Atoms Molecules and Ions Ch 2 cont HW


















- Slides: 27

Atoms, Molecules, and Ions Ch. 2 (cont. )

HW 2 2. Aluminum is a lightweight metal (ρ=2. 70 g/cm 3) used in aircraft construction, high-voltage transmission lines, and foils. What is its density in kg/m 3? 2. 70 x 103 kg/m 3 2/23/2021 2

HW 2 3. A resting adult requires about 240 m. L of pure oxygen/min and breathes about 12 times every minute. If inhaled air contains 20% oxygen by volume and exhaled air 16%, what is the volume of air per breath? (Assume that the volume of inhaled air is equal to that of exhaled air. ) 1. Need volume of O 2 required per breath 2. Need % O 2 used 3. Use above to figure out volume of air per breath 2/23/2021 3

HW 2 3. A resting adult requires about 240 m. L of pure oxygen/min and breathes about 12 times every minute. If inhaled air contains 20% oxygen by volume and exhaled air 16%, what is the volume of air per breath? (Assume that the volume of inhaled air is equal to that of exhaled air. ) 5. 00 x 102 m. L/breath 2/23/2021 4
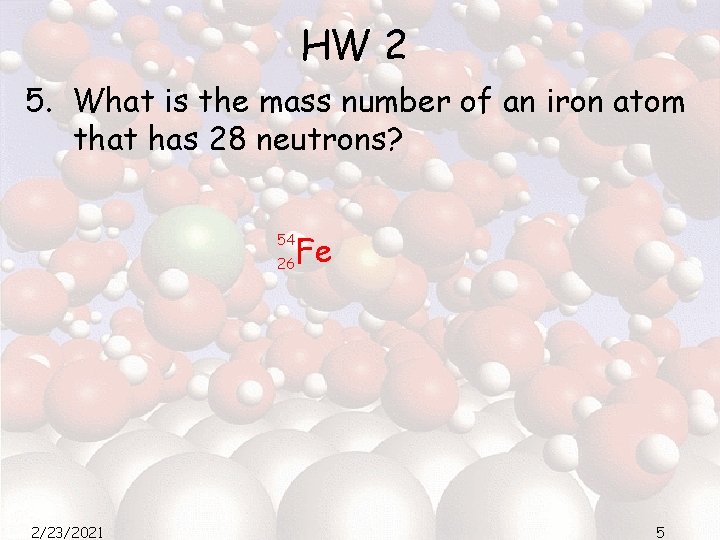
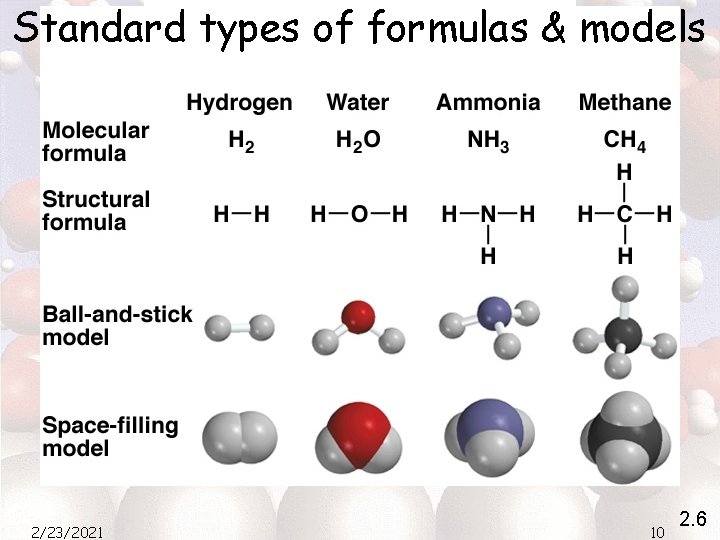
HW 2 5. What is the mass number of an iron atom that has 28 neutrons? 54 26 2/23/2021 Fe 5

Q 1 1. An object, of density 5. 40 g/cm 3 and mass 80. 0 g, is dropped into a 100 -m. L graduated cylinder containing exactly 50. 0 m. L of water. To what height will the water level rise in the cylinder? 64. 8 m. L 2. What subatomic particle(s) make up most of the mass of an atom? protons and neutrons 2/23/2021 6

• What element listed in Group IA is not an alkali metal? hydrogen 2/23/2021 7

• What noble gas has a mass number of 4? helium 2/23/2021 8

Heisenberg 2/23/2021 9

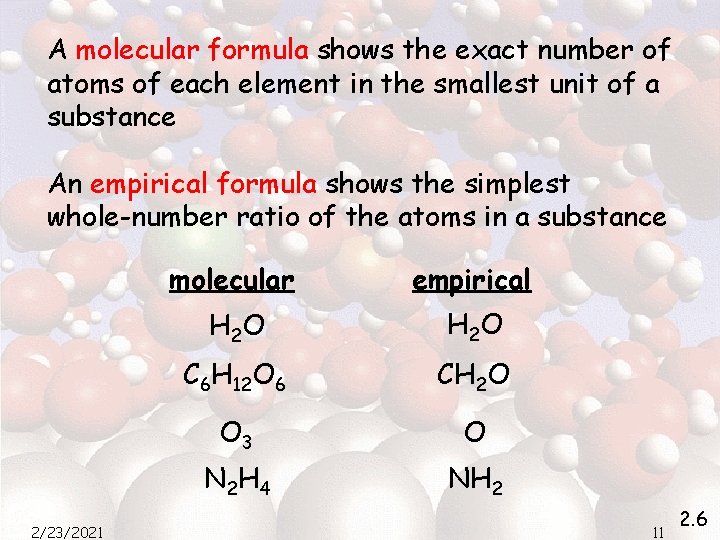
Standard types of formulas & models 2/23/2021 10 2. 6

A molecular formula shows the exact number of atoms of each element in the smallest unit of a substance An empirical formula shows the simplest whole-number ratio of the atoms in a substance 2/23/2021 molecular empirical H 2 O C 6 H 12 O 6 CH 2 O O 3 O N 2 H 4 NH 2 11 2. 6

ionic compounds consist of a cation and an anion • the formula is always the same as the empirical formula • the sum of the charges on the cation and anion in each formula unit must equal zero The ionic compound Na. Cl 2/23/2021 12 2. 6

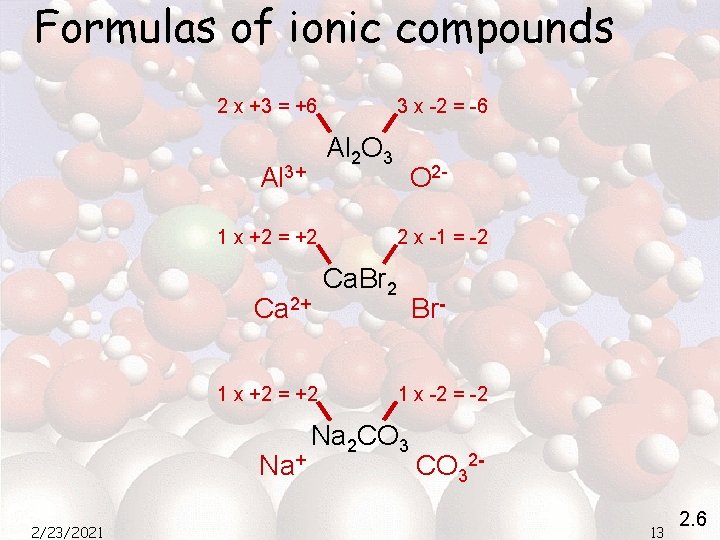
Formulas of ionic compounds 2 x +3 = +6 3 x -2 = -6 Al 2 O 3 Al 3+ 1 x +2 = +2 Ca 2+ 1 x +2 = +2 Na+ 2/23/2021 O 22 x -1 = -2 Ca. Br 2 Br 1 x -2 = -2 Na 2 CO 3213 2. 6

Some Polyatomic Ions (Table 2. 3) Memorize the polyatomic ions (names and formulae) in Table 2. 3 (p. 44, Chang)! 2/23/2021 14 2. 7

• Write the formula for the compound that results when NH 4 and SO 4 ions associate. Is this a molecular formula or an empirical formula? NH 4 has +1 charge SO 4 has -2 charge (NH 4)2(SO 4) 1 (NH 4)2 SO 4 ammonium sulfate (from Table 2. 3) ionic compound -> empirical formula 2/23/2021 15

Nomenclature • Figure 2. 13 (p. 47, Chang) • Ionic – Cation has only one charge – Cation has more than one charge • Molecular 2/23/2021 16

Nomenclature of ionic compounds The first element in the formula is identified by its English name, the second by appending the suffix –ide to its stem Chemical Symbol O S N P F Cl Br I Name as Stem First Element oxoxygen sulfur nitrogen phosph- phosphorus fluorine chlorine bromine iodiodine Name as Second Element oxide sulfide nitride phosphide fluoride chloride bromide iodide *Note that element can be atomic or polyatomic; first element usually refers to the cation and second element usually refers to the anion 2/23/2021 17

Nomenclature of ionic compounds • Ionic Compounds – often a metal + nonmetal – anion (nonmetal), add “ide” to stem 2/23/2021 Ba. Cl 2 barium chloride K 2 O potassium oxide Mg(OH)2 magnesium hydroxide KNO 3 potassium nitrate 18 2. 7

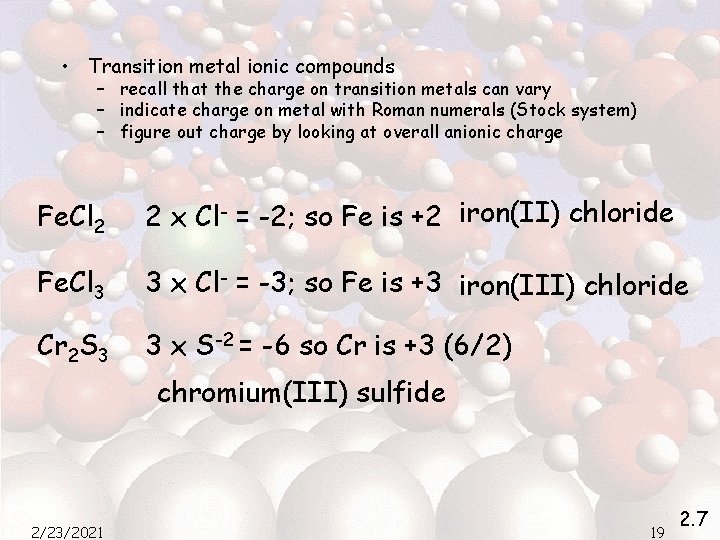
• Transition metal ionic compounds – recall that the charge on transition metals can vary – indicate charge on metal with Roman numerals (Stock system) – figure out charge by looking at overall anionic charge Fe. Cl 2 2 x Cl- = -2; so Fe is +2 iron(II) chloride Fe. Cl 3 3 x Cl- = -3; so Fe is +3 iron(III) chloride Cr 2 S 3 3 x S-2 = -6 so Cr is +3 (6/2) chromium(III) sulfide 2/23/2021 19 2. 7

Nomenclature of molecular compounds – as a general rule, molecular compounds are formed when nonmetallic elements combine (sometimes a nonmetal and a metalloid) – nonmetals or nonmetals + metalloids – element further left in periodic table is 1 st – element closest to bottom of group is 1 st – if more than one compound can be formed from the same elements, use Greek prefixes to indicate number of each kind of atom – last element ends in ide 2/23/2021 20 2. 7

Nomenclature of molecular compounds – Many molecular compounds contain hydrogen: Period 2 3 4 5 IVA CH 4 Si. H 4 Ge. H 4 Group VA VIA NH 3 H 2 O PH 3 H 2 S As. H 3 H 2 Se Sb. H 3 H 2 Te VIIA HF HCl HBr HI Noble Gas Ne Ar Kr Xe Note: The number of hydrogens that combined with the nonmetal equals the number of spaces to the right we have to move to get to the noble gas 2/23/2021 21

Nomenclature of molecular compounds – The number of each type of atom is specified with Greek prefixes Greek Prefixes mono- = 1 (often omitted) di=2 tri=3 tetra- = 4 penta- = 5 Examples: PF 5 = phosphorus pentafluoride HCl = hydrogen chloride N 2 O 5 = dinitrogen pentoxide 2/23/2021 hexa- = 6 hepta- = 7 octa= 8 nona- = 9 deca- = 10 Note: many compounds have common names, like water for H 2 O. 22

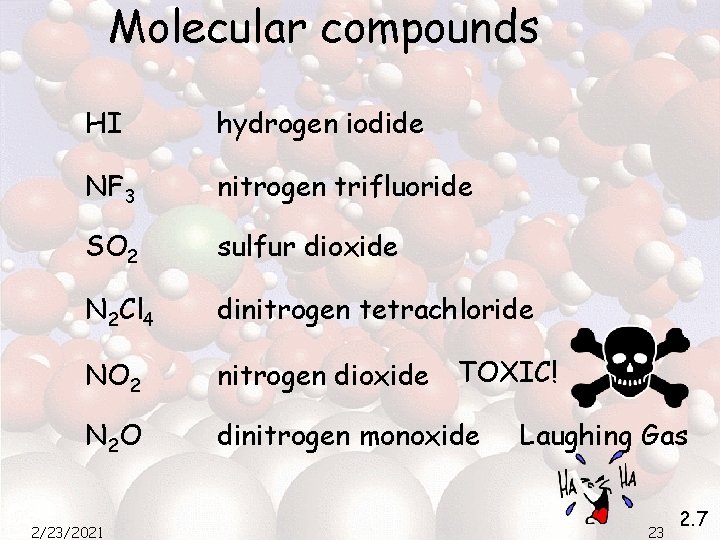
Molecular compounds HI hydrogen iodide NF 3 nitrogen trifluoride SO 2 sulfur dioxide N 2 Cl 4 dinitrogen tetrachloride NO 2 nitrogen dioxide TOXIC! N 2 O dinitrogen monoxide 2/23/2021 Laughing Gas 23 2. 7

An acid can be defined as a substance that yields hydrogen ions (H+) when dissolved in water. HCl • Pure substance, hydrogen chloride • Dissolved in water (H+ Cl-), hydrochloric acid An oxoacid is an acid that contains hydrogen, oxygen, and another element (see section 2. 7 & Tables 2. 5 and 2. 6… memorize these). HNO 3 H 2 CO 3 H 2 SO 4 HCl. O 3 2/23/2021 nitric acid carbonic acid sulfuric acid chloric acid 24 2. 7

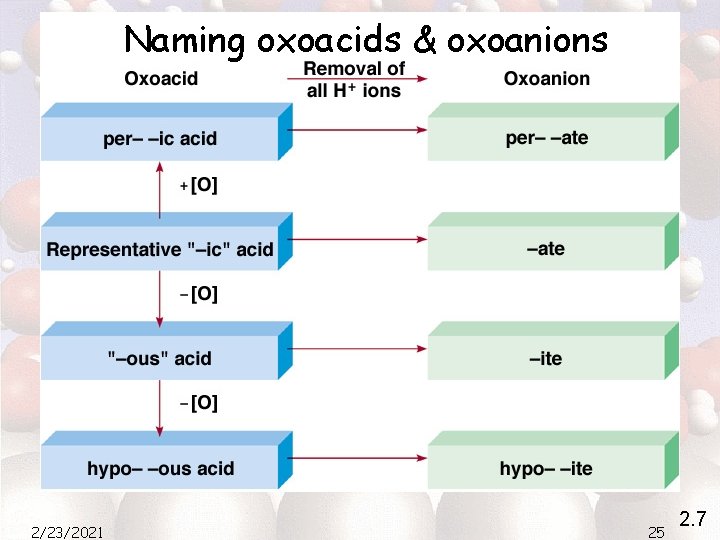
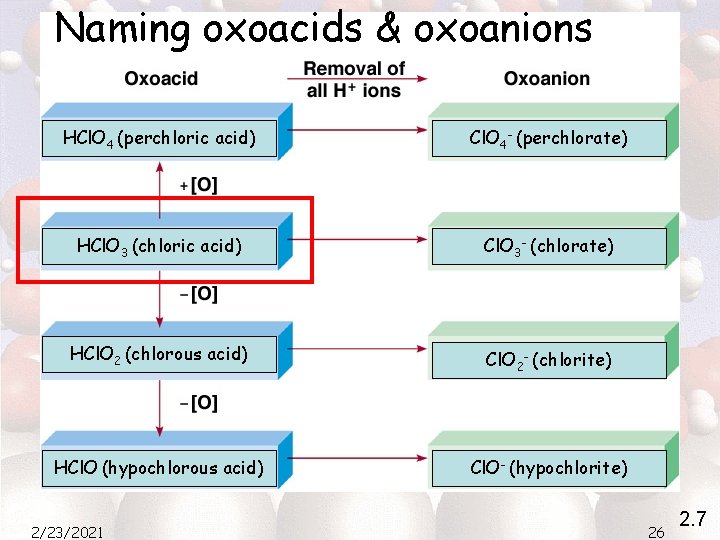
Naming oxoacids & oxoanions 2/23/2021 25 2. 7

Naming oxoacids & oxoanions HCl. O 4 (perchloric acid) Cl. O 4 - (perchlorate) HCl. O 3 (chloric acid) Cl. O 3 - (chlorate) HCl. O 2 (chlorous acid) Cl. O 2 - (chlorite) HCl. O (hypochlorous acid) Cl. O- (hypochlorite) 2/23/2021 26 2. 7

Practice with oxoacids • Name HNO 3, HNO 2, HNO • Write formulas for sulfuric acid and sulfurous acid (stem= sulf-) • Name the anions of HNO 3, HNO 2, HNO • Name the anions of sulfuric acid and sulfurous acid 2/23/2021 27