Atoms Molecules for Environmental Science Atoms building blocks

Atoms & Molecules for Environmental Science

Atoms • building blocks of matter • Smallest particle that retains its properties • dense nucleus in center – mostly space

Subatomic Particles particle size charge location proton large positive nucleus neutron large neutral nucleus electron small(1/2000 th) negative cloud
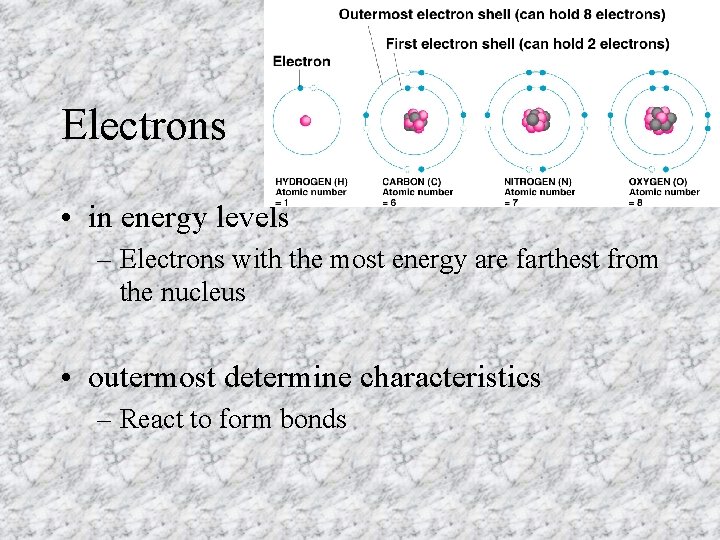
Electrons • in energy levels – Electrons with the most energy are farthest from the nucleus • outermost determine characteristics – React to form bonds

Symbols to Memorize

Atomic Number • Atomic # = # of Protons (whole number) • = # of electrons in a neutral atom – gives identity

Atomic Mass • Atomic mass number = weighted average of the masses of the isotopes of the element (decimal)

• mass number= # of protons + # of neutrons – 12 C has 6 protons + 6 neutrons – 13 C has 6 protons + 7 neutrons

Isotopes • atoms of the same element with a different mass – 12 C – 13 C – 14 C
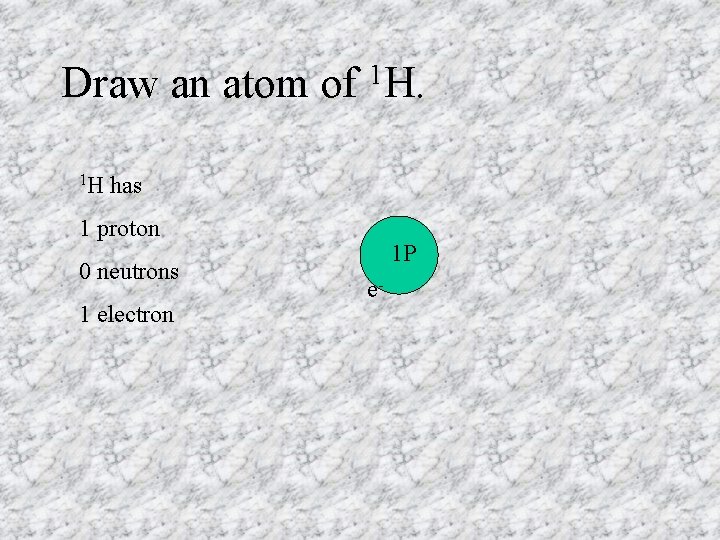
Draw an atom of 1 H 1 H. has 1 proton 0 neutrons 1 electron 1 P e-
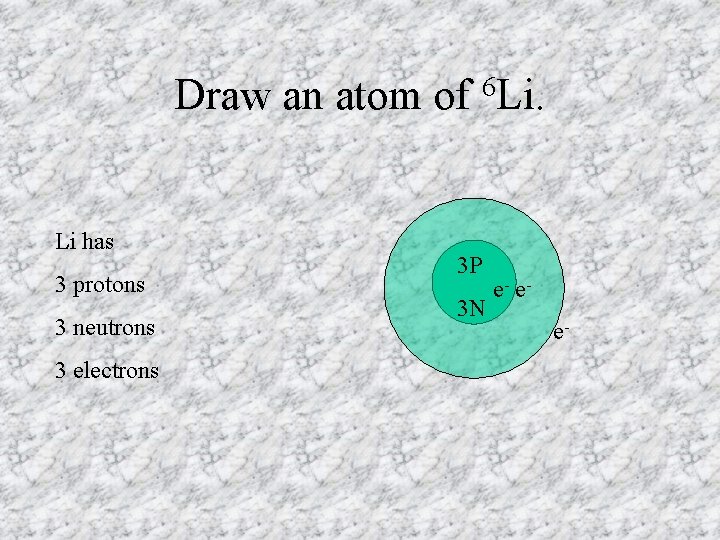
Draw an atom of Li has 3 protons 3 neutrons 3 electrons 6 Li. 3 P 3 N e- ee-
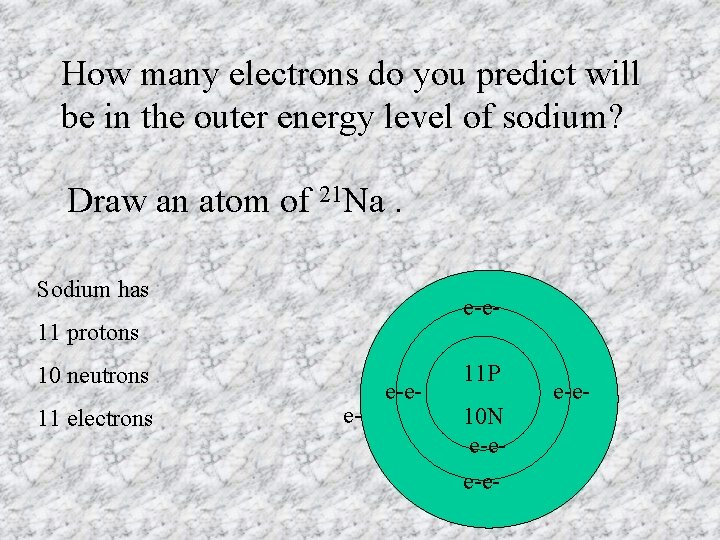
How many electrons do you predict will be in the outer energy level of sodium? Draw an atom of 21 Na. Sodium has e-e- 11 protons 10 neutrons 11 electrons e- e-e- 11 P 10 N e-ee-e-
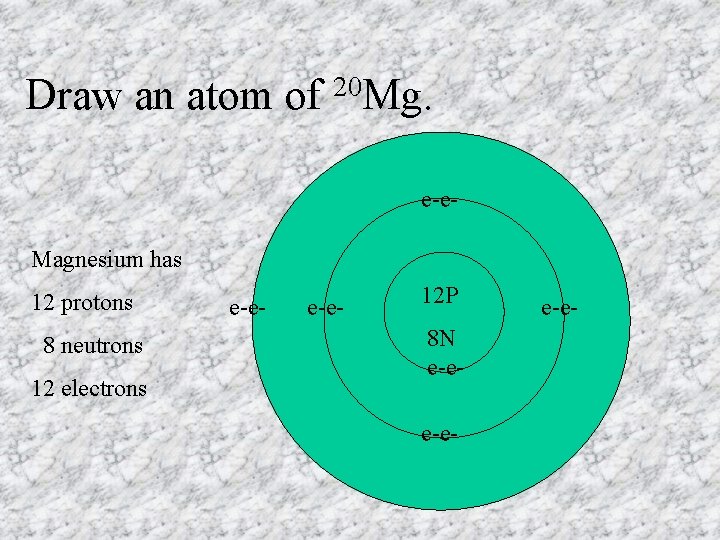
Draw an atom of 20 Mg. e-e- Magnesium has 12 protons 8 neutrons 12 electrons e-e- 12 P 8 N e-ee-e-
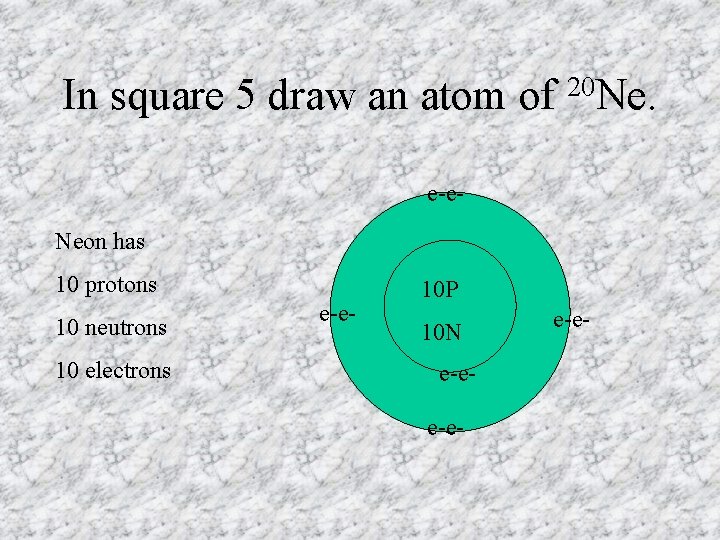
In square 5 draw an atom of 20 Ne. e-e. Neon has 10 protons 10 neutrons 10 electrons e-e- 10 P 10 N e-ee-e-

Draw the isotopes 14 C and 12 C. Carbon 12 has 6 protons e- 6 neutrons 6 electrons 6 P 6 N e-ee-e- e- Carbon 14 has 6 protons 8 neutrons 6 electrons 6 P e- 8 N e-ee-e- e-

Stable Atoms • have eight electrons in s & p orbitals in the outermost energy level • do not go through chemical reactions • Noble gases
- Slides: 16