Atoms l Atoms basic building blocks for all

Atoms l Atoms – basic building blocks for all earth materials; consist of 3 basic components: protons, neutrons, electrons

Atoms combine to form Elements l Elements – fundamental component that can not be broken down into other substances by ordinary chemical processes l Elements combine to form compounds.

General Facts About Minerals 2, 000 + minerals have been identified Most minerals are compounds, especially the silicate group (Si, O). l A few are “native elements” -- made of only one element, such as sulfur, gold. copper, and graphite (carbon) l

MINERALS NATIVE ELEMENTS Gold (Au) Silver (Ag) Platinum (Pt) Diamond (C) Graphite (C) Sulfur (S) Copper (Cu) Gold Copper Silver

Mineral Criteria 1. Naturally occurring – not manufactured 2. Inorganic – minerals not made of living things 3. SOLID substance 4. Have a definite chemical composition – may be a single element or combination 5. Orderly crystalline structure -atoms have specific arrangement

MINERAL FORMATION l CRYSTALLIZE FROM MAGMA or LAVA solidification of liquid magma/lava to solid with a definite internal arrangement of atoms into a regular repeating pattern l PRECIPITATE FROM SOLUTION dissolved minerals come out of solution (water) to form solids

Minerals are identified by their Physical Properties l Crystal Form – determines physical properties l Color l Streak l Luster – metallic, non-metallic l Hardness – Mohs Hardness Scale (1 -10) l Cleavage l Fracture l Density

A mineral’s physical properties are controlled by its internal arrangement of atoms regularly repeating, orderly pattern
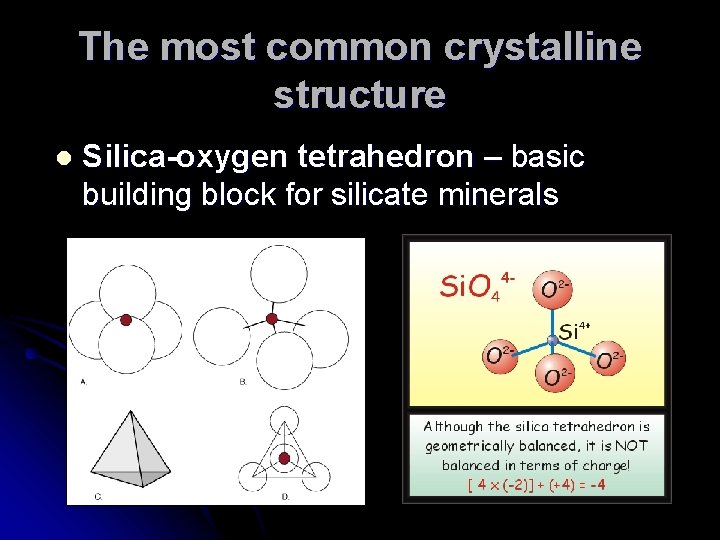
The most common crystalline structure l Silica-oxygen tetrahedron – basic building block for silicate minerals

Silica tetrahedron combine several different ways Five major types of silicate minerals based on their structure A) Isolated tetrahedron B) Single chain C) Double chains D) Sheet silicates E) 3 -D framework silicates


Minerals can have the same chemical composition (Carbon) but different physical properties because of their crystal structure Diamond Graphite

COLOR Color is not usually a definitive property of a mineral. l Some minerals have characteristics colors l Others vary due to chemical differences or impurities (atoms mixed inside the main elements) l However most minerals have a variety of colors. l

Some Colors of Quartz

STREAK l If you rub the sample across a streak plate, it will leave a colored powder. This streak is distinctive for minerals and is used to identify minerals.

Varieties of Hematite – all same color streak

HARDNESS – Mineral’s resistance to scratching or abrasion. Minerals with higher numbers will scratch minerals below

LUSTER – Does this look like it could be made from a metal? l Describes how light reflects off the surface l Categories: Metallic or Non-metallic Metallic – shines like a metal Many non-metallic minerals are SHINY because they are transparent or semi-transparent l Non-metallic: vitreous or glassy; silky; pearly; greasy; waxy, dull; earthy

Examples of metallic luster

More Examples of Metallic Luster Pyrite (Fe. S 2) Galena (Pb. S) PYRITE GALENA

Example of non-metallic luster l Vitreous--quartz

Example of non-metallic luster l Silky--example plagioclase feldspar

Non-metallic and metallic luster – earthy hematite – metallic hematite

Cleavage and Fracture Some minerals split along flat surfaces (called cleavage planes) when struck hard-this is called mineral cleavage l Other minerals break unevenly along rough or curved surfaces--this is called fracture l A few minerals have both cleavage and fracture ( mica ) l

Cleavage – due to weak bonds in the crystal structure Halite (Na. Cl) Fluorite (Ca. F 2) HALITE FLUORITE


Cleavage BIOTITE MUSCOVITE
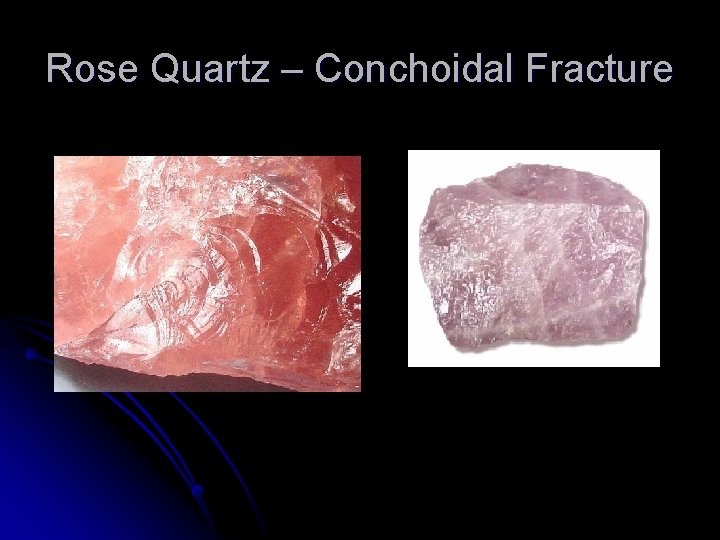
Rose Quartz – Conchoidal Fracture

Conchoidal Fracture - Quartz Obsidian

DENSITY = Mass ÷ Volume

Special or Distinctive Properties: 1. Smell, Feel, Taste (we will not taste samples) 2. Double refraction –when mineral is placed on a picture or letter it produces a double image (mineral must be transparent).

3. Magnetism – tested with common magnet. 4. Fluorescence – test mineral by placing under a black light.

5. Reaction to acid – place mineral in sink – put 12 drops of Hydrochloric acid on mineral sample. You MUST WEAR SAFETY GOGGLES & RINSE THE MINERAL WITH WATER WHEN DONE

Acid Test for Carbonates l Special Characteristics: Carbonates react with dilute HCl and other acids by fizzing or bubbling (releasing CO 2 gas)

Less than a dozen are common in most rocks l l l l l Quartz Feldspar (group) Muscovite (silver white mica) Biotite (black mica) Calcite Pyroxene Olivine Amphibole (group) Magnetite, limonite, and other iron oxides Pyrite

Common uses include: Use back page of ESRT for this information l l l Aluminum--packaging, transport, building Beryllium--gemstones, fluorescent lights Copper--electric cables, wires, switches Feldspar--glass and ceramics Iron--buildings, automobiles, magnets Calcite--toothpaste, construction
- Slides: 36