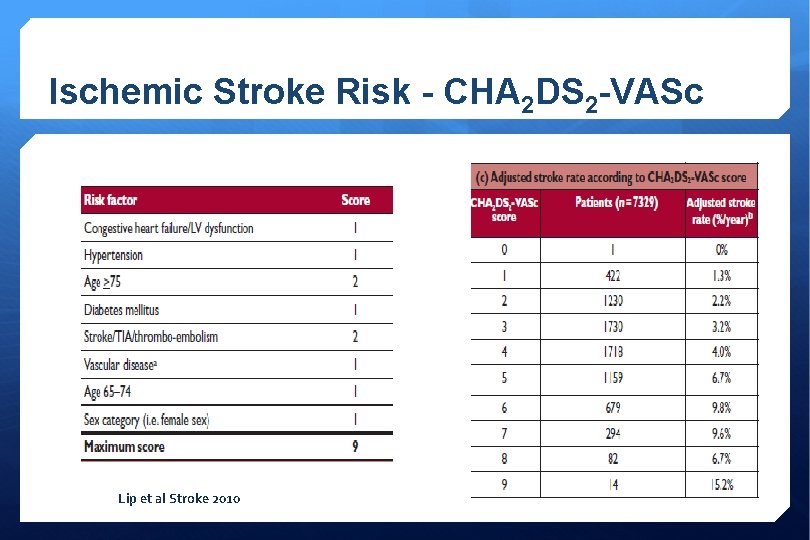
Ischemic Stroke Risk - CHA 2 DS 2 -VASc Lip et al Stroke 2010
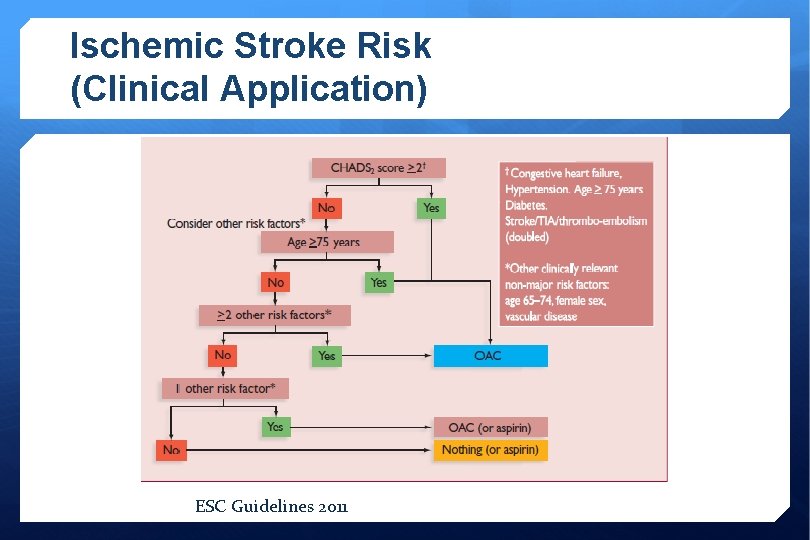
Ischemic Stroke Risk (Clinical Application) ESC Guidelines 2011
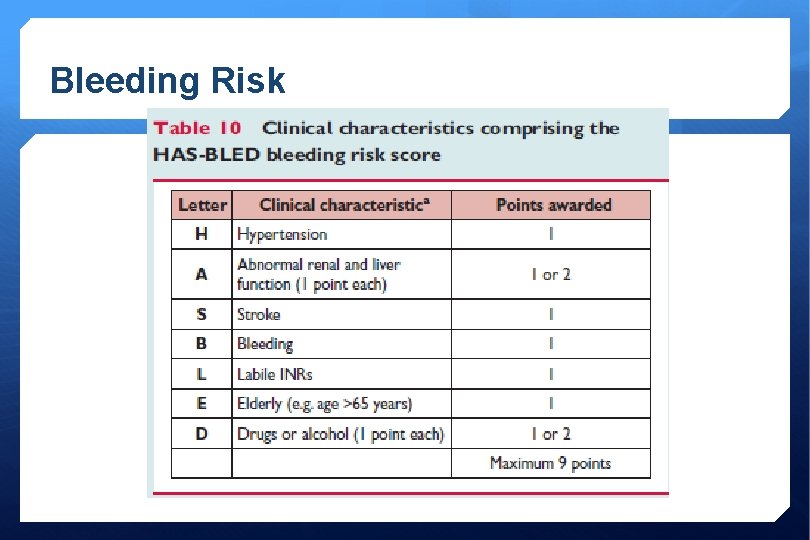
Bleeding Risk
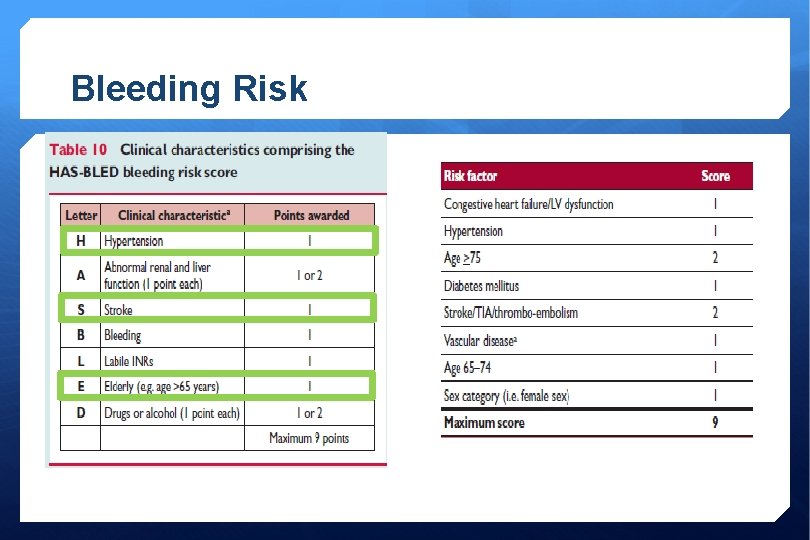
Bleeding Risk

Diagnostic Evaluation Time of onset Patients in AF with signs of HF require immediate rate control, cardioversion and echocardiogram Assess for stroke risk – CHADS 2 VA 2 Sc TFT’s, FBC, U&E, BP, ECG, fasting glucose, LFT’s

AMADEUS Trial Chronic kidney disease is associated with hypo and hypercoagulability Impact of renal function on outcomes of anticoagulated AF patients 4576 patients Mean age 70 years Apostolakis et al Eur Heart J. 2013; 34 (46): 3572 -3579

AMADEUS conclusion Mild renal impairment (Cr. Cl 60 m. L/min) doubles the risk of stroke and increased the risk of bleeding by almost 60% in anticoagulated patients with AF. Patients with a CHA 2 DS 2 VASc 1 -2 with Cr. Cl 60 m. L/min Associated with an 8 fold higher stroke risk

New Oral Anticoagulants (NOACs) Emerged as alternative to VKAs (warfarin) for thrombo-embolic prevention in non-valvular AF Predicable effect without need for monitoring Fewer drug interactions Shorter plasma half life Improved efficacy/ safety ratio
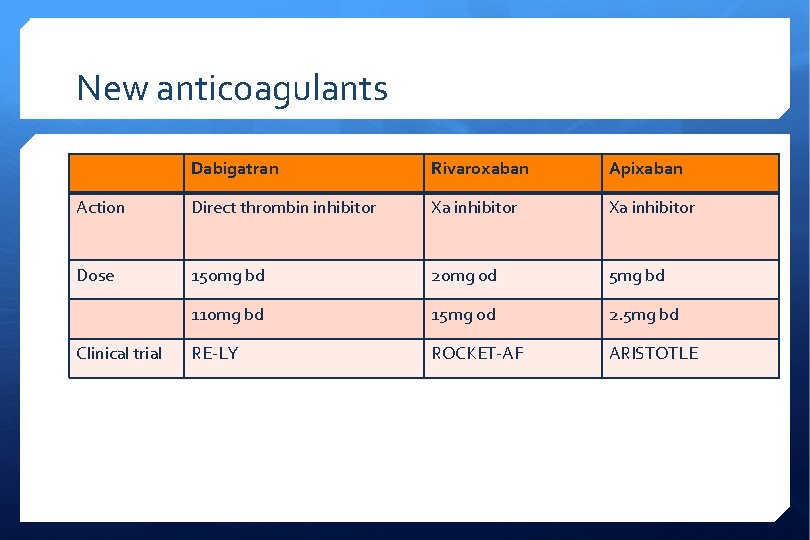
New anticoagulants Dabigatran Rivaroxaban Apixaban Action Direct thrombin inhibitor Xa inhibitor Dose 150 mg bd 20 mg od 5 mg bd 110 mg bd 15 mg od 2. 5 mg bd RE-LY ROCKET-AF ARISTOTLE Clinical trial
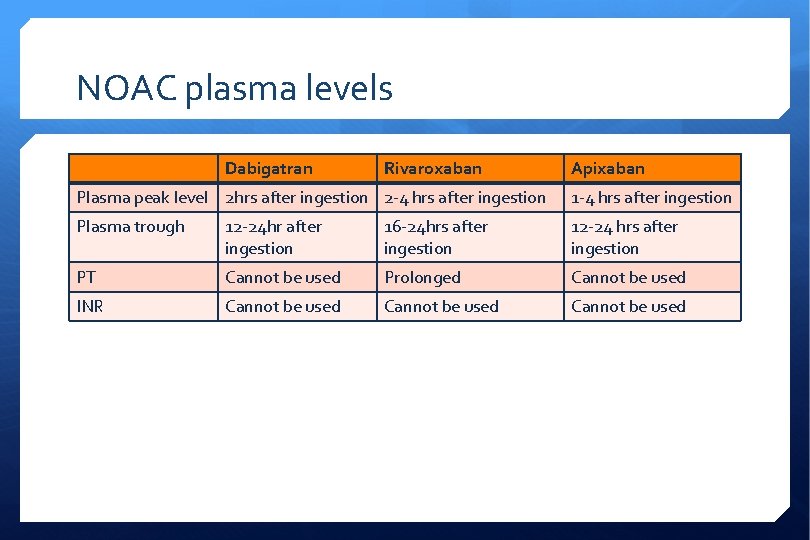
NOAC plasma levels Dabigatran Rivaroxaban Apixaban Plasma peak level 2 hrs after ingestion 2 -4 hrs after ingestion 1 -4 hrs after ingestion Plasma trough 12 -24 hr after ingestion 16 -24 hrs after ingestion 12 -24 hrs after ingestion PT Cannot be used Prolonged Cannot be used INR Cannot be used
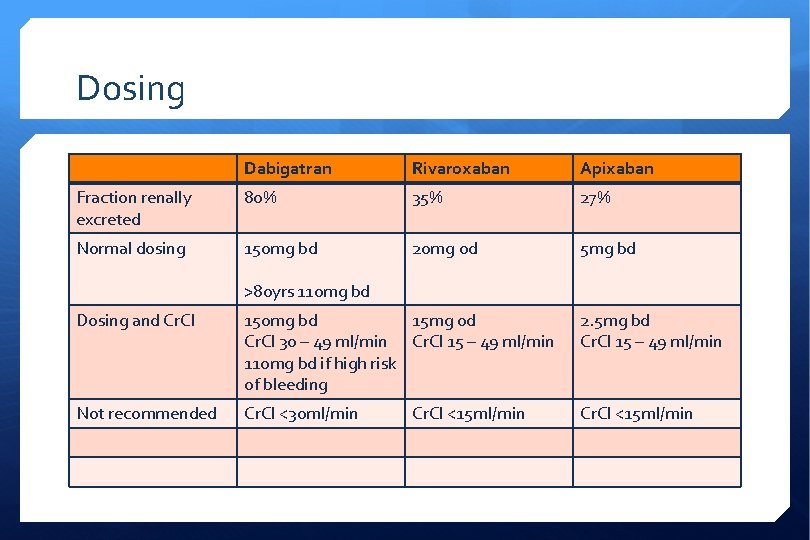
Dosing Dabigatran Rivaroxaban Apixaban Fraction renally excreted 80% 35% 27% Normal dosing 150 mg bd 20 mg od 5 mg bd >80 yrs 110 mg bd Dosing and Cr. Cl 150 mg bd 15 mg od Cr. Cl 30 – 49 ml/min Cr. Cl 15 – 49 ml/min 110 mg bd if high risk of bleeding 2. 5 mg bd Cr. Cl 15 – 49 ml/min Not recommended Cr. Cl <30 ml/min Cr. Cl <15 ml/min

Drug – Drug interactions These are new agents so thee jury is still out on drug – drug interactions, some are listed in the respective SPC E. g. Dabigatran – diclofenac risk of haemorrhage E. g. Rivaroxaban – enzyme inducers, phenytoin and carbamazepine All side effects and suspected Drug-drug interactions should be reported to the IMB

Management of bleeding in patients on dabigatran or rivaroxaban Ø There is currently no reversal agent or antidote for these medications. Ø Vitamin K is not effective. Ø In an acute overdose (<1 hr) activated charcoal may be helpful. Ø Supportive care and control of bleeding site are the mainstays of managing bleeding. Ø Possible options l l Red cell concentrate Platelet transfusions if the patient has also received anti-platelet agents. Haemodialysis may be effective in removing dabigatran but not rivaroxaban. Coagulation factors

Management of bleeding in patients on dabigatran or rivaroxaban Ø There is currently no reversal agent or antidote for these medications. Ø Vitamin K is not effective. Ø In an acute overdose (<1 hr) activated charcoal may be helpful. Ø Supportive care and control of bleeding site are the mainstays of managing bleeding. Ø Possible options l l Red cell concentrate Platelet transfusions if the patient has also received anti-platelet agents. Haemodialysis may be effective in removing dabigatran but not rivaroxaban. Coagulation factors

Swallowing Difficulties Pradaxa (Dabigatran) should be swallowed as a whole with water, with or without food. (1) Patients should be instructed not to open the capsule as this may increase the risk of bleeding. (1) The oral bioavailability may be increased by 75 % compared to the reference capsule formulation when the pellets are taken without the Hydroxypropylmethylcellulose (HPMC) capsule shell. .

Common Side effects (other than bleeding) Ø Dabigatran l Abdominal pain l Diarrhoea l Nausea l Dyspepsia Ø Rivaroxaban l GI side effects - Abdominal pain, diarrhoea, nausea, dyspepsia, constipation l Pruritus, rash (allergy uncommon) l Fever l Peripheral oedema l Fatigue l Headache, dizziness, syncope l Tachycardia, hypotension

Switching to/ from warfarin Warfarin to Dabigatran: Stop warfarin and start dabigatran when INR less than 2. Warfarin to Rivaroxaban: Stop warfarin and start rivaroxaban when INR less than 3 (n. b. rivaroxaban cause false high INR) Dabigatran to Warfarin: If Cr. Cl >50 ml/min – start warfarin 3 days before stopping dabigatran If Cr. Cl 30 -50 ml/min – start warfarin 2 days before stopping dabigatran Rivaroxaban to Warfarin Give both agents concurrently until INR is 2 or greater Test INR prior to next dose of rivaroxaban during this period

Missed Doses Ø Dabigatran l Take dose if up to 6 hours late. l Omit dose if over 6 hours late l Do not double up next dose. Ø Rivaroxaban l Take the tablet when you remember l Do not take more than 1 tablet in 1 day

Predictors for long term SR Short duration of AF Antiarrhythmic therapy Left atrial size Avoidance of triggers Upstream therapy

Upstream therapy to prevent fibrosis ACE inhibitors Statins Angiotension receptor blockers (ARBs) ? ? Omega 3


Questions? ?

Thank you
- Slides: 64