ATOMS THE BUILDING BLOCKS OF MATTER The Greek

ATOMS THE BUILDING BLOCKS OF MATTER

The Greek Model n Democritus concluded that matter could not be divided into smaller and smaller pieces forever. • The smallest piece would be obtained and would be. • He named this smallest piece of matter an. • He hypothesized that atoms were _____ and made of the same material.

Dalton’s Model n n All elements are composed of atoms. Atoms of the same element are exactly _____. Atoms of different elements are _______. Compounds are formed by joining of atoms of _____ elements.

Thomson’s Model n The atom is made of a puddinglike positively charged material throughout which negatively charged (_______) electrons were scattered, like plums in a pudding!

Rutherford’s Model n n Proposed that an atom is mostly empty space with a dense, positively charged ______in the center. Negatively charged electrons were scattered outside nucleus.
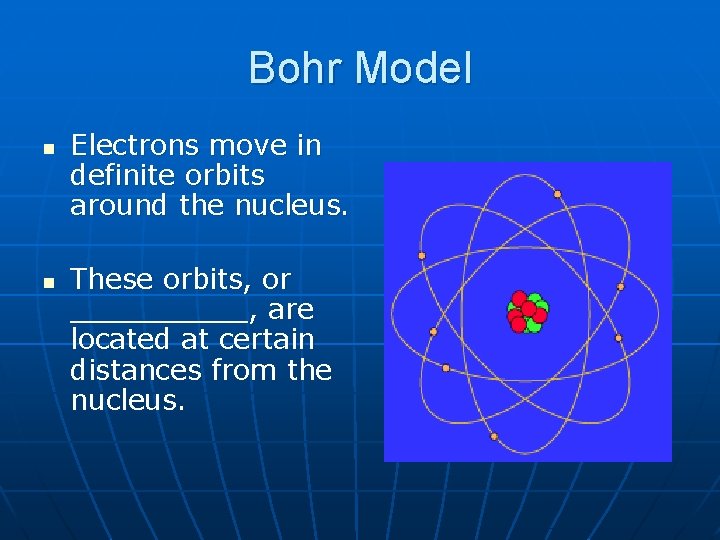
Bohr Model n n Electrons move in definite orbits around the nucleus. These orbits, or _____, are located at certain distances from the nucleus.

Wave Model n n n Electrons do not move around an atom in a _______ path. Scientists can only predict where an electron may be. Location is based on how much _____ the electron has.

Modern Atomic Theory n Based on Rutherford’s Model, Bohr’s Model and wave mechanics. • An atom has a small, positively charged nucleus surrounded by a large region in which there are enough electrons to make the atom _______.
- Slides: 8