ATOMS THE BASICS Atoms Building blocks of all

ATOMS– THE BASICS Atoms: • Building blocks of all things • Smallest particle that can still be considered an element Structure: Particle Charge Mass Where found Proton +1 1 amu Nucleus Neutron Neutral, no charge 1 amu Nucleus Electron -1 0. 00056 amu Electron cloud

ATOMS– THE BASICS Atomic Number and Number of protons, number of electrons Element symbol Average atomic mass of element ***Atomic mass = Protons + Neutrons The number of protons will always equal the number of electrons giving an overall neutral charge for the atom unless indicated.
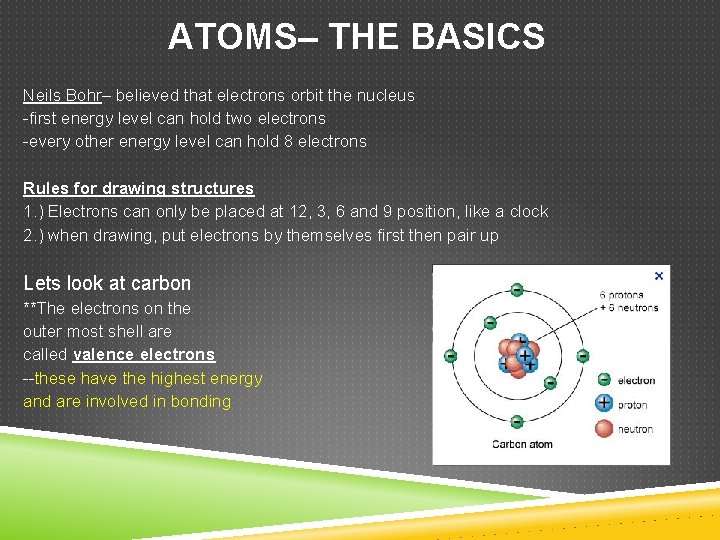
ATOMS– THE BASICS Neils Bohr– believed that electrons orbit the nucleus -first energy level can hold two electrons -every other energy level can hold 8 electrons Rules for drawing structures 1. ) Electrons can only be placed at 12, 3, 6 and 9 position, like a clock 2. ) when drawing, put electrons by themselves first then pair up Lets look at carbon **The electrons on the outer most shell are called valence electrons --these have the highest energy and are involved in bonding

ATOMS– THE BASICS Gilbert Newton Lewis– Lewis dot structures for valence electrons **Lewis dot structures are also known as electron dot diagrams C Electron Dot Diagrams The valence electrons of an atom are shown as dots around the symbol of the element.
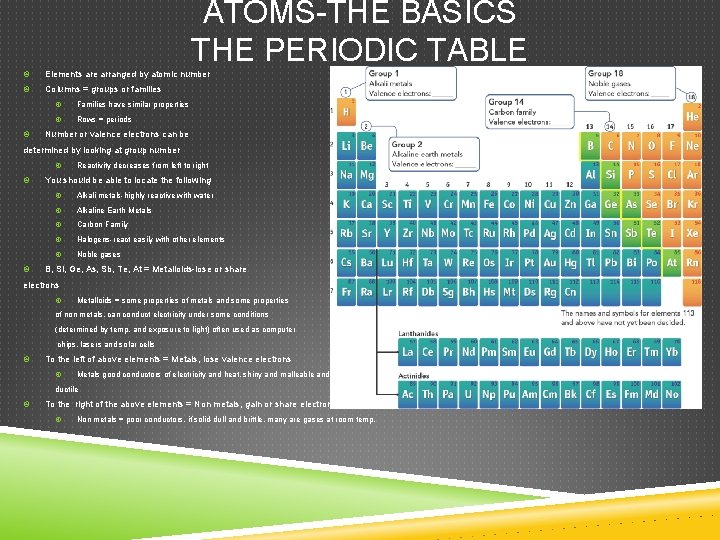
ATOMS-THE BASICS THE PERIODIC TABLE Elements are arranged by atomic number Columns = groups or families Families have similar properties Rows = periods Number or valence electrons can be determined by looking at group number Reactivity decreases from left to right You should be able to locate the following Alkali metals-highly reactive with water Alkaline Earth Metals Carbon Family Halogens-react easily with other elements Noble gases B, Si, Ge, As, Sb, Te, At = Metalloids-lose or share electrons Metalloids = some properties of metals and some properties of non metals, can conduct electricity under some conditions (determined by temp. and exposure to light) often used as computer chips, lasers and solar cells To the left of above elements = Metals, lose valence electrons Metals good conductors of electricity and heat, shiny and malleable and ductile To the right of the above elements = Non metals, gain or share electrons Non metals = poor conductors, if solid-dull and brittle, many are gases at room temp.

ATOMS-THE BASICS IONIC BONDING Ion- atom or group of atoms with a charge Ionic Bonding occurs between non metals and metals which are oppositely charged to form ionic compounds which are neutral Electrons are transferred, given away by one and taken by the other Metals Non Metals Lose electrons Gain electrons Positive charge Negative charge Cation Anion Number of electrons that are transferred is determined by number of electrons in valence shell Remember—we want full, stable, happy shells! Every valence electron wants to have a partner
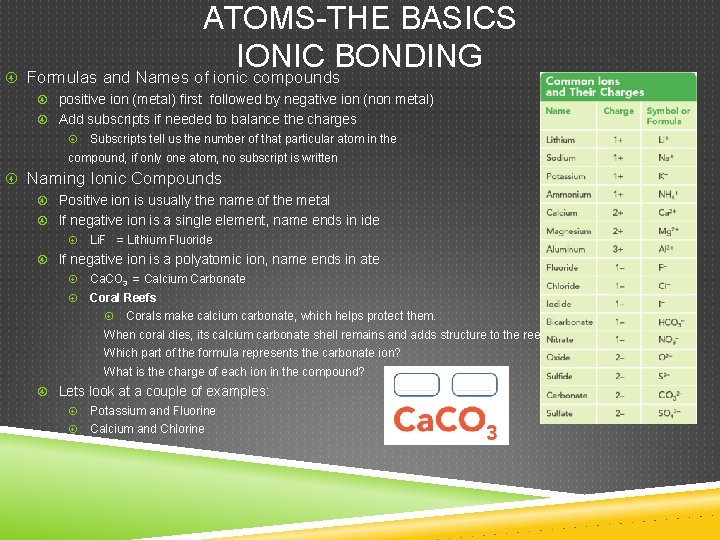
ATOMS-THE BASICS IONIC BONDING Formulas and Names of ionic compounds positive ion (metal) first followed by negative ion (non metal) Add subscripts if needed to balance the charges Subscripts tell us the number of that particular atom in the compound, if only one atom, no subscript is written Naming Ionic Compounds Positive ion is usually the name of the metal If negative ion is a single element, name ends in ide Li. F = Lithium Fluoride If negative ion is a polyatomic ion, name ends in ate Ca. CO 3 = Calcium Carbonate Coral Reefs Corals make calcium carbonate, which helps protect them. When coral dies, its calcium carbonate shell remains and adds structure to the reef. Which part of the formula represents the carbonate ion? What is the charge of each ion in the compound? Lets look at a couple of examples: Potassium and Fluorine Calcium and Chlorine

LETS REVIEW IONIC BONDING What is an ionic bond? Atoms will transfer one or more __________ to another to form the bond. Each atom is left with a __________ outer shell. An ionic bond forms between a __________ ion with a positive charge and a __________ ion with a negative charge. Example B 1: Sodium + Chlorine Example B 2: Magnesium + Iodine Example B 3: Potassium + Iodine Example B 4: Sodium + Oxygen Galena, or lead sulfide (Pb. S), has a structure similar to that of table salt. Halite Ions in ionic compounds are arranged in three-dimensional shapes called crystals. Some have a cubic shape.
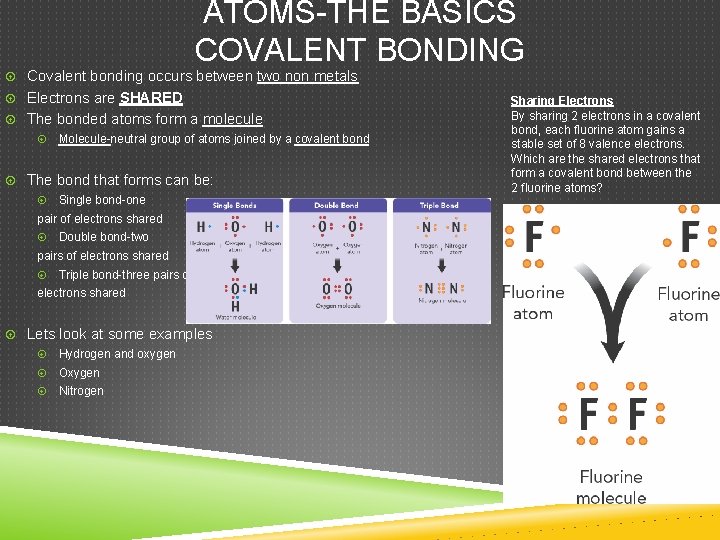
ATOMS-THE BASICS COVALENT BONDING Covalent bonding occurs between two non metals Electrons are SHARED The bonded atoms form a molecule Molecule-neutral group of atoms joined by a covalent bond The bond that forms can be: Single bond-one pair of electrons shared Double bond-two pairs of electrons shared Triple bond-three pairs of electrons shared Lets look at some examples Hydrogen and oxygen Oxygen Nitrogen Sharing Electrons By sharing 2 electrons in a covalent bond, each fluorine atom gains a stable set of 8 valence electrons. Which are the shared electrons that form a covalent bond between the 2 fluorine atoms?

ATOMS-THE BASICS COVALENT BONDING First atom, just its name, unless there are multiple of that atom Second atom, add prefix to indicate how many, then “ide” on end 1 - mono 2 - di 3 - tri 4 - tetra 5 - penta 6 - hexa 7 - hepta 8 - octa 9 - nona 10 - deca Name the elements in the same left-to-right order that they have on the periodic table, except that you would have to squeeze hydrogen in between nitrogen and oxygen. Example: CO = Carbon monoxide N 2 S 3 = Dinitrogen Tri. Sulfide

POLAR VS. NON POLAR COVALENT BONDS Although electrons are shared, they are not always shared equally Sharing is determined by electronegativity measure of the attraction of an atom for the electrons in a bond Higher electronegativity-greater the attraction for electrons Electronegativity increases as you go from left to right and bottom to top Non polar bond- equal sharing Polar bond- unequal sharing, causes atoms to have slight charge First check symmetry, then check electronegativity Symmetrical = could be non polar, then check electronegativity Non symmetrical = probably polar Nonpolar and Polar Bonds Hydrogen forms a nonpolar bond with another hydrogen atom. In hydrogen fluoride, fluorine attracts electrons more strongly than hydrogen does. The bond formed is polar.
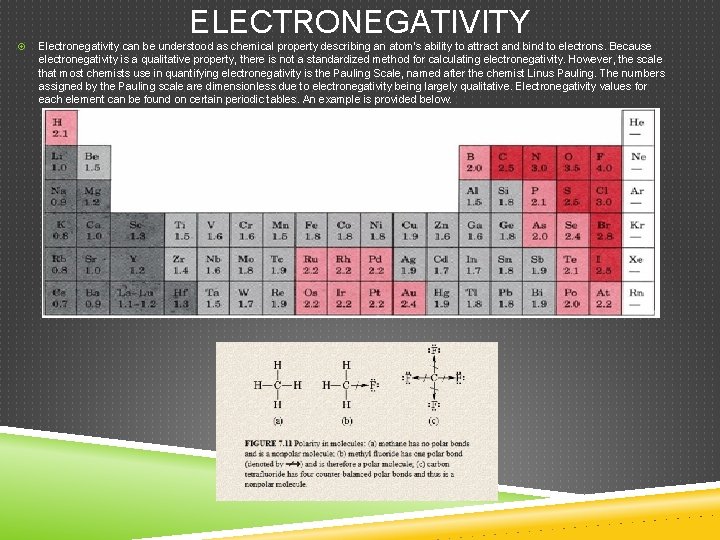
ELECTRONEGATIVITY Electronegativity can be understood as chemical property describing an atom's ability to attract and bind to electrons. Because electronegativity is a qualitative property, there is not a standardized method for calculating electronegativity. However, the scale that most chemists use in quantifying electronegativity is the Pauling Scale, named after the chemist Linus Pauling. The numbers assigned by the Pauling scale are dimensionless due to electronegativity being largely qualitative. Electronegativity values for each element can be found on certain periodic tables. An example is provided below.

FORCES BETWEEN POLAR MOLECULES Polar molecules are attracted to one and other due to their slight charges The forces that hold these molecules together are called van der Waals forces also pull molecules to each other Read page 118 van der Waals forces cause a geckos feet to grip smooth surfaces such as glass

LETS REVIEW COVALENT BONDING What is a covalent bond? Atoms __________ one or more electrons with each other to form the bond. Each atom is left with a __________ outer shell. A covalent bond forms between two __________. Example C 1: Hydrogen + Chlorine Example C 2: 2 Hydrogen + Oxygen Example C 3: Chlorine + Chlorine Example C 4: Oxygen + Oxygen
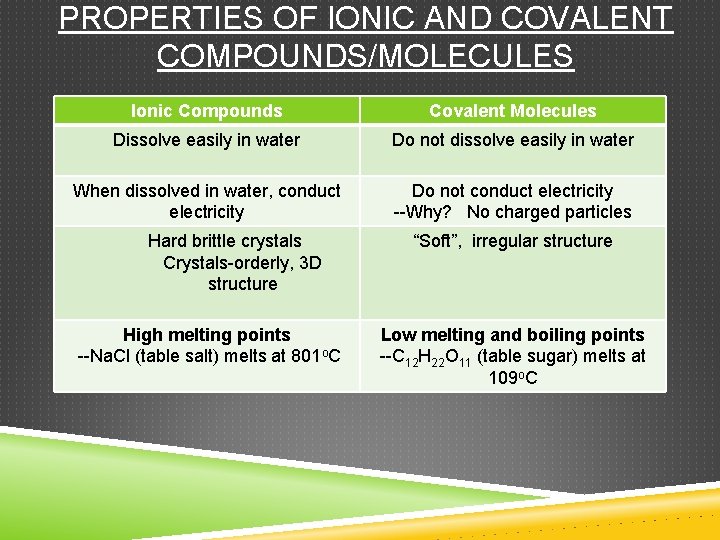
PROPERTIES OF IONIC AND COVALENT COMPOUNDS/MOLECULES Ionic Compounds Covalent Molecules Dissolve easily in water Do not dissolve easily in water When dissolved in water, conduct electricity Do not conduct electricity --Why? No charged particles Hard brittle crystals Crystals-orderly, 3 D structure High melting points --Na. Cl (table salt) melts at 801 o. C “Soft”, irregular structure Low melting and boiling points --C 12 H 22 O 11 (table sugar) melts at 109 o. C
- Slides: 15