Atoms The Building Blocks of Matter Honors Chemistry

Atoms: The Building Blocks of Matter Honors Chemistry Mrs. Partridge

Law of Conservation of Mass is neither created nor destroyed during chemical or physical reactions. Total mass of reactants = Total mass of products Antoine Lavoisier

Dalton’s Atomic Theory (1808) q All matter is composed of extremely small particles called atoms q Atoms of a given element are identical in size, mass, and other properties; atoms of different John Dalton elements differ in size, mass, and other properties q Atoms cannot be subdivided, created, or destroyed q Atoms of different elements combine in simple whole -number ratios to form chemical compounds q In chemical reactions, atoms are combined, separated, or rearranged

Modern Atomic Theory Several changes have been made to Dalton’s theory. Dalton said: Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties Modern theory states: Atoms of an element have a characteristic average mass which is unique to that element.
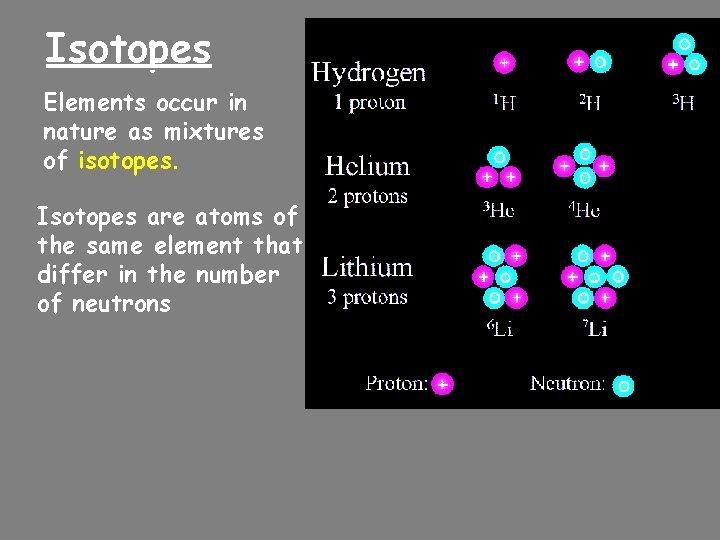
Isotopes Elements occur in nature as mixtures of isotopes. Isotopes are atoms of the same element that differ in the number of neutrons

Atomic Masses Atomic mass is the average of all the naturally isotopes of that element. Carbon = 12. 011 Isotope Symbol Composition of the nucleus % in nature Carbon-12 12 C 6 protons 6 neutrons 98. 89% Carbon-13 13 C 6 protons 7 neutrons 1. 11% Carbon-14 14 C 6 protons 8 neutrons <0. 01%

Modern Atomic Theory #2 Dalton said: Atoms cannot be subdivided, created, or destroyed Modern theory states: Atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions!
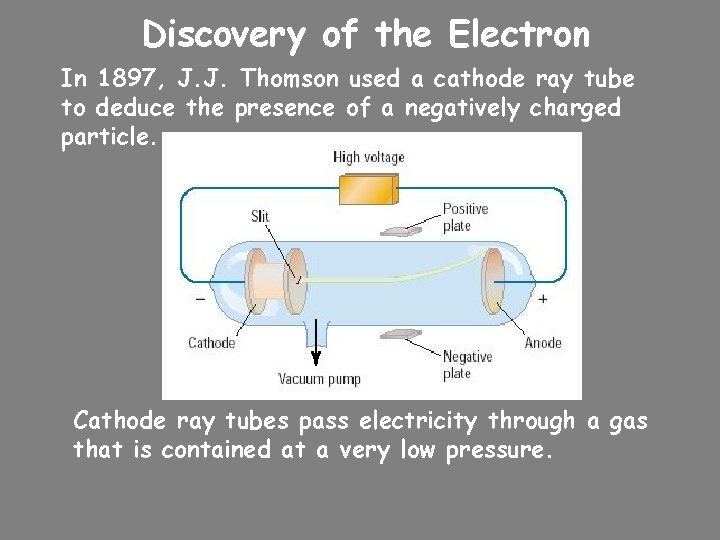
Discovery of the Electron In 1897, J. J. Thomson used a cathode ray tube to deduce the presence of a negatively charged particle. Cathode ray tubes pass electricity through a gas that is contained at a very low pressure.

Some Cathode Modern Ray Tubes

Thomson’s Atomic Model Thomson believed that the electrons were like plums embedded in a positively charged “pudding, ” thus it was called the “plum pudding” model.

Mass of the Electron 1909 – Robert Millikan determines the mass of the electron. The oil drop apparatus Mass of the electron is 9. 109 x 10 -31 kg

Conclusions from the Study of the Electron q Cathode rays have identical properties regardless of the element used to produce them. All elements must contain identically charged electrons. q. Atoms are neutral, so there must be positive particles in the atom to balance the negative charge of the electrons q Electrons have so little mass that atoms must contain other particles that account for most of the mass

Rutherford’s Gold Foil Experiment q Alpha particles are helium nuclei q Particles were fired at a thin sheet of gold foil q Particle hits on the detecting screen (film) are recorded

Try it Yourself! In the following pictures, there is a target hidden by a cloud. To figure out the shape of the target, we shot some beams into the cloud and recorded where the beams came out. Can you figure out the shape of the target?
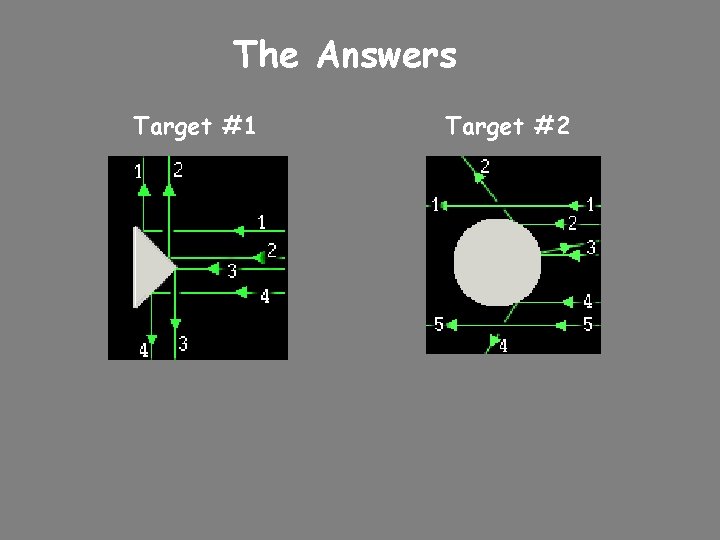
The Answers Target #1 Target #2

Rutherford’s Findings q Most of the particles passed right through q A few particles were deflected q VERY FEW were greatly deflected “Like howitzer shells bouncing off of tissue paper!” Conclusions: q The nucleus is small q The nucleus is dense q The nucleus is positively charged

Atomic Particles Particle Charge Electron -1 9. 109 x 10 -31 Electron cloud Proton +1 1. 673 x 10 -27 Nucleus 0 1. 675 x 10 -27 Nucleus Neutron Mass (kg) Location

The Atomic Scale § Most of the mass of the atom is in the nucleus (protons and neutrons) § Electrons are found outside of the nucleus (the electron cloud) § Most of the volume of the atom is empty space “q” is a particle called a “quark”
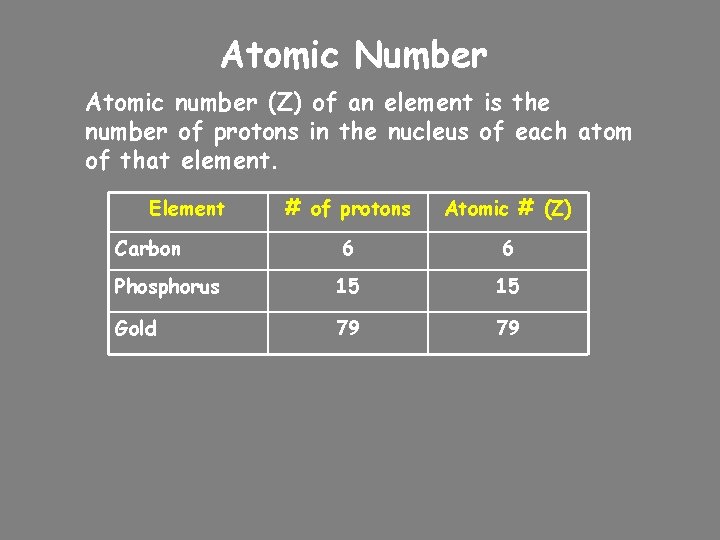
Atomic Number Atomic number (Z) of an element is the number of protons in the nucleus of each atom of that element. Element # of protons Atomic # (Z) 6 6 Phosphorus 15 15 Gold 79 79 Carbon

Isotopes…Again (must be on the test) Isotopes are atoms of the same element having different masses due to varying numbers of neutrons. Isotope Protons Electrons Neutrons Hydrogen– 1 (protium) 1 1 0 Hydrogen-2 (deuterium) 1 1 1 Hydrogen-3 (tritium) 1 1 2 Nucleus
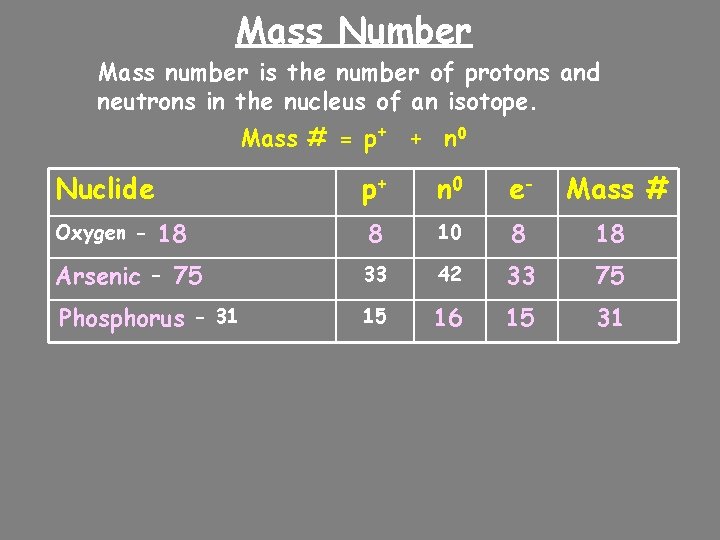
Mass Number Mass number is the number of protons and neutrons in the nucleus of an isotope. Mass # = p+ + n 0 Nuclide p+ n 0 e- 8 10 8 18 Arsenic - 75 33 42 33 75 Phosphorus - 31 15 16 15 31 Oxygen - 18 Mass #

Arrangement of Electrons in Atoms Electrons in atoms are arranged as LEVELS (n) SUBLEVELS (l) ORBITALS (ml)

QUANTUM NUMBERS The shape, size, and energy of each orbital is a function of 3 quantum numbers which describe the location of an electron within an atom or ion n (principal) ---> energy level l (orbital) ---> shape of orbital ml (magnetic) ---> designates a particular suborbital The fourth quantum number is not derived from the wave function s (spin) ---> spin of the electron (clockwise or counterclockwise: ½ or – ½)

QUANTUM NUMBERS So… if two electrons are in the same place at the same time, they must be repelling, so at least the spin quantum number is different! The Pauli Exclusion Principle says that no two electrons within an atom (or ion) can have the same four quantum numbers. If two electrons are in the same energy level, the same sublevel, and the same orbital, they must repel. Think of the 4 quantum numbers as the address of an electron… Country > State > City > Street

Energy Levels • Each energy level has a number called the PRINCIPAL QUANTUM NUMBER, n • Currently n can be 1 thru 7, because there are 7 periods on the periodic table
Energy Levels n=1 n=2 n=3 n=4
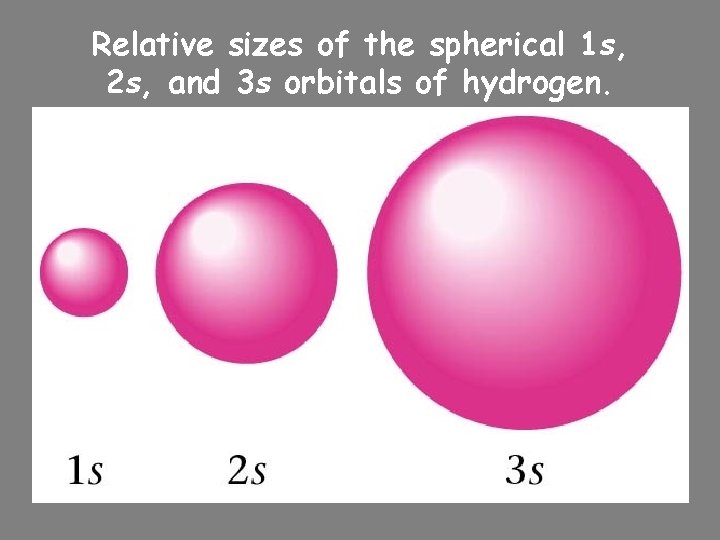
Relative sizes of the spherical 1 s, 2 s, and 3 s orbitals of hydrogen.

Types of Orbitals • The most probable area to find these electrons takes on a shape • So far, we have 4 shapes. They are named s, p, d, and f. • No more than 2 e- assigned to an orbital – one spins clockwise, one spins counterclockwise
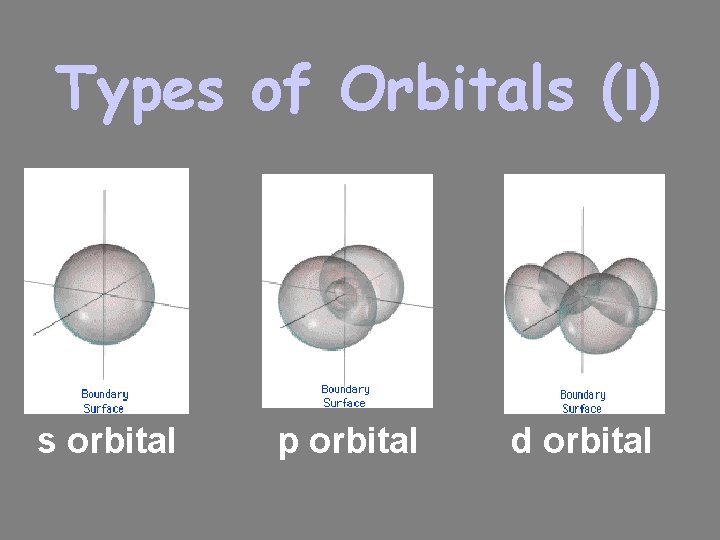
Types of Orbitals (l) s orbital p orbital d orbital
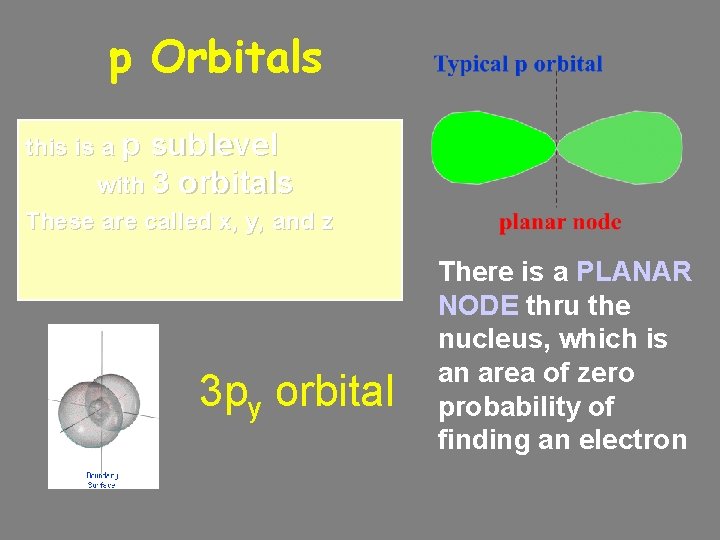
p Orbitals this is a p sublevel with 3 orbitals These are called x, y, and z 3 py orbital There is a PLANAR NODE thru the nucleus, which is an area of zero probability of finding an electron
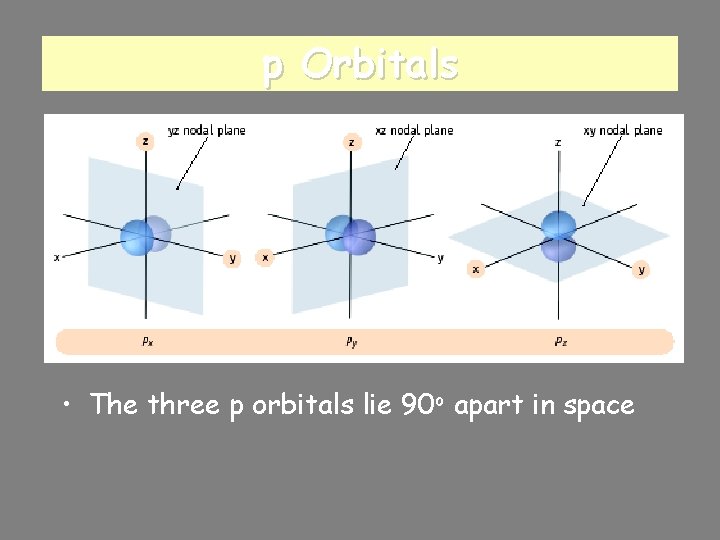
p Orbitals • The three p orbitals lie 90 o apart in space
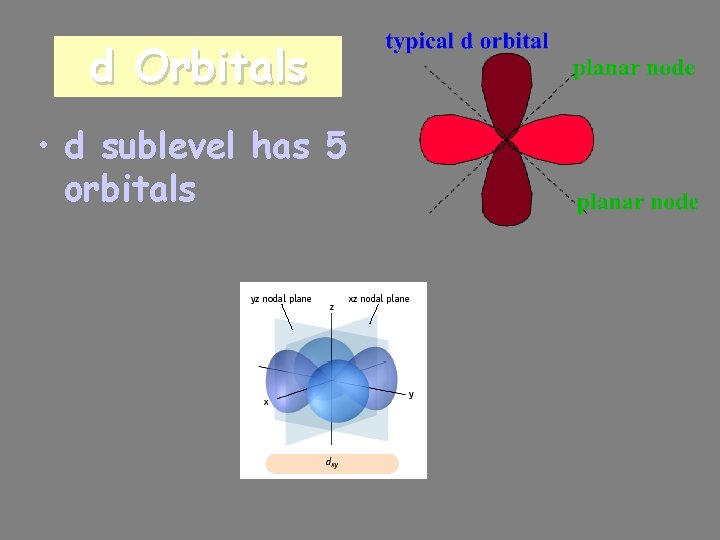
d Orbitals • d sublevel has 5 orbitals
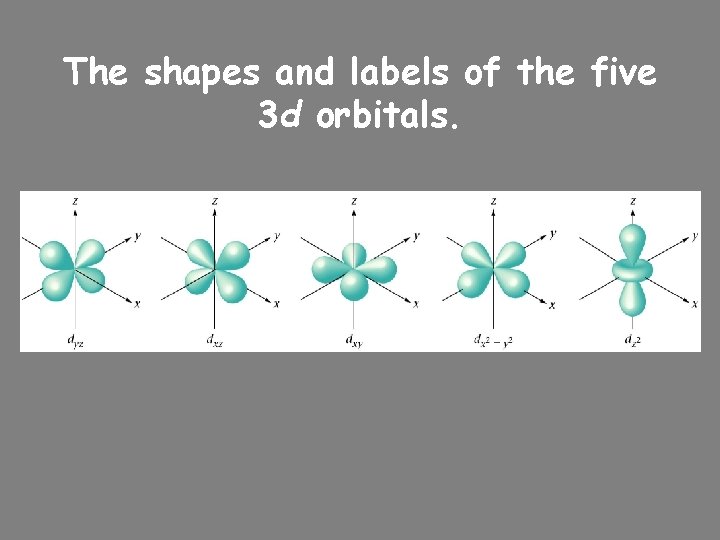
The shapes and labels of the five 3 d orbitals.

f Orbitals For l = 3, ---> f sublevel with 7 orbitals

Diagonal Rule • The diagonal rule is a memory device that helps you remember the order of the filling of the orbitals from lowest energy to highest energy • Aufbau principle states that electrons fill from the lowest possible energy to the highest energy

Diagonal Rule Steps: 1 s 2 s 3 s 1. Write the energy levels top to bottom. 2. Write the orbitals in s, p, d, f order. Write the same number of orbitals as the energy level. 3. Draw diagonal lines from the top right to the bottom left. 4. To get the correct order, 2 p 3 p 3 d follow the arrows! 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 5 g? 6 s 6 p 6 d 6 f 6 g? 6 h? 7 s 7 p 7 d 7 f 7 g? 7 h? By this point, we are past the current periodic table so we can stop. 7 i?

Why are d and f orbitals always in lower energy levels? • d and f orbitals require LARGE amounts of energy • It’s better (lower in energy) to skip a sublevel that requires a large amount of energy (d and f orbitals) for one in a higher level but lower energy This is the reason for the diagonal rule! BE SURE TO FOLLOW THE ARROWS IN ORDER!

How many electrons can be in a sublevel? Remember: A maximum of two electrons can be placed in an orbital. s orbitals p orbitals d orbitals f orbitals Number of electrons 1 3 5 7 2 6 10 14

Electron Configurations A list of all the electrons in an atom (or ion) • Must go in order (Aufbau principle) • 2 electrons per orbital, maximum • We need electron configurations so that we can determine the number of electrons in the outermost energy level. These are called valence electrons. • The number of valence electrons determines how many and what this atom (or ion) can bond to in order to make a molecule 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 14… etc.

Electron Configurations 4 2 p Energy Level Number of electrons in the sublevel Sublevel 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 14… etc.

Let’s Try It! • Write the electron configuration for the following elements: H Li N Ne K Zn
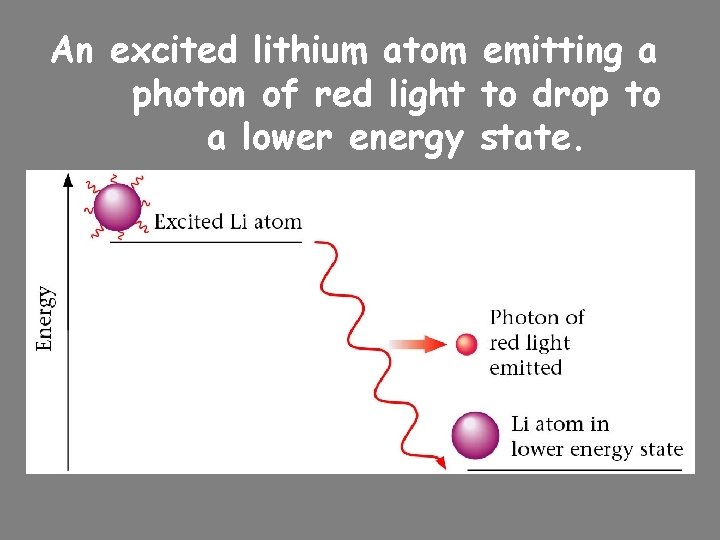
An excited lithium atom emitting a photon of red light to drop to a lower energy state.
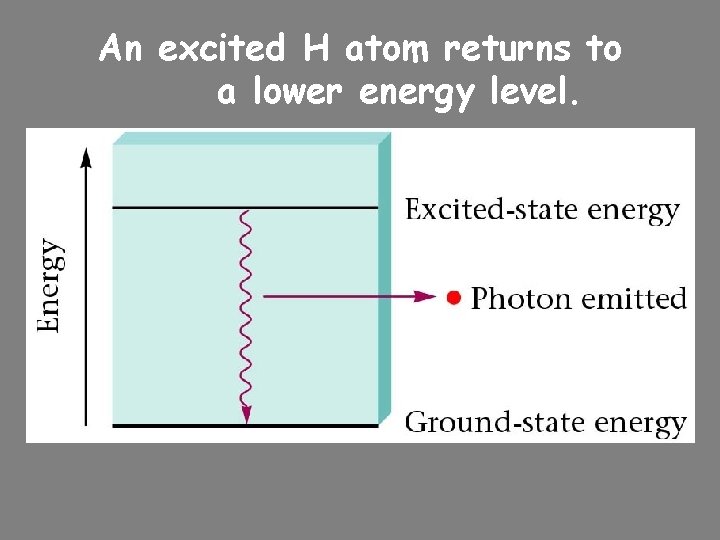
An excited H atom returns to a lower energy level.
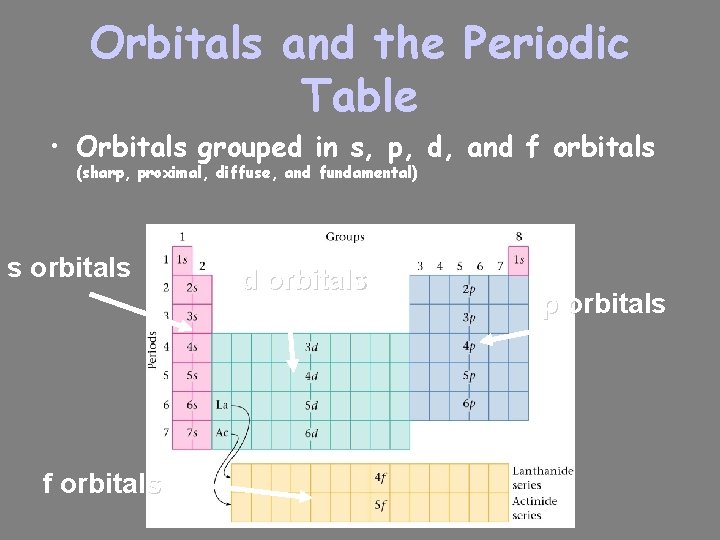
Orbitals and the Periodic Table • Orbitals grouped in s, p, d, and f orbitals (sharp, proximal, diffuse, and fundamental) s orbitals f orbitals d orbitals p orbitals

Shorthand Notation • A way of abbreviating long electron configurations • Since we are only concerned about the outermost electrons, we can skip to places we know are completely full (noble gases), and then finish the configuration

Shorthand Notation • Step 1: It’s the Showcase Showdown! Find the closest noble gas to the atom (or ion), WITHOUT GOING OVER the number of electrons in the atom (or ion). Write the noble gas in brackets [ ]. • Step 2: Find where to resume by finding the next energy level. • Step 3: Resume the configuration until it’s finished.
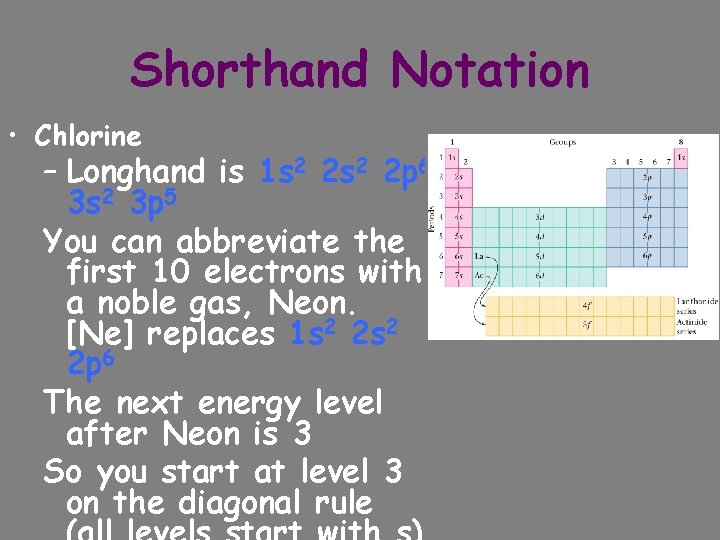
Shorthand Notation • Chlorine – Longhand is 1 s 2 2 p 6 3 s 2 3 p 5 You can abbreviate the first 10 electrons with a noble gas, Neon. [Ne] replaces 1 s 2 2 p 6 The next energy level after Neon is 3 So you start at level 3 on the diagonal rule

Practice Shorthand Notation • Write the shorthand notation for each of the following atoms: Cl K Ca I Bi

Valence Electrons are divided between core and valence electrons B 1 s 2 2 p 1 Core = [He] , valence = 2 s 2 2 p 1 Br [Ar] 3 d 10 4 s 2 4 p 5 Core = [Ar] 3 d 10 , valence = 4 s 2 4 p 5

Rules of the Game No. of valence electrons of a main group atom = Group number (for A groups) Atoms like to either empty or fill their outermost level. Since the outer level contains two s electrons and six p electrons (d & f are always in lower levels), the optimum number of electrons is eight. This is called the octet rule.

Keep an Eye On Those Ions! • Electrons are lost or gained like they always are with ions… negative ions have gained electrons, positive ions have lost electrons • The electrons that are lost or gained should be added/removed from the highest energy level (not the highest orbital in energy!)
![Keep an Eye On Those Ions! • Tin Atom: [Kr] 5 s 2 4 Keep an Eye On Those Ions! • Tin Atom: [Kr] 5 s 2 4](http://slidetodoc.com/presentation_image_h/8ef36566ff5207077758f508a37945ba/image-52.jpg)
Keep an Eye On Those Ions! • Tin Atom: [Kr] 5 s 2 4 d 10 5 p 2 Sn+4 ion: [Kr] 4 d 10 Sn+2 ion: [Kr] 5 s 2 4 d 10 Note that the electrons came out of the highest energy level, not the highest energy orbital!
![Keep an Eye On Those Ions! • Bromine Atom: [Ar] 4 s 2 3 Keep an Eye On Those Ions! • Bromine Atom: [Ar] 4 s 2 3](http://slidetodoc.com/presentation_image_h/8ef36566ff5207077758f508a37945ba/image-53.jpg)
Keep an Eye On Those Ions! • Bromine Atom: [Ar] 4 s 2 3 d 10 4 p 5 Br- ion: [Ar] 4 s 2 3 d 10 4 p 6 Note that the electrons went into the highest energy level, not the highest energy orbital!

Try Some Ions! • Write the longhand notation for these: FLi+ Mg+2 • Write the shorthand notation for these: Br. Ba+2 Al+3

Orbital Diagrams • Graphical representation of an electron configuration • One arrow represents one electron • Shows spin and which orbital within a sublevel • Same rules as before (Aufbau principle, d 4 and d 9 exceptions, two electrons in each orbital, etc. )

Orbital Diagrams • One additional rule: Hund’s Rule – In orbitals of EQUAL ENERGY (p, d, and f), place one electron in each orbital before making any pairs – All single electrons must spin the same way

Lithium Group 1 A Atomic number = 3 1 s 22 s 1 ---> 3 total electrons
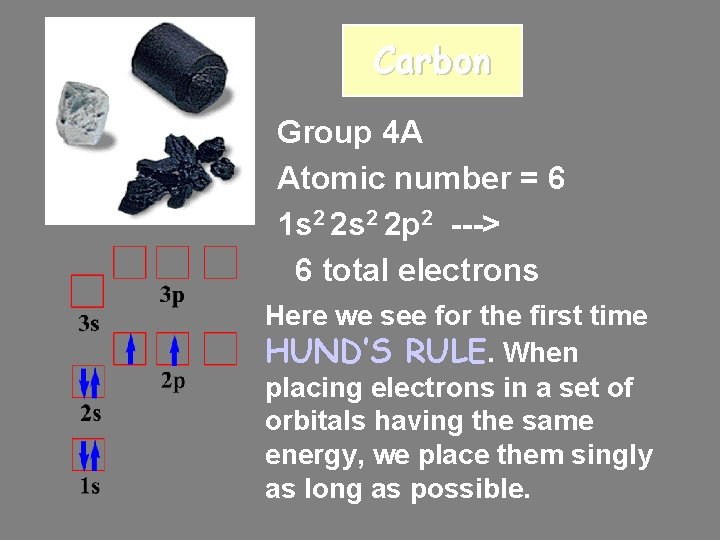
Carbon Group 4 A Atomic number = 6 1 s 2 2 p 2 ---> 6 total electrons Here we see for the first time HUND’S RULE. When placing electrons in a set of orbitals having the same energy, we place them singly as long as possible.
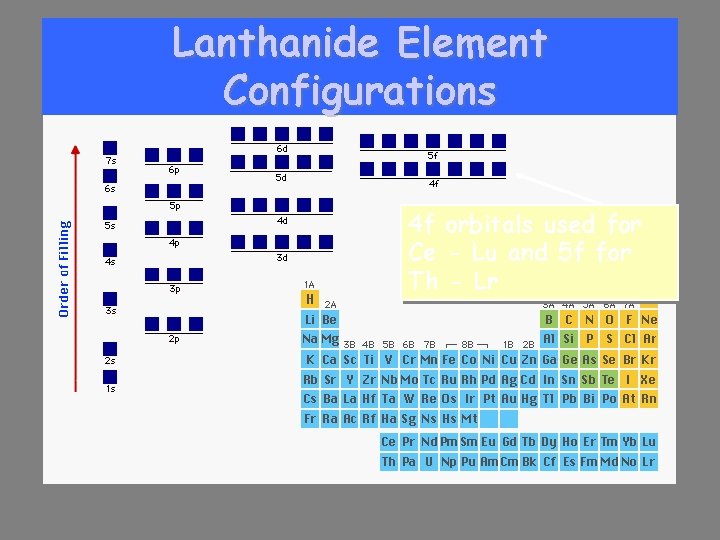
Lanthanide Element Configurations 4 f orbitals used for Ce - Lu and 5 f for Th - Lr
Draw these orbital diagrams! • Oxygen (O) • Chromium (Cr) • Mercury (Hg)
- Slides: 60