Atoms Nucleus and Radioactivity 20 October 2015 Background

Atom’s Nucleus and Radioactivity 20 October 2015 Background n Radioactivity and natural background exposure n Principles of radioactivity and human health n Application of the principles of radioactivity n

Isotopes n Atoms have specific number of protons, neutrons and electrons 8 16 n O Oxygen-16 (breath deep …) If the number of protons is unchanged but the number of neutrons goes up or down, what happens to the chemical properties of that element (i. e. , Periodic Table)? 8 17 O Oxygen-17 (breath deep …)

Isotopes … A Bit More Atoms of an element with same number of protons but different number of neutrons = isotopes n Conventional notation (Element - Mass) n Oxygen -17 or Oxygen -16 n Carbon -12 or Carbon -13 n Iodine -131 n Cesium -135 n Plutonium -237 n Uranium -235 n

Example: Isotopes of Hydrogen Hydroge n 1 Deuterium 1 1 H 2 H Stable Proton Tritium 1 3 H Unstable Neutron What is a stable versus an unstable isotope?

Stable Versus Unstable Isotopes n Stable over time Oxygen-16 n Carbon -12 n Carbon -13 n n Unstable: “atom changes” by releasing mass and/or energy (i. e. , radioactive) Carbon -14 (radioactive carbon) n Hydrogen-3 (radioactive hydrogen/tritium) n

Unstable Isotopes: Radioactive Emissions n n n Alpha (a): 2 protons + 2 neutrons (same as the helium atom) Beta (b): high energy electron Gamma (g): electromagnetic radiation with very short wavelengths (not visible to eye)

Natural or Background Radioactivity n Sources n n Cosmic rays from outer space Soils Water Building materials (rock) n n Nuclear sources Examples n n Annual Mean Exposure (OSU) Radon gas (Radon-222) - soils Beryllium (Beryllium-7) - atmosphere

Example: Radon Exposure in the Home n n Radon gas (Radon-222) Radioactive gas Human health effects - lungs Human health exposure n n Homes At-risk groups: smokers Risk mitigation - venting of basements Geologically based radioactivity: 100% natural

The Essence of Being Unstable: Radioactive Decay Uranium-238 92 protons + 146 neutrons (contrast with other atoms) n Spontaneous release of an alpha (a) particle (2 protons + 2 neutrons): atom now with only 90 protons and mass of 234, which is ____? n All isotopes of all elements with > 83 protons (Bismuth) are unstable and radioactively decay n
Periodic Table

n Types of Decay and Human Health Alpha (a) Two protons + two neutrons n Travel distance: stopped by sheet of paper (even air) n n Beta (b) high energy electron n Travel distance: ~10 meters; stopped by 1 cm aluminum block n n Gamma (g) high energy radiation n Travel distance: 100’s meters; stopped by 5 cm n
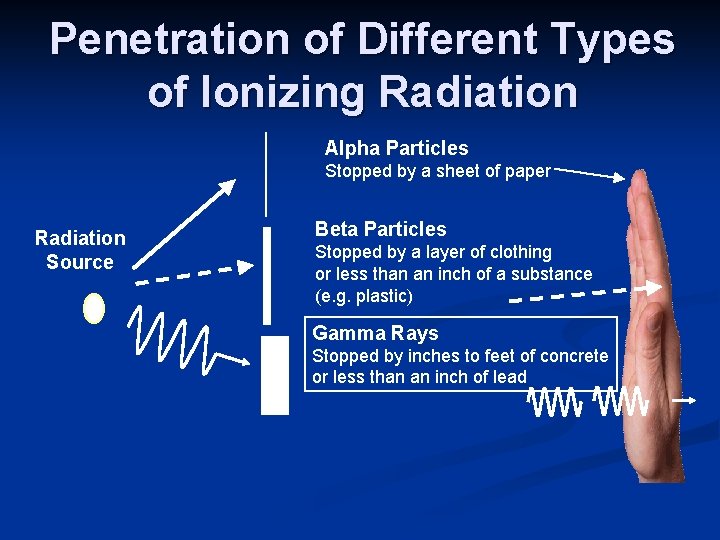
Penetration of Different Types of Ionizing Radiation Alpha Particles Stopped by a sheet of paper Radiation Source Beta Particles Stopped by a layer of clothing or less than an inch of a substance (e. g. plastic) Gamma Rays Stopped by inches to feet of concrete or less than an inch of lead

Radioactivity: Human Health n Radioactivity in biological tissues results in atoms being ionized n n Disrupts chemical bonds DNA as primary site of action (e. g. , mutations) Disrupts biochemistry Sensitivity greatest for actively growing cells n n Blood (Strontium-90) Bone marrow (Cesium-137) Thyroid (Iodine-131) Muscle (Pu-239)
Periodic Table

Radioactivity: Principles and Applications Background n Radioactivity is natural n Quantitative analysis of radioactivity n Principles of radioactivity and human health n Application of the principles of radioactivity n

Radioactive Decay Rate of decay to a stable state (no more spontaneous decay): specific for each isotope n Rate: unique terminology = “half-life” n Time for ½ (50%) of atomic nuclei to decay to the stable state (abbreviated t 1/2) n Range: fractions of second to billions of years (isotope specific) n Unaffected by the environment (e. g. , moisture, light, pressure, etc. ) n
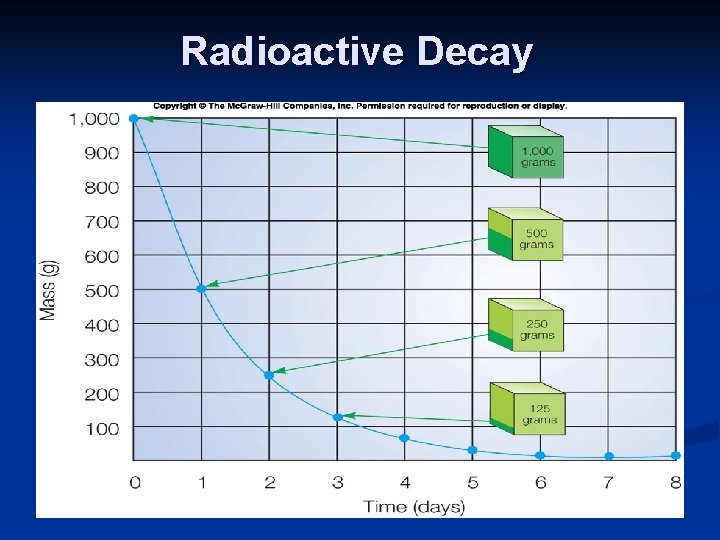
Radioactive Decay

Question The half-life of a radioactive element is ___. A. the time it takes for 50% of the atoms in a sample to spontaneously decay B. indicates that it is capable of reducing the life of an exposed human by 50% C. describes 50% of the energy that is released when the atomic nucleus decays D. is a measure of 50% the radioactivity that is emitted from each atomic nucleus that decays

Question As the temperature of a sample of radioactive element increases, the half-life will ___. A. B. C. D. Decrease Remain unchanged Increase None of the above

Question There are two radioactive sources, A and B; both have the same number of unstable nuclei at the beginning. After 10 days, the number of unstable nuclei remaining for B is more than A. Which of the following statements is correct? A. B. C. D. The mass of A is larger than that of B The mass of B is larger than that of A The half-life (t 1/2) of B is longer than that of A The half-life (t 1/2) of A is longer than that of B

Application of Atomic Principles of Radioactivity Medical science n Nuclear energy n Chernobyl Russia n Fukishima Japan n Dirty bomb n

Medical Science: Nuclear Applications n Nuclear imaging and radiopharmaceuticals Injection of radioactive element n Allow for distribution in body n Imagine body for presence or absence of radioactive element n Function: assessment of functional and dysfunctional tissues (e. g. , thyroid, kidneys, heart, liver) n n Examples

Nuclear Reactor: Power Generation (read section in text) Fuel rods Moderators Coolants Steam turbines Cooling towers Nuclear waste storage Comparison with fossil fuel power

Chernobyl Reactor and Sarcophagus - April 1986 n n n Complete core meltdown Sarcophagus - risk containment Human health (~4, 000 death) n Acute radiation poisoning – on site workers n Chronic exposure - residents n n n Iodine -131 Thyroid cancer and leukemia No detectable effects > 100 miles

Fukushima Daiichi Nuclear Power Plant Accident - March 2011

Question Fissionable (means what? ) uranium-233, uranium-235 and plutonium-239 are used in nuclear reactors as ___. A. B. C. D. E. Coolants Control rods Moderators Aspirators Fuels

Question Nuclear reactors and traditional fossil fuel combustion (gas and/or coal) plants share the following fundamental energy conversion feature. A. B. C. D. E. Nuclear reactor Control rods Power lines Aspirators Stem-driven generators to convert

Atom’s Nucleus and Radioactivity 20 October 2015 Background n Radioactivity and natural background exposure n Principles of radioactivity and human health n Application of the principles of radioactivity n
- Slides: 28