AtomsMoleculesIons Dr Ron Rusay Spring 2008 Copyright 2008














































- Slides: 54

Atoms-Molecules-Ions Dr. Ron Rusay Spring 2008 © Copyright 2008 R. J. Rusay

Aspects of Matter • Elements, Compounds, and Mixtures (Part 1) • Atomic Theory Today • Elements: The Periodic Table • Ions & Compounds • Compounds: Introduction to Ionic Bonding • Compounds: Formulas & Names (Part 2) • Mixtures: Separation Techniques (Part 3) • Atomic & Molar Masses (Part 4)

Modern History of the Atom ð 1909: Millikan determines charge and mass of e- ð 1913 -19: Rutherford & Bohr’s atom; The proton. http: //www. yrbe. edu. on. ca/~mdhs/science/chemistry/ch 2_2. htm ð 1927: Waves & Particles, Quantum Mechanics http: //www. nmsi. ac. uk/on-line/electron/section 3/1927. html ð 1932: James Chadwick “discovers” the neutron http: //www. nmsi. ac. uk/on-line/electron/section 3/1932 a. html © Copyright 1998 - 2008 R. J. Rusay

CHEMISTRY of the Atom Ernest Rutherford (1871 -1937)

Modern History of the Atom 1897: J. J. Thomson “discovers” the electron: http: //www. nmsi. ac. uk/on-line/electron/section 2/ Photo © The Nobel Foundation http: //pl. nobel. se/laureates/physics-1906 -1 -bio. html © Copyright 1998 - 2008 R. J. Rusay

Milliken Oil Drop Experiment

How does an atom relate to nanotechnology? Consider Powers of 10 (10 x) http: //www. eamesoffice. com/powers_of_ten. html http: //www. powersof 10. com/ Earth = 12, 760, 000 meters wide (12. 76 x 10 6), 12. 76 million meters (megameters) Plant Cell = 0. 00001276 meters wide (12. 76 x 10 -6) (12. 76 millionths of a meter) (12, 760 nanometers!) Nano scale is regarded as < 1, 000 nanometers ~1/50 the diameter of a human hair (anything less than a micron (10 -6 m). Chemists typically think in views and images of < 1 nanometer (eg. bond lengths and atomic sizes).

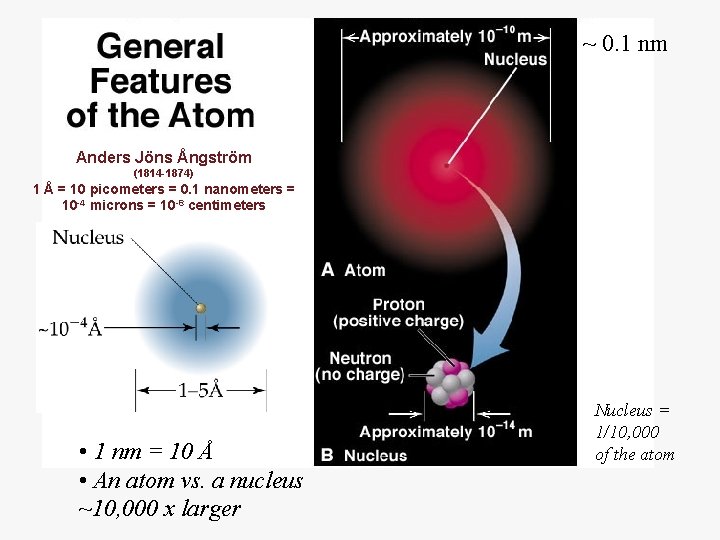
~ 0. 1 nm Anders Jöns Ångström (1814 -1874) 1 Å = 10 picometers = 0. 1 nanometers = 10 -4 microns = 10 -8 centimeters • 1 nm = 10 Å • An atom vs. a nucleus ~10, 000 x larger Nucleus = 1/10, 000 of the atom

Can we “see” and manipulate atoms using a microscope? Yes, using atomic force microscopy (AFM) and a variety of instruments such as Scanning Transmission Electron Microscopes. TEAM 0. 5: LBL’s Latest (2008) Transmission Electron Aberration-corrected Microscope Resolution: • 1 nm = 10 Å +/- 0. 5 Å • An atom vs. a nucleus ~10, 000 x larger

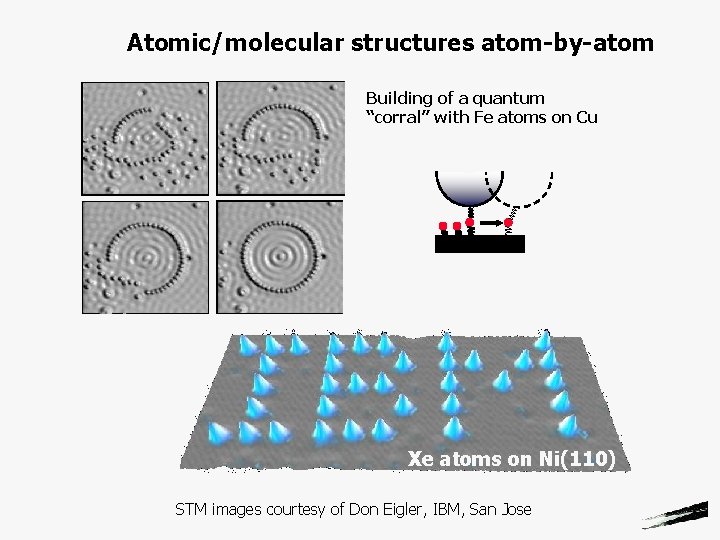
Atomic/molecular structures atom-by-atom Building of a quantum “corral” with Fe atoms on Cu Xe atoms on Ni(110) STM images courtesy of Don Eigler, IBM, San Jose

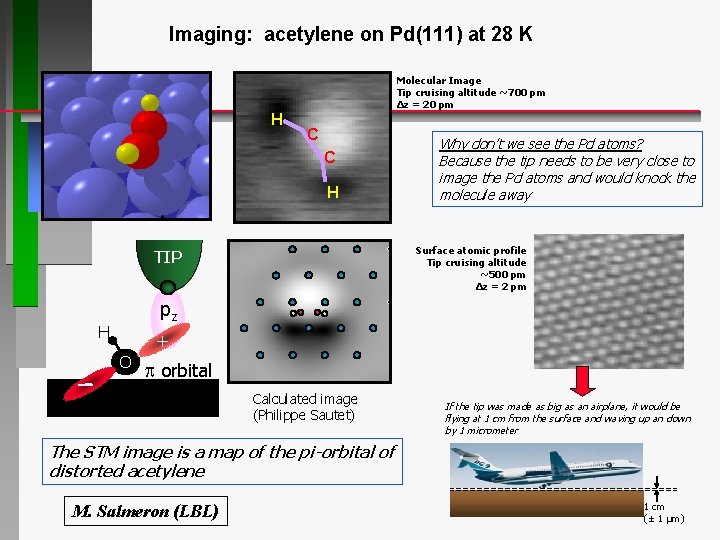
Imaging: acetylene on Pd(111) at 28 K Molecular Image Tip cruising altitude ~700 pm Δz = 20 pm H C C H Why don’t we see the Pd atoms? Because the tip needs to be very close to image the Pd atoms and would knock the molecule away Surface atomic profile Tip cruising altitude ~500 pm Δz = 2 pm TIP pz H + O p orbital Calculated image (Philippe Sautet) If the tip was made as big as an airplane, it would be flying at 1 cm from the surface and waving up an down by 1 micrometer The STM image is a map of the pi-orbital of distorted acetylene M. Salmeron (LBL) 1 cm (± 1 μm)

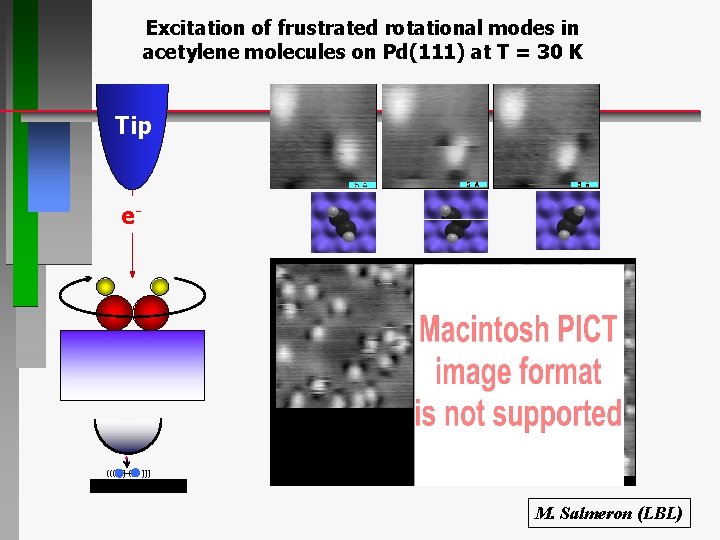
Excitation of frustrated rotational modes in acetylene molecules on Pd(111) at T = 30 K Tip e- ((( ) ( ))) M. Salmeron (LBL)

CHEMISTRY of the Atom FUNDAMENTAL PARTICLES: Mass Charge Nucleus: ð PROTON 1 amu +1 Symbol H+, H, p • 1. 67 x 10 -27 kg ð NEUTRON 1 amu 0 n • 1. 67 x 10 -27 kg _____________________ ð ELECTRON very small -1 e • 2000 x smaller than a proton or neutron The particle is said to “hold” or “bond” atoms together in molecules. © Copyright 1998 -2008 R. J. Rusay

Periodic Table • Mendeleev’s Table 1868 -1871 Mural at St. Petersburg University, Russia © Copyright 1998 -2008 R. J. Rusay


QUESTION The element found in the 6 A family (or group 16) and period four can be toxic and a micronutrient. What is the symbol for that element? You can use any help aid to answer this question except a breathing resource. 1. 2. 3. 4. Sb As Se Te

ANSWER Choice 3 pinpoints an element in both the 6 A column and row four. Remember to include the first row of Hydrogen and Helium when counting rows. Section 2. 7: An Introduction to the Periodic Table

Chemical Symbols & Historical Names What is the symbol for gold and what was its original name?

QUESTION

ANSWER 1) Phosphorus, Ph Pr Section 2. 7 An Introduction to the Periodic Table (p. 55) Phosphorus has a symbol of P.


QUESTION Of the following which would not be considered a metalloid? 1. Ge 2. Sb 3. Se 4. Spockonium, element #182 in the Star Trek Periodic Table.

ANSWER Choice 3, Se, is the only element (without considering the possibility of the fictional Spockonium) listed that is not on the accepted Periodic Table’s “stairway” between metals and nonmetals, therefore it is the only non-metalloid listed. Section 2. 7: An Introduction to the Periodic Table

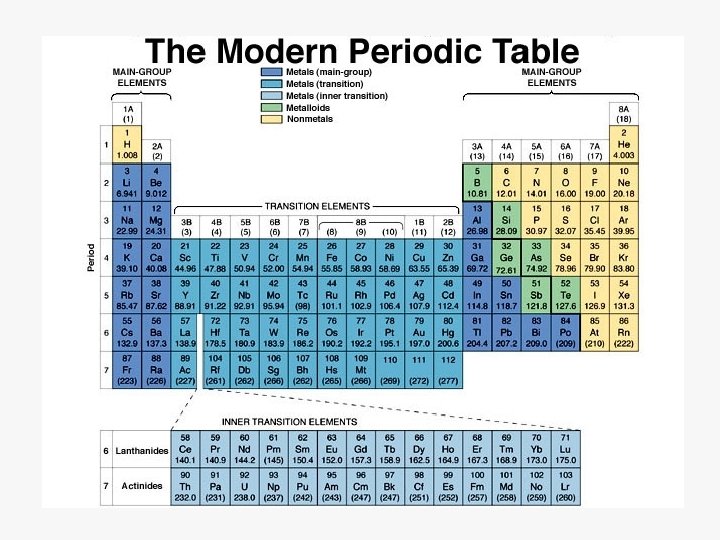
Periodic Table ð Elements are classified by: properties & atomic number metals, non-metals, metalloids ð Groups or Families (vertical) 1 A = alkali metals 2 A = alkaline earth metals 6 A (16) = chalcogens 7 A (17) = halogens 8 A (18) = noble gases ð Periods (horizontal) numbers 1 -7

Group 1 and Group 2 Metals

Using the Periodic Table

Atoms, Molecules & Ions ð Atoms (neutral electrostatic charge: # protons • • = # electrons ) # Protons = Atomic Number Atomic Mass = # Protons + # of Neutrons • Isotope: same atomic number but different atomic mass (different # of neutrons) © Copyright 1998 -2008 R. J. Rusay

QUESTION

ANSWER 4) a, d and b, e Section 2. 5 The Modern View of Atomic Structure: An Introduction (p. 49) Isotopes have the same number of protons and different numbers of neutrons, so they can be organized by atomic number.

Atoms, Molecules & Ions • Isotopes vary in their relative natural abundance. • Periodic Table’s atomic mass is a weighted average of all isotopic masses • The mass of sodium, Na, element #11 is listed as 22. 99 amu. Which isotope is naturally present in the larger amount: the isotope with 12 neutrons or with 13 neutrons? (There is a small percentage of the isotope with 11 neutrons. ) © Copyright 1998 -2008 R. J. Rusay

Atoms, Molecules & Ions ð Atomic Mass of Carbon: What is the “weighted” atomic mass? 12. 00000 x 98. 98/100 + 13. 00335 x 1. 011 = © Copyright 1998 -2008 R. J. Rusay 11. 8776 + 0. 13146 = 12. 0090 ð 12. 01

Atoms, Molecules and Ions • • • Atomic Number = 6 (atom’s identity) Carbon Atomic Mass = 13 (isotope 13) 6 protons; # neutrons = 13 - 6 neutral atom has 6 electrons © Copyright 1998 -2008 R. J. Rusay

QUESTION

ANSWER 1) 0%. Section 3. 1 Atomic Mass (p. 77) The 12. 011 is an average of the isotopic masses. None of the isotopes has an exact mass of 12. 011.

Atomic Symbols Mass number 39 K 19 Element Symbol Atomic number Also written as 39 K

Atoms, Molecules and Ions • • • Atomic Number = 12 (atom’s identity) Atomic Mass = 24 12 protons; # neutrons = 24 - 12 neutral atom has 12 electrons Ion contains 10 electrons: symbol? • 12 p+ + 10 e- = +2 © Copyright 1998 -2008 R. J. Rusay

Atoms, Molecules and Ions • • • Atomic Number = 17 (atom’s identity) Atomic Mass = ? # protons = ? ; # neutrons = ? neutral atom has ? electrons Ion contains 18 electrons: symbol? • 17 p+ + 18 e- = -1 © Copyright 1998 -2008 R. J. Rusay

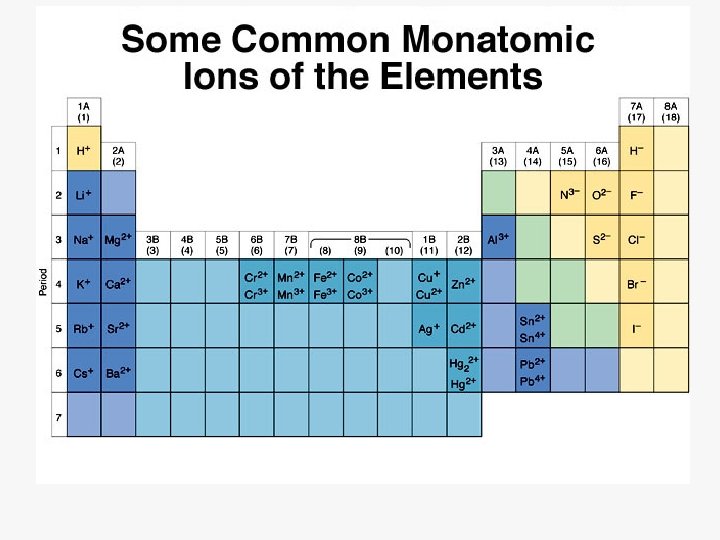
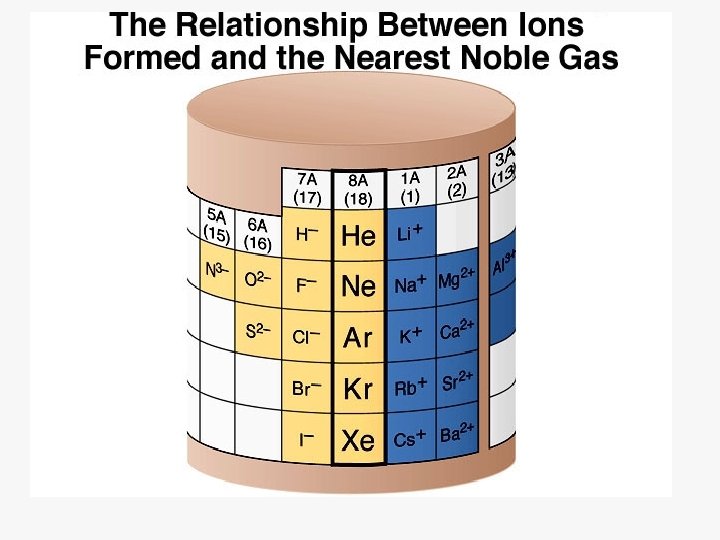
Ions ð Cation: ð Anion: ð Ionic A positive ion ð Mg 2+, NH 4+ A negative ion ð Cl , SO 42 Bonding: Force of attraction between oppositely charged ions.

QUESTION Calcium plays several critical roles in the functioning of human cells. However, this form of calcium is the ion made with 20 protons and 18 electrons. Therefore the ion would be… 1. 2. 3. 4. positive and called an anion. positive and called a cation. negative and called an anion. negative and called a cation.

ANSWER Choice 2 provides the correct relationship and name for an atom of calcium (20 protons = 20+) that has lost two electrons (now with 18–). The ion would have a +2 charge. Positive ions are called cations. Section 2. 6: Molecules and Ions

QUESTION Of the following, which would NOT qualify as an isotope of 35 Cl? 1. 2. 3. 4. 36 Cl 35 Cl– 37 Cl

ANSWER Choice 2 is NOT an isotope of 35 Cl. All three choices represent a form of chlorine but choice 2 has the same number of neutrons as the atom in the question, therefore it does not fit the criteria for isotopes (i. e. variable neutron number with constant proton number). Isotopes can be ions as well as neutral atoms. 37 Cl– and 37 Cl are also not isotopes. Section 2. 5: The Modern View of Atomic Structure: An Introduction

Worksheet: Atoms I Lab Manual: Pg. 157


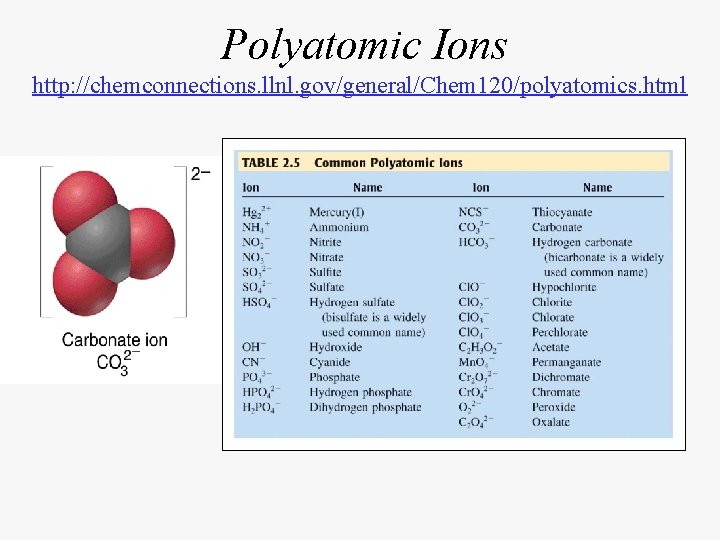
Polyatomic Ions http: //chemconnections. llnl. gov/general/Chem 120/polyatomics. html

Molecules ð Neutrally Charged ð Eg. Salt: Na. Cl -> 1 Na+ and 1 Cl ð What is the proportion of ions for a compound formed from Mg ion and chlorine? ð Mg 2+ and Cl ð 1 Mg 2+ combines with 2 Cl


Ionic vs. Covalent

Ionic vs. Covalent Metals generally combine with non- metals to form ionic compounds. Electrons are “lost” by the metal and “gained” by the non-metal following the octet rule. ð Non-metals generally combine with nonmetals to form covalent compounds where electrons are “shared”. Each pair of electrons ia a covalent bond. Eg. H 2 O ð Polyatomic ions have both covalent and ionic properties. Eg. hydroxide, OHð

QUESTION

ANSWER 2) Ions are formed by changing the number of protons in an atom’s nucleus. Section 2. 6 Molecules and Ions (p. 52) The removal of a proton from the nucleus requires extraordinary amounts of energy. It will only happen during a nuclear reaction.

Chemical Formulas ð Molecular Formula: Elements’ Symbols = atoms Subscripts = relative numbers of atoms ð How many atoms of each element are in the following componds? Mg. Cl 2 CCl 4 Na. OH (NH 4 )2 CO 3 C 20 H 26 N 2 O (Ibogaine, not ionic)

QUESTION

ANSWER 4) 8 Section 2. 8 Naming Simple Compounds (p. 57) 57 Remember to multiply the number of oxygen atoms in the parentheses by the subscript outside of the parentheses.