Atoms Building Blocks of Matter Atoms Smallest particle

Atoms Building Blocks of Matter

Atoms • Smallest particle of matter to have similar properties • Atoms are particles of an element

Subatomic Particles • All atoms are made up of three primary subatomic particles: – Protons • Positively-charged particle in the nucleus • Has a relative mass of 1 Atomic Mass Unit (amu) – Neutrons • Neutrally-charged particle in the nucleus • Has a relative mass of 1 Atomic Mass Unit (amu) • New theories suggest that Neutrons are slightly larger than Protons, and may be made from a Proton and an Electron that have combined

Subatomic Particles – Electrons • Negatively-charged particle outside the nucleus • Has NO relative mass – Actual mass is 1/1936 th of 1 amu – Quarks • Smaller particles that make up Protons, Neutrons, and Electrons • Have their own branch of Theoretical Physics

Properties of Atoms • Atomic Number – Differentiates between elements – Equal to the number of protons • Atomic Mass – Equal to the number of Nucleons (particles in the nucleus, Protons and Neutrons)

Properties of Atoms • Normal atoms are neutral, having the same number of protons and electrons – If an atom gains electrons the atom becomes negatively charged – If an atom loses electrons the atom becomes positively charged

Writing Elements • Write the Symbol • Write the Mass Number at the upper-left of the symbol • Write the Atomic Number at the lowerleft of the symbol • Any charge goes at the upper-right hand corner NOT ALL OF THESE ARE NECESSARY +4 Ag 108 47

Counting Particles • Protons = Atomic Number • Neutrons = Atomic Mass – Atomic Number • Electrons = Protons – ADJUST FOR CHARGE – Negative charge means MORE electrons – Positive Charge means LESS electrons 35 17 Cl

Charge • Atoms will gain, lose, or share electrons to become chemically stable • “Rule of Octet” – Atoms in Groups 1 – 14 will lose electrons down to zero • become positive ions – Atoms in Groups 15, 16, 17, will gain electrons up to eight • become negative ions – Group 18 will not gain, lose, or share electrons • Have 8 electrons anyway

Oxidation Number • The number of electrons an atom will gain, lose, or share in order to become stable • The GROUP number indicates the number of outer-shell electrons – Groups 1, 2 – Groups 13 -18 (except He) • Always 4 or less – Go with the LOWER number of the two possibilities

The Periodic Table Periodic means in a regular, repeating fashion Many early designs were based on use, physical properties, and even spelling

Mendeleev’s Contributions • Organized first working Periodic Table – according to chemical properties and atomic mass • Left gaps for undiscovered elements – He was able to predict many properties of these undiscovered elements based on their location – “Ekasilicon”
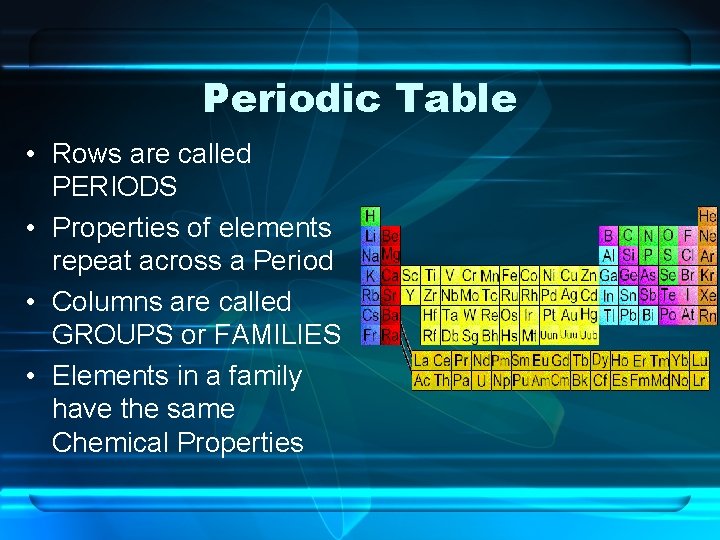
Periodic Table • Rows are called PERIODS • Properties of elements repeat across a Period • Columns are called GROUPS or FAMILIES • Elements in a family have the same Chemical Properties

Group 1 • Alkali Metals • Highly reactive metals • Have one outer shell electron
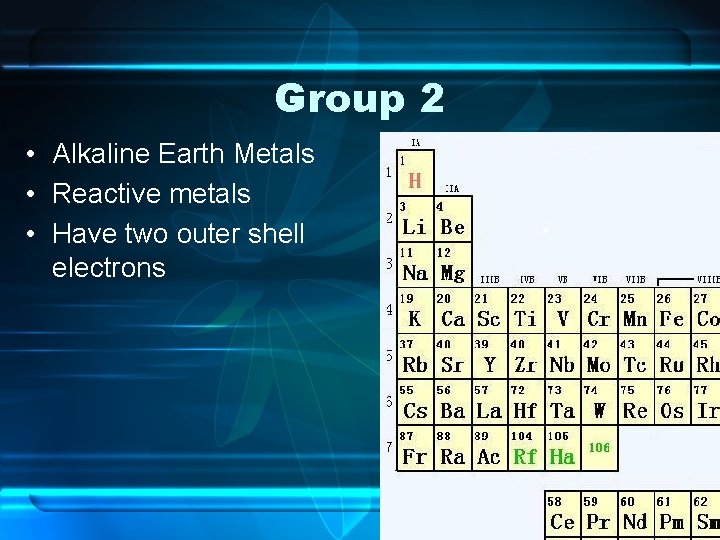
Group 2 • Alkaline Earth Metals • Reactive metals • Have two outer shell electrons
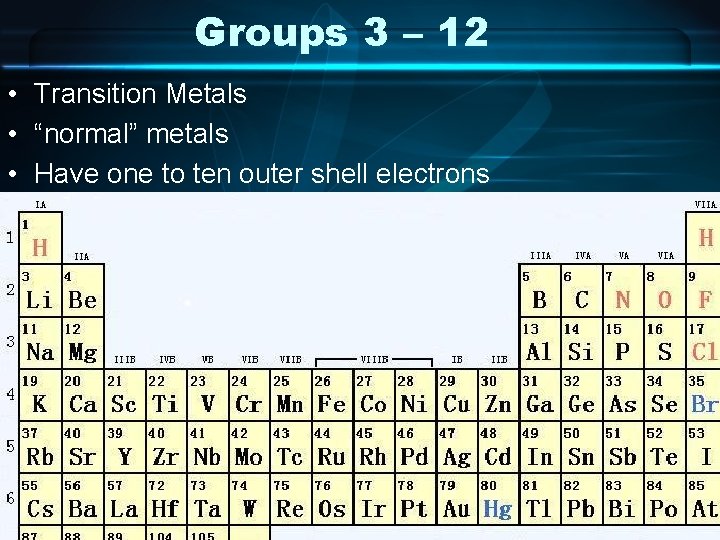
Groups 3 – 12 • Transition Metals • “normal” metals • Have one to ten outer shell electrons

Metals v. Nonmetals • • • METALS Located to the left of the Stair-Step Lustrous Malleable & Ductile Good conductors of heat and electricity Solid at room temperature (except Hg) • • • NONMETALS Located to the right of the Stair-Step Waxy or dull Brittle Poor conductors of heat and electricity (good insulators) Gas at room temperature (some exceptions)

Metalloids • May have properties of metals and nonmetals • Located on the stair-step • Some are semiconductors: they only conduct electricity under certain conditions – This makes metalloids excellent switches for computers

Group 17 • Halogens – Means “Salt Former” • Highly reactive nonmetals • Have seven outer shell electrons

Group 18 • Noble Gases • Completely unreactive nonmetals – Will not bond with any atom • Have a full outer shell of electrons – Eight for all but Helium – He has two outer shell electrons

Chemical Properties • Based on the number of Valence electrons – Electrons on the outside of the atom – Called valence shell electrons, outer shell electrons • Each element in a group has the same number of valence shell electrons – Groups 1, 2 is the group number – Groups 13 -18, drop the “ 1” • Helium has 2 • Groups 3 -12 use a different set of electrons

Drawing a model of an atom • Draw a nucleus • List the number of protons and neutrons in the nucleus • Add energy levels for the number of electrons – An energy level can hold electrons equal to the number of elements in that Period – 2, 8, 8, 18, 32

Differing Atoms of the Same Element • Isotopes – Atoms of an element with a different number of neutrons – Has the same chemical and physical properties as any other isotope of that element
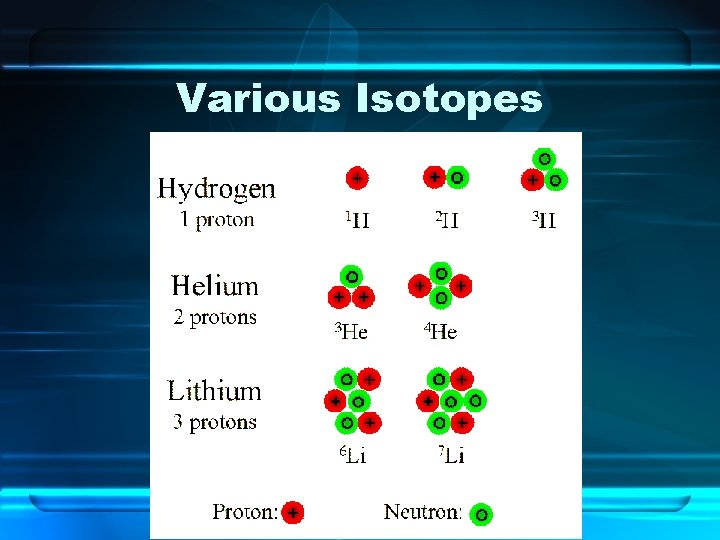
Various Isotopes

Differing Atoms of the Same Element • Allotropes – Named when two or more natural or physical forms of an element exists • Ex. for Carbon: Graphite, Diamond, Buckminsterfullerenes • Ex. for Oxygen: O 2, O 3 - Ozone
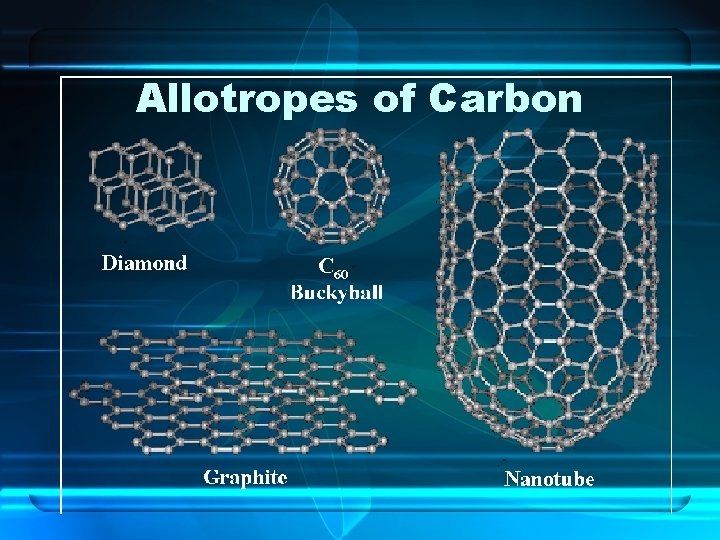
Allotropes of Carbon
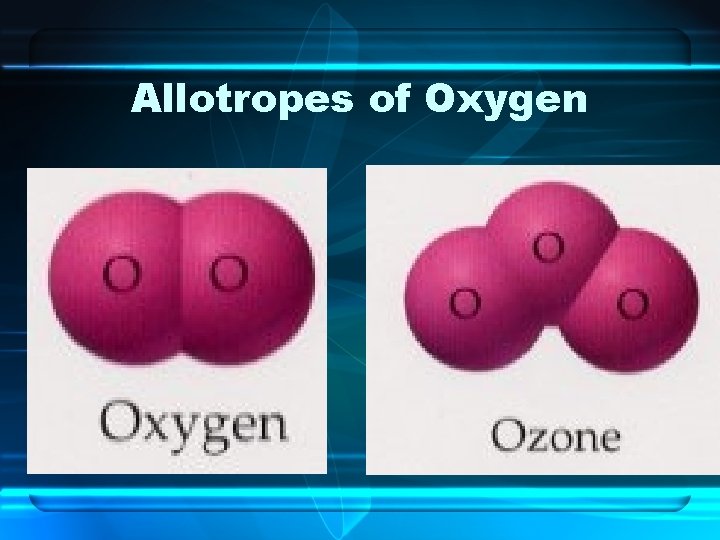
Allotropes of Oxygen

REVIEW • • Atoms Elements Subatomic Particles Atomic Number Atomic Mass Charge Periodic Table Mendeleev • • Periods Groups Families Metals Nonmetals Drawing Atoms Isotopes Allotropes
- Slides: 28